Scalable Preparation of Oxazaspiro Compounds via Mild Alkaline Cyclization for Global Pharmaceutical Supply Chains
Introduction to Advanced Oxazaspiro Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing complex heterocyclic scaffolds, particularly those exhibiting broad-spectrum bioactivity such as oxazaspiro compounds. Patent CN111484507B discloses a groundbreaking preparation method that addresses critical bottlenecks in the synthesis of nitrogen-oxygen heterocyclic spiro-ring compounds, which serve as vital intermediates for cardiovascular and central nervous system therapeutics. This technology leverages a streamlined three-step sequence starting from substituted saturated azacycloalkanes, utilizing mild alkaline hydrolysis followed by condensation and final cyclization. Unlike traditional approaches that rely on extreme conditions, this innovation offers a pragmatic solution for generating high-purity intermediates like Compound I-1, which are essential for developing ROMK inhibitors and muscarinic antagonists. By shifting away from cryogenic and toxic reagent dependencies, this method establishes a new benchmark for efficiency in fine chemical manufacturing.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of oxygen and azaspiro frameworks has been plagued by severe operational constraints that hinder industrial adoption. Prior art, such as the synthesis of Compound IX reported in Chemical Communications, necessitates the use of lithium bis(trimethylsilyl)amide (LiHMDS) at cryogenic temperatures of -78°C, imposing immense energy burdens and requiring specialized cooling infrastructure. Furthermore, this route involves ozonolysis, a hazardous oxidation step that complicates post-treatment and poses significant safety risks due to the instability of ozone and intermediate peroxides. Another documented pathway for Compound I-1 relies on osmium tetroxide (OsO4) for oxidative cleavage, a reagent known for its extreme toxicity and high cost, alongside a disappointingly low overall yield of merely 7.7% across the first three steps. These factors collectively render conventional methods economically unviable and environmentally unsustainable for large-scale production.
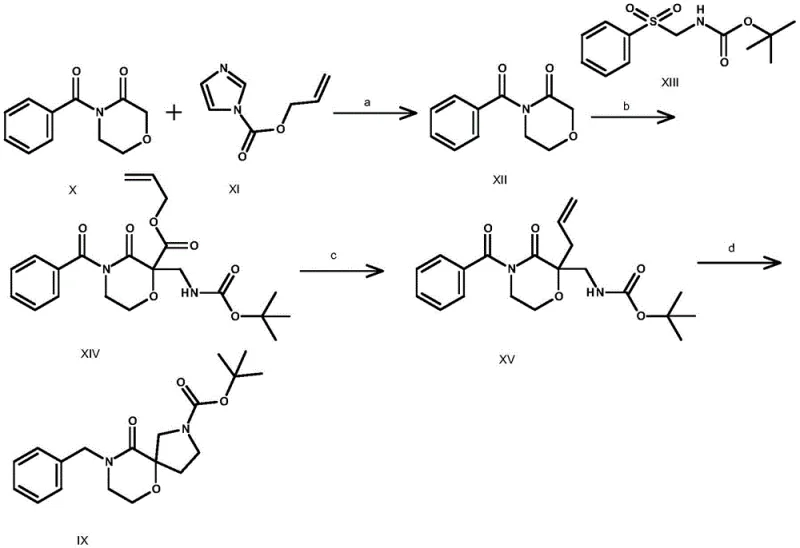
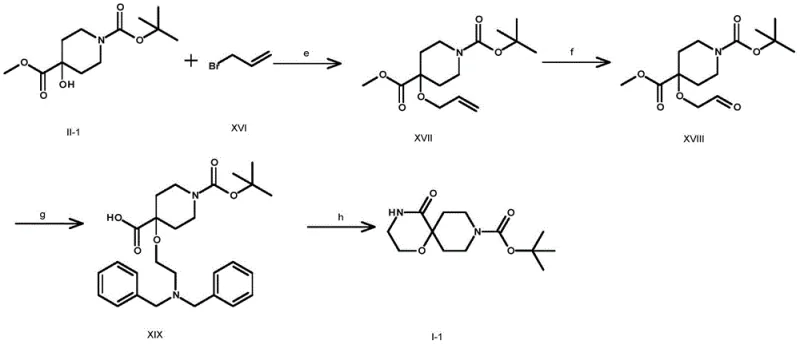
The Novel Approach
In stark contrast, the methodology outlined in CN111484507B introduces a paradigm shift by employing readily available alkaline reagents and standard coupling chemistry to achieve the same structural complexity. The process initiates with a straightforward hydrolysis of ester precursors using bases like sodium hydroxide or potassium hydroxide at moderate temperatures ranging from 0°C to 80°C, completely eliminating the need for ultra-low temperature control. The subsequent amidation step utilizes efficient coupling agents such as HATU or EDCI/HOBt to link the acid intermediate with haloalkylamines, ensuring high conversion rates without the generation of heavy metal waste. Finally, the ring-closing step is achieved through base-mediated intramolecular nucleophilic substitution, often facilitated by phase transfer catalysts, which proceeds smoothly at elevated temperatures up to 100°C. This approach not only simplifies the operational workflow but also significantly enhances the safety profile and economic feasibility of the synthesis.
Mechanistic Insights into Alkaline Hydrolysis and Cyclization
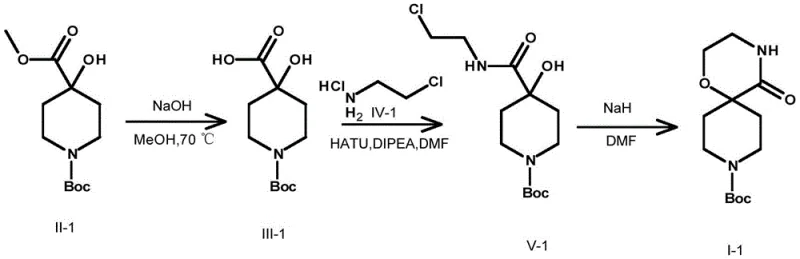
The core of this synthetic strategy lies in the precise manipulation of nucleophilicity and leaving group ability under controlled pH environments. In the initial hydrolysis stage, the hydroxide ion acts as a potent nucleophile attacking the carbonyl carbon of the ester group in Compound II, leading to the formation of the carboxylate anion which is subsequently protonated to yield Compound III. This step is critical for exposing the reactive acid functionality required for the next coupling event. The condensation reaction between Compound III and the halogenated alkylamine (Compound IV) proceeds via an activated ester intermediate formed by the coupling reagent, facilitating the attack of the amine nitrogen to form the stable amide bond in Compound V. The final cyclization is driven by the deprotonation of the hydroxyl group on the spiro-center by a strong base like sodium hydride, generating an alkoxide that intramolecularly displaces the halogen atom on the side chain. This intramolecular SN2 reaction is entropically favored and results in the formation of the rigid oxazaspiro ring system found in Compound I.
Impurity control is inherently managed through the mildness of the reaction conditions, which suppresses competing elimination reactions and over-oxidation often seen in harsher protocols. The use of specific bases and phase transfer catalysts, such as 18-crown-6 or tetrabutylammonium bromide, further enhances selectivity by solubilizing the ionic species in organic solvents, ensuring that the cyclization occurs exclusively at the desired position. Additionally, the avoidance of transition metals like palladium or osmium eliminates the risk of metal contamination, a critical quality attribute for pharmaceutical intermediates intended for human use. The robustness of this mechanism allows for consistent reproducibility, as evidenced by the high purity levels (up to 97.0%) achieved in the experimental examples without the need for complex chromatographic purifications.
How to Synthesize Oxazaspiro Compounds Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control during the coupling and cyclization phases to maximize yield and minimize byproduct formation. The protocol is designed to be flexible, accommodating various protecting groups such as Boc or Cbz on the nitrogen atom, which allows for downstream functionalization if necessary. Operators should ensure that the hydrolysis step is driven to completion, typically monitored by TLC, before proceeding to the amidation to prevent the carryover of unreacted ester which could complicate purification. The final cyclization step benefits from the addition of iodide salts which can act as catalysts by converting chloro-leaving groups into more reactive iodo-species in situ. For a detailed breakdown of the specific reagent quantities and workup procedures demonstrated in the patent examples, please refer to the standardized guide below.
- Hydrolyze substituted saturated azacycloalkane (Compound II) under alkaline conditions using bases like NaOH or KOH to obtain the carboxylic acid intermediate (Compound III).
- Condense Compound III with a halogenated alkylamine salt (Compound IV) using coupling agents such as HATU or EDCI/HOBt to form the amide precursor (Compound V).
- Perform intramolecular cyclization of Compound V under basic conditions using sodium hydride or potassium tert-butoxide at elevated temperatures to close the oxazaspiro ring (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis method represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with complex heterocyclic intermediates. The elimination of cryogenic requirements removes the dependency on specialized low-temperature reactors and the high energy consumption associated with maintaining -78°C environments, leading to substantial operational expenditure reductions. Furthermore, the removal of toxic and regulated substances like osmium tetroxide and ozone simplifies waste disposal protocols and reduces the regulatory burden on manufacturing facilities, thereby enhancing overall site compliance and sustainability metrics. The use of commodity chemicals such as sodium hydroxide and standard coupling reagents ensures a stable and resilient supply chain, less susceptible to the volatility often seen with specialty organometallic catalysts.
- Cost Reduction in Manufacturing: The replacement of expensive organolithium reagents and precious metal catalysts with inexpensive inorganic bases and organic coupling agents drives down the raw material cost per kilogram significantly. By avoiding the need for specialized cryogenic equipment and hazardous gas handling systems for ozone, capital expenditure for plant setup is also drastically reduced, allowing for more competitive pricing models for the final API intermediates.
- Enhanced Supply Chain Reliability: Sourcing of key reagents like HATU, EDCI, and simple alkali metals is globally stable, unlike niche catalysts which may face supply disruptions. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without the need for highly specialized infrastructure, ensuring continuity of supply even during regional logistical challenges or facility maintenance periods.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of heavy metals make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The simplified workup procedures, which often involve standard extraction and crystallization rather than complex chromatography, reduce solvent consumption and waste generation, aligning with green chemistry principles and facilitating smoother environmental permitting processes for expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazaspiro synthesis technology, derived directly from the patent specifications and comparative analysis with prior art. These insights are intended to clarify the operational benefits and quality assurances provided by this method for potential partners and licensees. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production portfolios for cardiovascular and CNS drug candidates.
Q: How does this new method improve safety compared to prior art routes?
A: The novel method eliminates the need for cryogenic conditions (-78°C) and hazardous reagents like ozone and osmium tetroxide found in previous literature, significantly reducing operational risks and equipment costs.
Q: What represents the key cost-saving factor in this synthesis route?
A: By utilizing common alkaline bases and standard coupling reagents instead of expensive organolithium reagents and transition metal catalysts, the process drastically reduces raw material costs and simplifies waste treatment.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (0-100°C) and avoid ultra-low temperatures and toxic heavy metals, making the pathway highly adaptable for multi-kilogram to ton-scale manufacturing in standard reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazaspiro Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes for high-value pharmaceutical intermediates like oxazaspiro compounds. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with stringent purity specifications. We leverage our rigorous QC labs to guarantee that every batch meets the exacting standards required for downstream API synthesis, minimizing the risk of delays in your drug development timeline. Our commitment to technological excellence allows us to offer customized manufacturing solutions that optimize both cost and quality for our global clientele.
We invite you to collaborate with us to leverage this advanced preparation method for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-purity oxazaspiro intermediates while optimizing your overall manufacturing economics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
