Advanced Palladium-Catalyzed Synthesis of Chiral Indole-2,3-Fused Eight-Membered Rings for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Chiral Indole-2,3-Fused Eight-Membered Rings for Commercial Scale-Up
The landscape of asymmetric catalytic synthesis is continually evolving to address the complex structural demands of modern bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112479982A, which discloses a highly efficient preparation method for chiral indole-2,3-octatomic carbocyclic compounds. These unique scaffolds are pivotal in the development of next-generation therapeutics, yet their synthesis has historically been fraught with challenges. The patented technology leverages a sophisticated palladium-catalyzed enamine isomerization and intramolecular asymmetric arylation strategy to construct these intricate architectures. By utilizing easily prepared 2-(2-iodoarylamino)cyclooct-1-ene-1-carboxylate derivatives as starting materials, this method achieves the rapid construction of the target core in a single operational step. This represents a paradigm shift for reliable pharmaceutical intermediate suppliers seeking to access these valuable motifs without the prohibitive costs associated with multi-step classical routes.
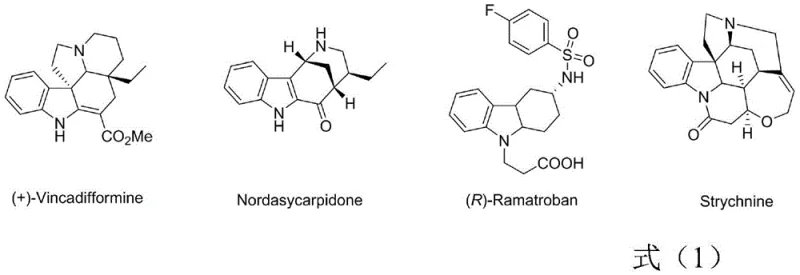
The biological significance of indole-fused ring systems cannot be overstated, as evidenced by their ubiquity in potent natural products and pharmaceutical agents. As illustrated in the structural diversity of compounds like (+)-Vincadifformine, Nordasycarpidone, and Strychnine, the fusion of an indole core with carbocyclic rings often imparts critical three-dimensional rigidity and specific binding affinity required for biological activity. However, while five and six-membered fused indoles are well-explored, the eight-membered variants remain a synthetic frontier. The ability to access these larger ring systems with high stereocontrol opens new avenues for drug discovery, particularly for targets where conformational flexibility is key. This patent provides the chemical toolkit necessary to explore this underutilized chemical space effectively.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral indole-2,3-fused ring compounds has relied heavily on strategies that utilize pre-formed indole substrates. Conventional approaches typically involve asymmetric cycloaddition, asymmetric aromatic hydrogenation, or asymmetric allylation reactions catalyzed by transition metals or organocatalysts. For instance, prior art describes the use of prolinol-catalyzed Diels-Alder reactions or platinum-BIPHEP complex-catalyzed hydrogenations. While effective for generating five or six-membered fused rings, these strategies face severe steric and entropic barriers when attempting to close larger rings. The nucleophilicity of the indole ring, often exploited in these traditional methods, is insufficient to drive the formation of eight-membered rings efficiently. Consequently, the synthesis of chiral indole-2,3-fused eight-membered rings has remained largely unrealized, creating a bottleneck in the supply chain for researchers targeting this specific pharmacophore.
The Novel Approach
The methodology outlined in patent CN112479982A circumvents these limitations through a clever redesign of the synthetic precursor. Instead of starting with a branched substituted indole, the process begins with a 2-(2-iodoarylamino)cyclooct-1-ene-1-carboxylate derivative. This strategic shift allows the reaction to proceed via a palladium-catalyzed cascade involving enamine isomerization followed by intramolecular asymmetric arylation. This tandem process constructs a new ring and a chemical bond simultaneously while establishing a chiral center at the C2 benzylic position of the indole. The result is a direct, one-step entry into the chiral indole-2,3-octatomic carbocyclic scaffold. This approach not only simplifies the synthetic sequence but also offers exceptional control over stereochemistry, addressing the critical need for high-purity pharmaceutical intermediates in early-stage drug development.

Mechanistic Insights into Palladium-Catalyzed Enamine Isomerization and Intramolecular Asymmetric Arylation
The success of this transformation hinges on the precise interplay between the palladium catalyst, the chiral ligand, and the silver salt additive. The reaction mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the substrate. This is followed by a crucial enamine isomerization step, which rearranges the double bond within the eight-membered ring to generate a reactive nucleophilic species in situ. The chiral ligand, selected from a library of sophisticated phosphines such as (S)-BINAP, (S)-SegPhos, or (S)-DiFluorPhos, creates a sterically defined environment around the metal center. This chiral pocket dictates the facial selectivity of the subsequent intramolecular arylation, ensuring the formation of the desired enantiomer. The silver salt plays a dual role, acting as a halide scavenger to regenerate the active cationic palladium species and potentially assisting in the activation of the nucleophile. This synergistic catalytic system operates effectively across a temperature range of room temperature to 100°C, demonstrating remarkable robustness.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise constructions. By forming the ring and the stereocenter in a single catalytic cycle, the potential for generating diastereomeric impurities associated with intermediate isolation is eliminated. The reaction conditions are mild enough to preserve sensitive functional groups, yet vigorous enough to drive the cyclization to completion. Experimental data indicates that the process tolerates a wide array of substituents, including electron-donating alkoxy groups and electron-withdrawing halogens like fluorine and chlorine. This broad substrate scope suggests that the catalytic cycle is resilient to electronic variations on the aryl ring, a critical factor for cost reduction in pharmaceutical intermediate manufacturing where diverse analog libraries must be synthesized rapidly. The high yields and enantiomeric ratios observed confirm that the transition state is tightly controlled, minimizing side reactions such as beta-hydride elimination or homocoupling.
How to Synthesize Chiral Indole-2,3-Octatomic Carbocyclic Compound Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the catalytic components and the choice of solvent. The standard protocol involves charging a reaction vessel with the iodo-precursor, a palladium source such as bis(dibenzylideneacetone)palladium or palladium acetate, and a chiral diphosphine ligand. A silver salt, typically silver phosphate or silver carbonate, is added to facilitate the catalytic turnover. The reaction is conducted in solvents like tetrahydrofuran, toluene, or dichloromethane, with concentrations optimized to balance reaction rate and solubility. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 2-(2-iodoarylamino)cyclooct-1-ene-1-carboxylate derivative, palladium catalyst, chiral ligand, and silver salt in an organic solvent.
- Heat the reaction mixture to a temperature between room temperature and 100°C, maintaining the reaction for 0.1 to 72 hours depending on substrate reactivity.
- Upon completion, quench with acetic acid and ice water, extract with ethyl acetate, wash with saturated brine, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical elegance. The primary value proposition lies in the drastic simplification of the supply chain for these complex intermediates. Traditional routes to fused indole systems often require multiple protection/deprotection steps and the sourcing of exotic chiral pool starting materials. In contrast, this method utilizes readily available 2-(2-iodoarylamino)cyclooct-1-ene-1-carboxylate derivatives, which can be synthesized from commodity chemicals. This shift significantly reduces the dependency on volatile specialty material markets and enhances supply continuity. Furthermore, the operational simplicity of the reaction—requiring only standard heating and stirring equipment—lowers the barrier for contract manufacturing organizations (CMOs) to adopt the process, thereby increasing the number of qualified vendors in the supply base.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by its atom economy and step efficiency. By consolidating ring formation and stereocenter installation into a single step, the process eliminates the cumulative yield losses inherent in multi-step syntheses. The removal of expensive chiral auxiliaries or resolution steps further drives down the cost of goods sold (COGS). Additionally, the use of catalytic amounts of palladium and ligands, rather than stoichiometric chiral reagents, minimizes the consumption of high-value materials. The mild reaction conditions also translate to lower energy consumption, as the process does not require cryogenic temperatures or extreme pressures. These factors collectively contribute to a more cost-effective manufacturing profile, allowing for competitive pricing in the final API market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, a critical metric for supply chain stability. The tolerance for various functional groups means that a single platform technology can be applied to generate a wide range of analogs without needing to re-optimize the entire process for each new derivative. This flexibility allows manufacturers to respond quickly to changing R&D demands. Moreover, the post-treatment workup is straightforward, involving simple aqueous quenching and extraction, which reduces the turnaround time between batches. This efficiency supports reducing lead time for high-purity pharmaceutical intermediates, enabling faster progression through clinical trial phases.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous reagents. The solvents used, such as THF and toluene, are standard in the industry and can be readily recovered and recycled, aligning with green chemistry principles. The high selectivity of the reaction minimizes the generation of difficult-to-separate byproducts, simplifying waste stream management. This environmental compatibility is increasingly important for meeting regulatory standards in major pharmaceutical markets. The ability to scale this process safely and sustainably ensures a long-term, reliable supply of these critical building blocks for the global healthcare industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for effective technology transfer and process optimization.
Q: What is the primary advantage of this palladium-catalyzed method over traditional synthesis routes?
A: This method allows for the one-step construction of both a ring and a chemical bond while establishing a chiral center at the C2 benzylic position, overcoming the historical difficulty of synthesizing eight-membered fused rings compared to five or six-membered analogs.
Q: What types of substrates are compatible with this asymmetric arylation process?
A: The process demonstrates broad substrate applicability, tolerating various substituents on the aryl ring including alkyl, alkoxy, and halogen groups such as fluorine, chlorine, and bromine.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the reaction utilizes readily available raw materials, operates under mild conditions (room temperature to 100°C), and involves simple post-treatment steps like extraction and chromatography, making it highly amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indole-2,3-Octatomic Carbocyclic Compound Supplier
The development of complex chiral scaffolds like indole-2,3-fused eight-membered rings requires a partner with deep technical expertise and a commitment to quality. NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in transition metal catalysis and asymmetric synthesis, ensuring that the transition from laboratory bench to pilot plant is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications. Our infrastructure is designed to handle sensitive catalytic reactions with precision, ensuring reproducibility and safety at every scale.
We invite you to collaborate with us to leverage this innovative synthetic technology for your drug discovery programs. Whether you require custom synthesis of specific analogs or scale-up of the core scaffold, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this efficient palladium-catalyzed method can accelerate your timeline and optimize your budget.
