Advanced X-Binaphane Ligand Technology for High-Efficiency Asymmetric Catalysis and Commercial API Production
Introduction to Next-Generation Chiral Ligand Technology
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for higher purity and efficiency in the production of active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN115340572B, which introduces a novel class of bisphosphine ligands featuring a unique xanthene skeleton combined with a binaphthyl framework. This innovation, specifically exemplified by the X-Binaphane ligand, addresses critical limitations in stereoselectivity and catalyst stability that have long challenged process chemists. By integrating a rigid xanthene backbone with the proven chiral environment of binaphthyl units, this technology offers a robust solution for the asymmetric hydrogenation of challenging substrates like cyclic imines. For R&D directors and procurement strategists, understanding the structural advantages and synthetic accessibility of this ligand is paramount for optimizing current catalytic processes and securing a reliable supply chain for high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional bisphosphine ligands, while foundational to modern asymmetric synthesis, often suffer from inherent geometric constraints that limit their efficacy in specific transformation classes. Many established ligands fail to provide an optimal bite angle or chiral pocket depth necessary for discriminating between enantiotopic faces of bulky or electronically complex substrates. Furthermore, the synthesis of highly specialized ligands frequently involves multi-step sequences with low overall yields, reliance on expensive chiral pool starting materials, or the use of air-sensitive intermediates that complicate handling and storage. These factors collectively contribute to elevated manufacturing costs and supply chain vulnerabilities, particularly when scaling from gram-scale laboratory optimization to multi-kilogram commercial production. The inability to fine-tune the P-M-P coordination angle precisely often results in suboptimal turnover numbers and lower enantiomeric excess, necessitating costly recrystallization steps to meet stringent pharmaceutical purity specifications.
The Novel Approach
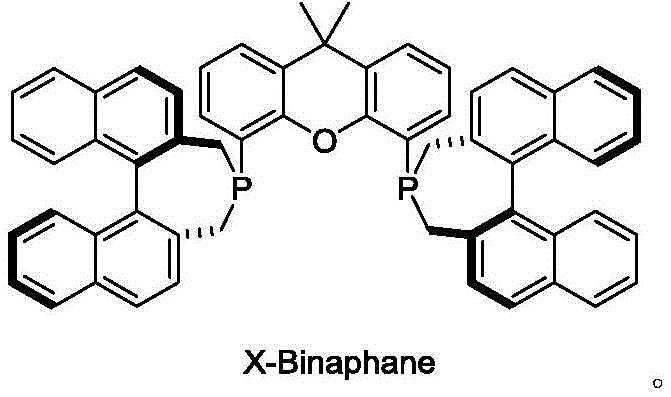
The approach detailed in patent CN115340572B represents a paradigm shift by engineering a ligand architecture that maximizes steric control while maintaining synthetic simplicity. The X-Binaphane ligand leverages the rigidity of the xanthene scaffold to enforce a specific geometry around the metal center, ensuring that the two phosphorus atoms coordinate in a manner that approaches a 180-degree angle. This trans-coordination characteristic, combined with the deep chiral pocket generated by the binaphthyl moieties, creates a highly defined asymmetric environment. This structural precision allows for superior differentiation of substrate enantiofaces, leading to exceptional stereoselectivity even in difficult reductions. Moreover, the synthetic route utilizes commodity chemicals and straightforward coupling reactions, drastically reducing the barrier to entry for large-scale manufacturing and offering a distinct advantage over more convoluted ligand syntheses that rely on scarce natural products.
Mechanistic Insights into Ir-Catalyzed Asymmetric Hydrogenation
The exceptional performance of the X-Binaphane ligand stems from its ability to modulate the electronic and steric properties of the transition metal center, typically iridium or rhodium. When complexed with a metal precursor such as [Ir(COD)Cl]2, the ligand forms a cationic species where the wide P-M-P angle facilitates the coordination of the substrate and hydrogen in a highly organized transition state. The binaphthyl groups project into the coordination sphere, creating a "chiral pocket" that effectively shields one face of the incoming imine substrate. This steric hindrance forces the hydride transfer to occur exclusively from the less hindered face, thereby dictating the absolute configuration of the resulting amine product. The rigidity of the xanthene backbone prevents conformational flexibility that could otherwise lead to non-selective background reactions, ensuring that the catalytic cycle proceeds with high fidelity.
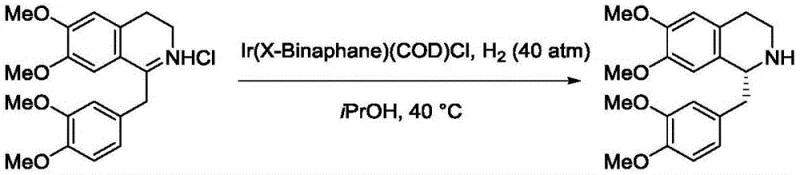
In practical applications, this mechanistic advantage translates directly to process efficiency. For instance, in the asymmetric hydrogenation of dihydropapaverine derivatives, the Ir-X-Binaphane complex has demonstrated the capability to achieve conversion rates exceeding 99% with enantiomeric excess values reaching up to 98-99%. This level of performance is critical for the synthesis of polyhydroisoquinoline alkaloids, which serve as key intermediates for potent pharmaceuticals including muscle relaxants and antitussives. The ability to obtain such high optical purity directly from the reaction mixture minimizes downstream purification burdens, reducing solvent consumption and waste generation. This aligns perfectly with the industry's push towards greener chemistry and more sustainable manufacturing practices, making the X-Binaphane system an attractive candidate for replacing legacy catalysts in existing production lines.
How to Synthesize X-Binaphane Efficiently
The synthesis of the X-Binaphane ligand is designed for operational robustness, avoiding the need for cryogenic conditions or ultra-high vacuum techniques that often hinder scale-up. The process begins with the functionalization of a 9,9-dimethylxanthene core, which is subjected to double lithiation followed by iodination to generate a reactive diiodide intermediate. This intermediate is then converted into a diphosphine precursor through a sequence involving halogen-lithium exchange and subsequent reduction, yielding a stable diphosphine building block. The final and most critical step involves the coupling of this xanthene-derived diphosphine with a 2,2'-dichloromethyl-1,1'-binaphthyl derivative. This nucleophilic substitution reaction, typically mediated by a strong base like sodium hydride in tetrahydrofuran, constructs the final P-C bonds that link the chiral binaphthyl arms to the central xanthene hub. The detailed standardized synthetic steps for this high-efficiency route are provided in the guide below.
- Preparation of the xanthene backbone via double lithiation of 9,9-dimethylxanthene followed by iodination to form the diiodide intermediate.
- Conversion of the diiodide to a diphosphine precursor through halogen-lithium exchange and reaction with diethyl chlorophosphite, followed by reduction with LiAlH4.
- Final coupling of the diphosphine xanthene derivative with 2,2'-dichloromethyl-1,1'-binaphthyl under basic conditions to yield the target X-Binaphane ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the X-Binaphane ligand technology offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the substantial cost reduction in API manufacturing enabled by the ligand's accessible synthetic route. Unlike many bespoke chiral ligands that depend on expensive, naturally sourced chiral pools, X-Binaphane is constructed from readily available industrial chemicals. This decoupling from volatile biological supply chains ensures price stability and consistent availability, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the high catalytic activity allows for lower catalyst loading, meaning less precious metal is required per kilogram of product, which directly impacts the bill of materials and reduces the burden on metal scavenging processes downstream.
- Cost Reduction in Manufacturing: The synthetic pathway for X-Binaphane eliminates the need for complex resolution steps or rare earth catalysts often associated with traditional ligand production. By utilizing straightforward coupling reactions and common solvents, the overall process mass intensity is significantly lowered. This efficiency translates into a lower cost of goods sold (COGS) for the final chiral intermediate, allowing pharmaceutical companies to maintain healthier margins or pass savings on to healthcare providers. Additionally, the high selectivity reduces the formation of unwanted diastereomers, minimizing the loss of valuable starting materials and further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as xanthene derivatives and binaphthyl precursors ensures a resilient supply chain. These materials are produced by multiple global suppliers, reducing dependency on single-source vendors. This diversification is crucial for maintaining business continuity in the face of geopolitical disruptions or logistical bottlenecks. Moreover, the stability of the ligand and its metal complexes simplifies storage and transportation requirements, as they do not demand extreme cold chain logistics, thereby reducing warehousing costs and expanding the potential supplier base to include regional manufacturers who can offer faster lead times.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as refluxing in THF or using standard inert atmosphere techniques, are fully compatible with existing multipurpose reactor infrastructure found in most CDMO facilities. This compatibility facilitates rapid technology transfer from lab to plant without the need for capital-intensive equipment upgrades. From an environmental perspective, the high atom economy and reduced solvent usage associated with the high-yield reactions contribute to a smaller environmental footprint. This alignment with green chemistry principles helps companies meet increasingly stringent regulatory standards regarding waste disposal and emissions, avoiding potential fines and enhancing corporate sustainability profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of X-Binaphane technology in industrial settings. These insights are derived directly from the experimental data and embodiments disclosed in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this ligand into your specific process workflow.
Q: What distinguishes the X-Binaphane ligand from conventional bisphosphine ligands?
A: X-Binaphane features a unique combination of a xanthene skeleton and a binaphthyl backbone, creating a P-M-P coordination angle closer to 180 degrees and a deeper chiral pocket, which significantly enhances stereoselectivity in asymmetric hydrogenation compared to traditional ligands.
Q: Is the synthesis of X-Binaphane scalable for industrial production?
A: Yes, the synthetic route described in patent CN115340572B utilizes readily available starting materials and standard organic transformations such as lithiation and nucleophilic substitution, making it highly suitable for commercial scale-up without requiring exotic reagents.
Q: What types of reactions can the Ir-X-Binaphane complex catalyze?
A: The iridium complex of X-Binaphane is particularly effective for the asymmetric hydrogenation of cyclic imines, such as dihydropapaverine derivatives, achieving excellent yields and enantiomeric excess (ee) values up to 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable X-Binaphane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands like X-Binaphane in accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ligand or catalytic intermediate meets the highest international standards. We understand that consistency is key in pharmaceutical manufacturing, and our robust quality management systems are designed to deliver that reliability consistently.
We invite you to collaborate with our technical team to explore how this innovative ligand technology can optimize your specific synthesis challenges. Whether you require a Customized Cost-Saving Analysis for your current catalytic process or need to evaluate the feasibility of switching to the X-Binaphane system, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and pilot-scale quotations. Let us help you secure a competitive edge through superior catalytic performance and supply chain security.
