Advanced Oxidation Technology for O-Nitro-P-Methylsulfonyl Benzoic Acid: A Strategic Upgrade for Global Pharmaceutical Supply Chains
Advanced Oxidation Technology for O-Nitro-P-Methylsulfonyl Benzoic Acid: A Strategic Upgrade for Global Pharmaceutical Supply Chains
The global demand for high-purity pharmaceutical intermediates continues to drive innovation in synthetic methodology, particularly for complex aromatic acids bearing multiple electron-withdrawing groups. A pivotal advancement in this domain is documented in Chinese Patent CN101503383B, which discloses a novel preparation method for o-nitro-p-methylsulfonyl benzoic acid, a critical building block in the synthesis of various bioactive molecules. This technology represents a paradigm shift from traditional, hazardous oxidation protocols to a cleaner, more efficient process utilizing in-situ generated persulfuric acid. By leveraging the unique oxidative properties of persulfuric acid formed from the mixture of hydrogen peroxide and oleum, this method addresses longstanding challenges regarding catalyst toxicity, waste management, and raw material efficiency. For R&D directors and procurement strategists seeking a reliable pharma intermediates supplier, understanding the mechanistic advantages of this patent is essential for securing a sustainable and cost-effective supply chain for high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzoic acids containing strong electron-withdrawing substituents, such as nitro and sulfonyl groups, has relied heavily on aggressive oxidation conditions that pose significant environmental and operational risks. The most prevalent conventional method involves the use of concentrated nitric acid as the oxidant, often necessitating the presence of vanadium pentoxide (V2O5) as a catalyst to overcome the deactivating effects of the substituents on the aromatic ring. While United States Patent 5,424,481 demonstrated that this approach could achieve yields exceeding 80%, the process is fraught with severe drawbacks that modern manufacturing facilities strive to eliminate. The oxidation with nitric acid inevitably generates large volumes of nitrogen dioxide (NO2) gas, a toxic reddish-brown pollutant that requires extensive scrubbing systems to meet environmental regulations. Furthermore, the reliance on vanadium pentoxide introduces a highly toxic heavy metal into the process stream, complicating waste acid treatment and requiring rigorous purification steps to ensure the final product meets stringent heavy metal specifications required for pharmaceutical applications. Alternative "green" methods utilizing direct hydrogen peroxide oxidation have also been explored, such as those described in European Patent WO 2004/058698A1; however, these processes often suffer from poor atom economy, requiring up to ten times the stoichiometric amount of hydrogen peroxide due to its rapid decomposition and lower oxidative potential, thereby rendering them economically unviable for large-scale production.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN101503383B introduces a sophisticated oxidation strategy that utilizes fresh persulfuric acid (H2S2O8) generated in situ from the reaction of hydrogen peroxide and oleum. This novel approach fundamentally alters the reaction landscape by creating an oxidizing species with a significantly higher electrode potential than hydrogen peroxide alone, thereby enabling the efficient oxidation of the methyl group to a carboxylic acid without the need for toxic vanadium catalysts. The process cleverly mitigates the instability of hydrogen peroxide by reacting it with oleum at low temperatures, which not only generates the active persulfuric acid but also reduces the water content that typically accelerates peroxide decomposition. This results in a much higher utilization rate of the oxidant, drastically reducing the molar excess required compared to direct hydrogen peroxide methods. Moreover, the byproduct of the oxidation is sulfuric acid, which is compatible with the reaction medium and can potentially be recycled or reused in upstream processes, aligning perfectly with the principles of green chemistry. For a cost reduction in fine chemical manufacturing, this elimination of heavy metal catalysts and the reduction in oxidant consumption translates directly into lower raw material costs and simplified downstream processing, making it an attractive option for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into In-Situ Persulfuric Acid Oxidation
The core of this technological breakthrough lies in the generation and reactivity of persulfuric acid, a powerful oxidizing agent formed through the interaction of hydrogen peroxide and sulfur trioxide present in oleum. The reaction mechanism begins with the careful mixing of 50% hydrogen peroxide and oleum (containing 20-60% free SO3) at temperatures maintained below 30°C to prevent thermal runaway and premature decomposition. Under these conditions, the sulfur trioxide reacts with hydrogen peroxide to form persulfuric acid (H2S2O8) according to the equation 2H2SO4 + H2O2 → H2S2O8 + 2H2O, although the actual pathway involves the direct reaction of SO3 with H2O2. The resulting persulfuric acid possesses a higher standard electrode potential than hydrogen peroxide, making it thermodynamically capable of oxidizing the deactivated methyl group on the o-nitro-p-methylsulfonyl toluene substrate. This enhanced reactivity allows the reaction to proceed efficiently at moderate temperatures (80-130°C) without the need for external catalytic promotion by transition metals. The choice of solvent is also critical; the substrate is dissolved in 70% sulfuric acid, a concentration carefully selected to ensure complete solubility of the organic starting material while minimizing the risk of electrophilic aromatic sulfonation, which could occur in higher concentrations of sulfuric acid or oleum. This balance ensures that the oxidation occurs selectively at the benzylic position, preserving the integrity of the aromatic ring and the existing nitro and sulfonyl substituents.
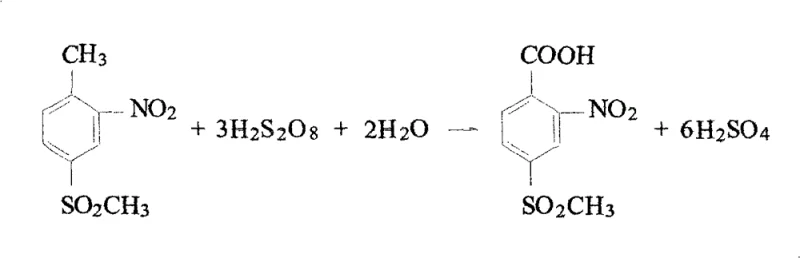
From a mechanistic standpoint, the stoichiometry of the reaction dictates that three moles of hydrogen peroxide are theoretically required to oxidize one mole of the toluene derivative to the corresponding benzoic acid, as illustrated in the reaction scheme. However, practical implementation often requires a molar ratio of hydrogen peroxide to substrate ranging from 9:1 to 15:1 to drive the reaction to completion and account for side reactions or decomposition. Despite this excess, the in-situ generation of persulfuric acid ensures that the effective concentration of the oxidant remains high throughout the addition period, which is typically conducted over 4 to 5 hours to manage the exothermic nature of the oxidation. The reaction mixture is then cooled to induce crystallization, allowing for the isolation of the product via suction filtration. Impurity control is inherently managed by the absence of metal catalysts, which eliminates the formation of metal-organic complexes that are difficult to remove. The crude product typically achieves a purity of 80% to 95% by liquid chromatography, and further purification via alkaline dissolution and acidification can elevate the purity to 99%, meeting the rigorous standards required for high-purity pharmaceutical intermediates. This level of control over the impurity profile is a key consideration for R&D teams evaluating the feasibility of this route for GMP manufacturing.
How to Synthesize O-Nitro-P-Methylsulfonyl Benzoic Acid Efficiently
The implementation of this oxidation protocol requires precise control over reaction parameters to maximize yield and safety, particularly during the generation of the unstable persulfuric acid intermediate. The process is divided into two distinct stages: the preparation of the oxidant and the subsequent oxidation of the substrate. Operators must adhere to strict temperature controls, ensuring that the mixing of hydrogen peroxide and oleum occurs under cooling conditions to prevent violent decomposition. Once the persulfuric acid is prepared, it is added dropwise to the heated solution of the substrate in sulfuric acid, maintaining the reaction temperature within the optimal window of 80°C to 130°C. Detailed standard operating procedures regarding addition rates, stirring speeds, and quenching protocols are critical for reproducibility. For a comprehensive understanding of the specific operational parameters and safety measures required for laboratory or pilot-scale execution, please refer to the standardized synthesis guide below.
- Preparation of Persulfuric Acid: Mix hydrogen peroxide (50% concentration) with oleum (20-60% free SO3) at low temperature (below 30°C) to generate fresh persulfuric acid (H2S2O8) in situ.
- Substrate Dissolution: Dissolve o-nitro-p-methylsulfonyl toluene in 70% sulfuric acid solution to ensure solubility while preventing unwanted sulfonation of the benzene ring.
- Oxidation Reaction: Slowly add the prepared persulfuric acid to the substrate solution while maintaining a reaction temperature between 80°C and 130°C for 4 to 5 hours, followed by cooling and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the persulfuric acid oxidation method described in CN101503383B offers substantial strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the waste treatment infrastructure required for production. By eliminating the use of vanadium pentoxide, manufacturers avoid the complex and costly processes associated with heavy metal recovery and the disposal of toxic metal-containing waste streams. This not only reduces the direct costs of waste management but also mitigates regulatory risks associated with environmental compliance in increasingly stringent global markets. Furthermore, the avoidance of nitric acid oxidation means that facilities do not need to invest in expensive gas scrubbing systems to handle nitrogen oxide emissions, leading to significant capital expenditure savings and lower operational overheads. The process inherently promotes a cleaner production environment, which is a key metric for sustainability-focused supply chains.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the improved efficiency of the oxidant. Unlike direct hydrogen peroxide oxidation methods that require vast excesses of the reagent due to decomposition, the in-situ generation of persulfuric acid stabilizes the active oxygen species, allowing for a significant reduction in the total amount of hydrogen peroxide consumed per kilogram of product. Although the process uses oleum, the resulting sulfuric acid byproduct can often be integrated back into the process or sold, offsetting raw material costs. Additionally, the removal of the expensive and toxic vanadium catalyst eliminates a major cost center associated with catalyst procurement and the specialized equipment needed to handle it. These factors combine to create a manufacturing route with a markedly lower variable cost structure compared to traditional nitric acid or direct peroxide methods.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the raw materials required for this synthesis—hydrogen peroxide, oleum, and sulfuric acid—are commodity chemicals with robust and stable global supply chains. This contrasts sharply with specialized catalysts or reagents that may be subject to supply disruptions or geopolitical constraints. The simplicity of the reagent profile ensures that production can be sustained continuously without the risk of bottlenecks caused by the unavailability of niche materials. Moreover, the robustness of the reaction conditions allows for flexible manufacturing schedules, enabling suppliers to respond rapidly to fluctuations in market demand for o-nitro-p-methylsulfonyl benzoic acid. This reliability is crucial for downstream pharmaceutical customers who require consistent delivery of high-purity intermediates to maintain their own production timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial application in mind. The use of common reactor materials compatible with sulfuric acid environments facilitates easy scale-up from pilot plants to multi-ton commercial production without the need for exotic metallurgy. The environmental profile of the process is another key scalability driver; with no heavy metals and reduced gaseous emissions, obtaining environmental permits for expansion is significantly streamlined. The ability to recycle mother liquors or utilize the spent acid reduces the overall environmental footprint, aligning with the corporate social responsibility goals of modern chemical enterprises. This ease of scale-up ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable even as volume requirements grow.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common questions regarding the process parameters, safety considerations, and product quality. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for assessing the compatibility of this route with existing manufacturing capabilities and quality assurance frameworks.
Q: Why is persulfuric acid preferred over nitric acid for oxidizing electron-withdrawing substituted toluenes?
A: Persulfuric acid offers a significantly higher electrode potential than hydrogen peroxide alone and avoids the generation of toxic nitrogen dioxide (NO2) gas associated with nitric acid oxidation. Furthermore, it eliminates the need for toxic vanadium pentoxide catalysts, simplifying waste acid treatment and reducing environmental compliance costs.
Q: How does the use of oleum improve the stability of hydrogen peroxide in this process?
A: Mixing hydrogen peroxide with oleum generates persulfuric acid, which effectively increases the concentration of the active oxidizing species while reducing the water content that typically accelerates hydrogen peroxide decomposition. This stabilization allows for a more controlled and efficient oxidation reaction.
Q: What purity levels can be achieved with this oxidation method?
A: The crude product obtained via suction filtration typically exhibits a purity of 80% to 95% as analyzed by liquid chromatography. Subsequent purification steps, such as dissolution in alkali followed by acidification, can yield high-quality crystals with purity reaching 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Nitro-P-Methylsulfonyl Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, greener synthetic routes is critical for the long-term competitiveness of the pharmaceutical industry. Our technical team has extensively analyzed the persulfuric acid oxidation technology and possesses the expertise to implement this method effectively at commercial scales. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, high-volume manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of o-nitro-p-methylsulfonyl benzoic acid meets the exacting standards required for API synthesis. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your operational efficiency.
We invite you to collaborate with us to explore how this innovative oxidation method can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our mastery of this technology can drive value for your organization. Let us be your partner in achieving sustainable and cost-effective chemical manufacturing.
