Revolutionizing Aromatic Sulfonyl Chloride Production: A Continuous Flow Strategy for High-Purity Pharma Intermediates
Revolutionizing Aromatic Sulfonyl Chloride Production: A Continuous Flow Strategy for High-Purity Pharma Intermediates
In the highly competitive landscape of fine chemical manufacturing, the ability to produce critical intermediates with superior purity, safety, and cost-efficiency is paramount. The recent disclosure of patent CN111718286A introduces a transformative industrial production method for aromatic hydrocarbon sulfonyl chlorides bearing electron-donating groups. This technology represents a significant departure from traditional batch processing, leveraging a sophisticated two-step continuous flow protocol that utilizes liquid sulfur trioxide and chlorosulfonic acid in a controlled solvent system. For R&D directors and procurement strategists alike, this patent offers a compelling roadmap for optimizing the synthesis of key pharmaceutical and agrochemical intermediates. By shifting from hazardous batch reactions to a streamlined pipeline continuous reaction system, manufacturers can achieve a qualitative leap in production stability and intrinsic safety. This report delves deep into the technical nuances of this innovation, analyzing how it addresses long-standing pain points in sulfonation chemistry while providing a robust foundation for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of arene sulfonyl chlorides with electron-donating groups has been plagued by significant environmental and safety challenges inherent to traditional batch processes. The conventional approach typically involves the direct reaction of aromatic hydrocarbons with a large excess of chlorosulfonic acid, a method that generates substantial quantities of waste sulfuric acid and waste hydrochloric acid as byproducts. This not only creates a massive burden on waste treatment facilities but also leads to severe pollution issues that are increasingly difficult to manage under modern environmental regulations. Furthermore, the separation of the desired product from the waste acid matrix is notoriously difficult, often requiring complex downstream processing that erodes overall yield and purity. From a safety perspective, the use of large volumes of chlorosulfonic acid in batch reactors poses a high risk; the exothermic nature of the reaction combined with the potential for unorganized hydrogen chloride emissions creates a hazardous working environment. The reliance on intermittent reaction systems also means that large amounts of dangerous chemicals are stored within the system at any one time, increasing the potential severity of any accidental release.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111718286A proposes a refined, two-stage continuous process that fundamentally alters the reaction landscape. The innovation lies in the strategic decoupling of sulfonation and acyl chlorination, utilizing liquid sulfur trioxide for the initial sulfonation step rather than relying solely on chlorosulfonic acid. This approach allows for a homogeneous continuous sulfonation reaction in the presence of a solvent and a positioning agent, which dramatically improves selectivity and reduces side reactions. By employing a continuous dynamic multiphase plug flow reaction system—formed by combining dynamic mixers with pipeline reactors—the process ensures efficient mass and heat transfer, eliminating the hot spots common in batch kettles. Crucially, this new route avoids the generation of hydrogen chloride gas during the main reaction phases, resulting in a much cleaner process profile. The ability to realize pipeline continuous reaction means that the inventory of hazardous reagents is minimized, inherently safer, and easier to control, marking a definitive upgrade over the antiquated batch methodologies.
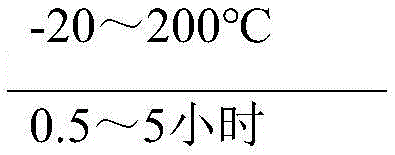
Mechanistic Insights into Solvent-Mediated Continuous Sulfonation and Acyl Chlorination
The core of this technological advancement rests on a precise understanding of reaction kinetics and thermodynamics within a solvent-mediated environment. In the first stage, liquid sulfur trioxide is premixed with a solvent and preheated or cooled to a specific temperature range of -20~200°C before being introduced to the aromatic hydrocarbon. The inclusion of a positioning agent, such as N-fluoro-diphenyl sulfonamide, acetic acid, or ammonium acetate, is critical; it acts to direct the sulfonic acid group to the desired position on the benzene ring, thereby suppressing the formation of unwanted ortho or meta isomers. The solvent system itself plays a dual role: it liquefies the reaction mixture to facilitate continuous flow and modulates the activity of the highly reactive sulfur trioxide. When the semi-finished product is liquid, it can even serve as the solvent, further inhibiting side reactions and enhancing yield. This careful orchestration of reagents ensures that the sulfonation proceeds with high regioselectivity, a factor that is often the bottleneck in producing high-purity intermediates for sensitive pharmaceutical applications.
Following sulfonation, the process transitions seamlessly into the acyl chlorination phase, where the sulfonated intermediate reacts with chlorosulfonic acid in the presence of a catalyst. The patent specifies a variety of effective catalysts, including DMF, triethylamine, pyridine, and 4-dimethylaminopyridine, which greatly promote the conversion of the sulfonic acid group to the sulfonyl chloride. Similar to the first step, this reaction is conducted in a continuous flow system where the chlorosulfonic acid is premixed with a solvent and temperature-controlled between -20~200°C. The continuous nature of this acyl chlorination ensures that the slightly excessive chlorosulfonic acid is efficiently utilized and subsequently hydrolyzed, allowing for easy separation of the acid layer. This mechanistic design not only maximizes the atom economy of the process but also ensures that the final product, the aromatic hydrocarbon sulfonyl chloride, is obtained with exceptional purity after desolventizing. The synergy between the positioning agent in step one and the catalyst in step two creates a robust chemical environment that consistently delivers high-quality output suitable for demanding downstream syntheses.
How to Synthesize Aromatic Sulfonyl Chloride Efficiently
The implementation of this continuous flow synthesis requires a precise adherence to the operational parameters defined in the patent to ensure optimal yield and safety. The process begins with the preparation of the sulfonation feed, where liquid sulfur trioxide and the chosen solvent are mixed and temperature-adjusted before entering the dynamic mixer alongside the aromatic substrate and positioning agent. Following the residence time in the pipeline reactor, the effluent is immediately directed to the acyl chlorination stage, where it meets the catalyst-containing chlorosulfonic acid stream. The detailed standardized synthesis steps, including specific flow rates, residence times, and workup procedures for various substrates like toluene or acetanilide, are critical for reproducibility and are outlined below for technical reference.
- Premix liquid sulfur trioxide with a solvent and preheat to -20~200°C, then add a positioning agent for homogeneous continuous sulfonation with the aromatic hydrocarbon.
- Conduct the acyl chlorination reaction by premixing chlorosulfonic acid with a solvent and catalyst, reacting with the sulfonated product in a continuous pipeline system.
- Hydrolyze excess chlorosulfonic acid, separate the acid layer, and remove the solvent to isolate the high-purity aromatic sulfonyl chloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the process described in CN111718286A offers profound strategic benefits that extend far beyond simple chemical efficiency. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials; by substituting a significant portion of expensive chlorosulfonic acid with liquid sulfur trioxide for the sulfonation step, the overall material cost structure is significantly optimized. Liquid sulfur trioxide is not only more economical on a molar basis but also offers superior atom economy, meaning less waste is generated per unit of product. This reduction in waste translates directly into lower disposal costs and reduced regulatory burden, which are critical factors in the total cost of ownership for fine chemical manufacturing. Furthermore, the elimination of hydrogen chloride gas generation removes the need for extensive scrubbing systems and associated maintenance, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The economic argument for adopting this technology is compelling, primarily driven by the substitution of high-cost reagents with more efficient alternatives. By utilizing liquid sulfur trioxide for the initial sulfonation, the process avoids the need for the massive excess of chlorosulfonic acid required in traditional methods, leading to substantial raw material savings. Additionally, the continuous flow nature of the reaction improves energy efficiency through better heat management, reducing the utility costs associated with heating and cooling large batch reactors. The simplified downstream processing, characterized by easy acid separation and desolventizing, minimizes labor and equipment time, contributing to a leaner and more cost-effective production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this continuous process ensures a more reliable and consistent output of high-purity intermediates. The ability to run the reaction in a pipeline system means that production can be scaled up or down with greater flexibility compared to fixed-volume batch reactors, allowing manufacturers to respond more agilely to market demand fluctuations. Moreover, the reduced dependency on complex additive packages and the use of widely available solvents and reagents mitigate the risk of supply disruptions. The inherent stability of the continuous process also leads to fewer batch failures and off-spec products, ensuring that delivery schedules are met with greater certainty and reducing the need for safety stock inventory.
- Scalability and Environmental Compliance: The scalability of this technology is perhaps its most significant advantage for long-term growth, as the continuous dynamic multiphase plug flow reaction system can be expanded by numbering up reactors rather than scaling up vessel size. This approach maintains the same reaction conditions and safety profile regardless of production volume, facilitating a smoother path from pilot plant to commercial scale. Environmentally, the process aligns perfectly with green chemistry principles by minimizing waste generation and eliminating hazardous gas emissions, making it easier for facilities to maintain compliance with increasingly stringent environmental regulations. The reduced footprint of the pipeline equipment compared to large batch tanks also allows for higher production capacity within existing facility boundaries, maximizing asset utilization.
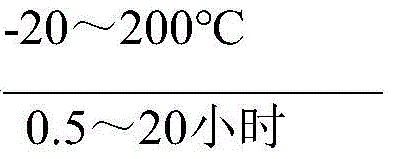
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced sulfonation technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility of adopting this continuous flow strategy for industrial production. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or process optimization within their own manufacturing networks.
Q: How does this new process improve safety compared to traditional batch methods?
A: The novel process utilizes a continuous dynamic multiphase plug flow reaction system which significantly reduces the inventory of dangerous chemicals like liquid sulfur trioxide and chlorosulfonic acid within the reactor at any given time, thereby enhancing intrinsic safety compared to large batch kets.
Q: What are the primary cost drivers reduced in this manufacturing method?
A: By replacing a large portion of expensive chlorosulfonic acid with liquid sulfur trioxide for the initial sulfonation step, raw material costs are drastically lowered. Additionally, the elimination of hydrogen chloride gas generation reduces waste treatment expenses.
Q: Can this method handle aromatic hydrocarbons with strong electron-donating groups?
A: Yes, the process is specifically designed for aromatic hydrocarbons with electron supply groups (such as dialkylamino, alkoxy, or alkyl groups) by utilizing specific positioning agents and solvent systems to control regioselectivity and prevent side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Sulfonyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of cutting-edge synthetic methodologies like the one described in CN111718286A is key to maintaining a competitive edge in the global fine chemical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to industrial reality is seamless and efficient. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that the production of complex aromatic sulfonyl chlorides requires not just chemical expertise but also a deep dedication to safety and environmental stewardship, values that are central to our operational philosophy.
We invite you to collaborate with us to leverage this advanced continuous flow technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this process can optimize your budget without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to jointly explore the full potential of this innovative manufacturing strategy for your supply chain.
