Advanced Imidazo Naphthyridine Compounds for High Efficiency OLED Manufacturing and Supply
Advanced Imidazo Naphthyridine Compounds for High Efficiency OLED Manufacturing and Supply
The rapid evolution of the organic light-emitting diode (OLED) industry demands host materials that surpass the limitations of traditional carbazole-based systems. Patent CN111320626B introduces a groundbreaking class of organic electroluminescent materials based on the imidazo[1,5-a][1,8]naphthyridine scaffold, designed specifically to address the critical bottlenecks of thermal instability and carrier imbalance found in legacy hosts. This technological leap offers a robust solution for next-generation display and lighting applications, providing a foundation for devices with exceptional longevity and color purity. By integrating nitrogen-rich heterocyclic structures, these compounds achieve a superior balance between hole and electron transport, which is essential for maximizing internal quantum efficiency in phosphorescent OLED architectures.
![General chemical structure of imidazo[1,5-a][1,8]naphthyridine derivatives for OLED applications](/insights/img/imidazo-naphthyridine-oled-material-supplier-20260307203736-01.png)
For procurement leaders and supply chain directors, the adoption of this new chemical scaffold represents a strategic opportunity to secure a reliable OLED material supplier capable of delivering high-performance components. The structural versatility allows for extensive customization of the aryl and heteroaryl substituents, enabling fine-tuning of energy levels to match specific dopant requirements. This flexibility ensures that manufacturers can optimize their device stacks for various emission colors, from deep blue to efficient red, without being constrained by the rigid properties of older host materials like CBP. The patent data underscores a clear trajectory towards materials that not only perform better but are also synthesized via scalable, robust chemical pathways suitable for industrial expansion.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on CBP (4,4'-Bis(N-carbazolyl)-1,1'-biphenyl) as a standard host material for phosphorescent OLEDs due to its high triplet energy level. However, CBP exhibits significant drawbacks that hinder the performance and reliability of modern high-resolution displays. Primarily, CBP possesses a low glass transition temperature (Tg) of approximately 62°C, which leads to morphological instability in the emitting layer under the thermal stress of device operation. This crystallization tendency causes dark spot formation and rapid efficiency roll-off, drastically shortening the operational lifetime of the display panel. Furthermore, CBP is predominantly a hole-transporting material with poor electron mobility, creating a severe imbalance in carrier injection that forces the recombination zone to shift, thereby reducing overall device efficiency and increasing driving voltage requirements.
The Novel Approach
The innovative approach detailed in the patent data utilizes an imidazo[1,5-a][1,8]naphthyridine core, which intrinsically offers a more balanced electronic structure compared to simple carbazole dimers. This novel scaffold demonstrates a markedly higher glass transition temperature, with specific embodiments like Compound 29 achieving a Tg of 99°C, ensuring exceptional morphological stability even under prolonged thermal cycling. The incorporation of multiple nitrogen atoms within the fused ring system enhances electron affinity, effectively balancing the hole and electron transport rates within the emissive layer. This equilibrium allows for a broader recombination zone and more efficient exciton formation, directly translating to higher current efficiency and power efficiency metrics as validated by the comparative device data showing substantial improvements over CBP-based controls.
Mechanistic Insights into Lithiation and Suzuki Coupling Synthesis
The synthesis of these advanced OLED materials relies on a precise sequence of organometallic transformations, beginning with a critical cryogenic lithiation step. As illustrated in the preparation of Intermediate 1, the process initiates with the treatment of 2-bromo-1,8-naphthyridine with n-butyllithium at a strictly controlled temperature of -50°C. This low-temperature environment is paramount to suppress unwanted nucleophilic attacks on the electron-deficient naphthyridine ring and to ensure selective deprotonation or halogen-lithium exchange at the desired position. Following the formation of the lithiated species, the reaction mixture is treated with a methyl ester, such as methyl benzoate, to introduce the ketone functionality. This acylation step constructs the necessary carbon framework for the subsequent cyclization, establishing the core connectivity required for the final heterocyclic system.
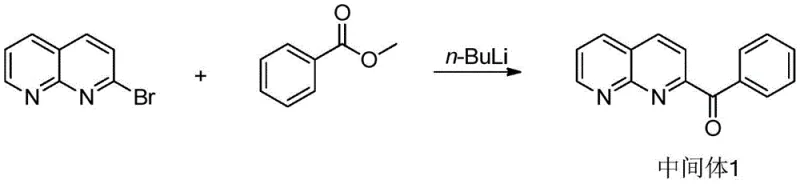
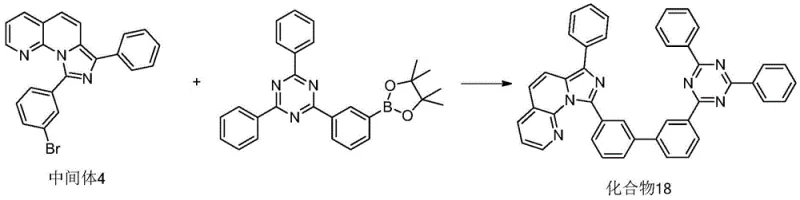
Following the construction of the ketone intermediate, the synthesis proceeds through a condensation-cyclization cascade to form the imidazo-naphthyridine core, followed by a palladium-catalyzed cross-coupling to install the functional aryl groups. The cyclization involves reacting the ketone with a bromobenzaldehyde derivative and ammonium acetate in acetic acid at elevated temperatures around 130°C. The final diversification is achieved via a Suzuki-Miyaura coupling reaction, where the brominated intermediate reacts with various boronic acids or esters in the presence of a tetrakis(triphenylphosphine)palladium catalyst. This step, typically conducted at 110°C in a dioxane-water solvent system, allows for the modular attachment of complex substituents like triazines or spiro-acridines, enabling the fine-tuning of the material's HOMO-LUMO gap and triplet energy levels for specific device applications.
How to Synthesize Imidazo[1,5-a][1,8]naphthyridine Efficiently
The manufacturing protocol for these high-performance host materials involves a streamlined three-stage process that balances yield optimization with impurity control. The initial stage focuses on the generation of the key ketone intermediate through cryogenic lithiation, requiring rigorous temperature management to prevent side reactions. The second stage involves the thermal cyclization to close the imidazole ring, followed by purification via column chromatography to remove unreacted aldehydes and ammonium salts. The final stage employs robust palladium catalysis to couple the core with charge-transporting moieties, a step that benefits from the use of phase-transfer conditions to enhance reaction kinetics. Detailed standardized synthesis steps for scaling this pathway are provided in the guide below.
- Perform cryogenic lithiation of 2-bromo-1,8-naphthyridine using n-BuLi at -50°C followed by acylation with methyl benzoate to form the ketone intermediate.
- Execute cyclization by reacting the ketone intermediate with bromobenzaldehyde and ammonium acetate in acetic acid at 130°C to construct the imidazo-naphthyridine core.
- Finalize the synthesis via Suzuki-Miyaura coupling using tetrakis(triphenylphosphine)palladium catalyst at 110°C to attach diverse aryl substituents.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this new class of imidazo-naphthyridine materials offers profound strategic advantages for supply chain resilience and cost management in electronic chemical manufacturing. The synthetic route avoids the use of exotic or scarce precious metal catalysts beyond standard palladium systems, which are widely available and easily recoverable, thereby mitigating supply risks associated with specialized reagents. Furthermore, the high thermal stability of the final product reduces the failure rate of downstream OLED panels, indirectly lowering the total cost of ownership for display manufacturers by extending product lifecycles. The ability to synthesize these materials using conventional batch reactors without requiring extreme pressure or vacuum conditions simplifies the engineering requirements for commercial scale-up, facilitating a smoother transition from laboratory grams to metric ton production volumes.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for complex multi-step protection and deprotection strategies often required in carbazole chemistry, significantly streamlining the production workflow. By utilizing readily available starting materials like bromo-naphthyridines and substituted benzaldehydes, the raw material costs are kept competitive compared to proprietary host materials with convoluted supply chains. Additionally, the high yields reported in the patent examples, such as the 81% yield for Compound 18, indicate an efficient atom economy that minimizes waste disposal costs and maximizes output per batch, contributing to substantial overall cost savings in electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on standard organic transformations like Suzuki coupling ensures that the production process is compatible with existing infrastructure at most fine chemical contract manufacturing organizations. This compatibility reduces the lead time for high-purity OLED material qualification, as suppliers do not need to invest in specialized equipment to handle hazardous or unstable intermediates. The robustness of the imidazo-naphthyridine core also implies a longer shelf life for the raw materials, reducing inventory write-offs and ensuring a steady supply continuity even during fluctuations in market demand for consumer electronics.
- Scalability and Environmental Compliance: The process generates minimal hazardous byproducts, primarily consisting of inorganic salts and aqueous waste streams that are straightforward to treat in standard wastewater facilities. The absence of heavy metal contaminants in the final product, achieved through efficient purification steps, aligns with stringent environmental regulations for electronic waste and RoHS compliance. This environmental friendliness, combined with the scalability of the liquid-phase reactions, positions these materials as a sustainable choice for large-volume production, supporting the industry's shift towards greener manufacturing practices without compromising on performance metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of imidazo[1,5-a][1,8]naphthyridine compounds in OLED device stacks. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance benchmarks and processing requirements. Understanding these factors is crucial for R&D teams evaluating material substitution and for procurement officers assessing long-term supply viability.
Q: Why are imidazo[1,5-a][1,8]naphthyridine compounds superior to traditional CBP hosts?
A: Unlike CBP which suffers from low glass transition temperatures (62°C) and unbalanced hole/electron transport, these novel compounds exhibit significantly higher thermal stability (Tg up to 99°C) and balanced carrier mobility, leading to extended device lifespan and efficiency.
Q: What represents the critical control point in the synthesis of these OLED materials?
A: The cryogenic lithiation step at -50°C is critical for preventing side reactions and ensuring high regioselectivity, while the final Suzuki coupling requires precise moisture control to maintain catalyst activity and maximize yield.
Q: Can these materials be scaled for commercial OLED production?
A: Yes, the synthetic route utilizes standard industrial reactions like lithiation and Suzuki coupling which are well-established for scale-up, allowing for reliable production from kilogram to metric ton quantities without compromising purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazo[1,5-a][1,8]naphthyridine Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to support the commercialization of these advanced OLED host materials. Our facility is equipped with state-of-the-art reactors capable of handling cryogenic lithiation and high-temperature cyclization safely and efficiently, ensuring consistent quality across batches. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications required for electronic grade chemicals. Our rigorous QC labs utilize advanced analytical techniques to verify the absence of metallic impurities and confirm the structural integrity of every lot, guaranteeing that our clients receive materials that meet the highest industry standards for display manufacturing.
We invite global partners to collaborate with us to leverage these technological advancements for their next-generation display products. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and device architecture. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the rapidly evolving OLED market with our reliable supply of high-performance organic electroluminescent materials.
