Optimizing Beta-Alkoxypropionamide Production for High-Performance Electronic and Industrial Solvents
The global demand for high-performance, environmentally benign solvents in the semiconductor and fine chemical sectors has necessitated a rigorous re-evaluation of traditional synthetic pathways. Patent CN102245561A introduces a transformative methodology for the production of beta-alkoxypropionamides, a class of compounds increasingly vital as replacements for toxic halogenated solvents and reproductive toxins like NMP. This technical insight report analyzes the proprietary two-step transesterification and amidation process, highlighting its potential to redefine supply chain reliability for electronic chemical manufacturers. By decoupling the ester exchange from the final amidation, the process achieves unprecedented selectivity and conversion rates, directly addressing the critical pain points of purity and cost-efficiency faced by R&D directors and procurement managers alike.
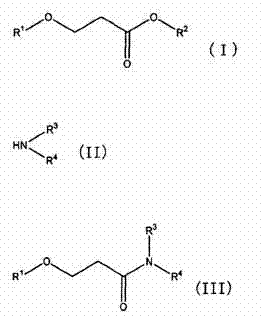
The conventional manufacturing landscape for amide-based solvents has long been plagued by inefficiencies that compromise both economic viability and environmental compliance. Traditional one-pot methods, where transesterification and amidation occur simultaneously, suffer from thermodynamic limitations that result in incomplete conversions and complex separation challenges. Specifically, the coexistence of unreacted amines and by-product alcohols, such as methanol, often leads to the formation of difficult-to-break azeotropes, necessitating energy-intensive distillation columns and reducing overall yield. Furthermore, legacy processes frequently rely on expensive starting materials like dialkylacrylamides or require harsh acidic conditions that generate significant quantities of heavy, tarry by-products, complicating waste treatment and increasing the total cost of ownership for the final solvent product.
In stark contrast, the novel approach detailed in the patent utilizes a strategic "separate reaction" protocol that fundamentally alters the reaction kinetics and equilibrium. By first synthesizing a polyol ester exchanger intermediate through a base-catalyzed transesterification, the process creates a highly reactive species that readily undergoes subsequent amidation. This sequential methodology allows for the removal of volatile by-products prior to the addition of the amine, effectively driving the equilibrium towards the desired product without the interference of competing side reactions. The result is a streamlined workflow that eliminates the need for intermediate neutralization and separation steps, thereby reducing processing time and enhancing the scalability of the operation for industrial-scale production facilities.
Mechanistic Insights into Base-Catalyzed Transesterification and Amidation
The core of this technological advancement lies in the precise manipulation of the catalytic environment during the initial transesterification phase. The utilization of basic catalysts, specifically alkali metal alkoxides like sodium methoxide, serves a dual purpose: it accelerates the nucleophilic attack of the polyol hydroxyl groups on the ester carbonyl while maintaining high selectivity for the mono-esterified intermediate. Unlike acid catalysts, which can promote dehydration and polymerization side reactions leading to heavy ends, the basic system preserves the integrity of the beta-alkoxy structure. This mechanistic control is crucial for minimizing the formation of degradation products such as dimethylacrylamide (DMAA), which poses significant risks due to its high polymerization potential and tendency to cause blockages in industrial piping systems.
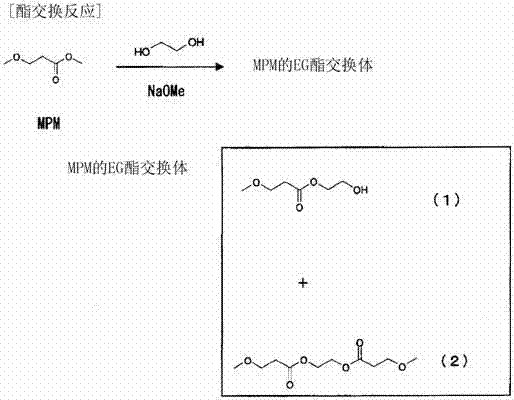
Furthermore, the impurity profile of the final beta-alkoxypropionamide is rigorously controlled through temperature modulation and the specific choice of polyol. The patent data indicates that maintaining the amidation temperature between 30°C and 120°C is critical; exceeding this range triggers thermal decomposition pathways that generate unsaturated impurities. The structural diversity of the polyol component—ranging from ethylene glycol to glycerin and diethylene glycol—allows for fine-tuning the steric and electronic properties of the intermediate ester exchanger. This flexibility ensures that the subsequent nucleophilic substitution by the amine proceeds with minimal resistance, resulting in a final product with a purity profile that meets the stringent specifications required for resist strippers and precision cleaning agents in the electronics industry.
How to Synthesize Beta-Alkoxypropionamides Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and thermal management to maximize the benefits of the separate reaction design. The process begins with the preparation of the polyol ester exchanger, where the molar ratio of polyol to beta-alkoxypropionate is optimized to balance conversion efficiency with downstream separation costs. Following the establishment of the intermediate, the amidation step is initiated under controlled conditions that leverage the residual basicity of the first step, often eliminating the need for additional catalyst loading. For a comprehensive breakdown of the specific operational parameters, reagent grades, and safety protocols required for laboratory and pilot-scale execution, please refer to the standardized synthesis guide below.
- Mix beta-alkoxypropionate (e.g., MPM) with a polyhydric alcohol (e.g., ethylene glycol) and a basic catalyst (e.g., sodium methoxide) in a reactor.
- Heat the mixture to 40-150°C to perform the transesterification reaction, forming a polyol ester exchanger intermediate.
- Add the amine (e.g., dimethylamine) to the reaction mixture and maintain temperature at 30-120°C to complete the amidation without intermediate neutralization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing route offers substantial advantages by shifting the dependency away from volatile and expensive acrylamide derivatives toward more stable and commoditized ester feedstocks. The ability to utilize beta-alkoxypropionates, which can be derived from widely available acrylates and alcohols via Michael addition, creates a more resilient supply chain less susceptible to the price fluctuations associated with specialized monomer markets. Additionally, the elimination of the neutralization and separation steps between the transesterification and amidation phases significantly reduces the consumption of auxiliary chemicals and solvents, leading to a drastic simplification of the unit operations required. This process intensification translates directly into lower capital expenditure for new production lines and reduced operating expenses for existing facilities looking to retrofit their solvent manufacturing capabilities.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high selectivity achieved, which minimizes the loss of valuable raw materials into waste streams. By avoiding the use of expensive dialkylacrylamides and instead utilizing cost-effective beta-alkoxypropionates, the direct material cost per kilogram of the final active ingredient is significantly lowered. Moreover, the continuous nature of the reaction sequence reduces energy consumption associated with heating and cooling cycles, further enhancing the overall cost competitiveness of the produced specialty chemical.
- Enhanced Supply Chain Reliability: The reliance on common industrial chemicals such as ethylene glycol, methanol, and dimethylamine ensures a robust and diversified supplier base, mitigating the risk of single-source bottlenecks. The simplified process flow also shortens the overall production cycle time, allowing for faster turnaround on custom orders and improved responsiveness to market demand spikes. This agility is particularly valuable for clients in the semiconductor sector who require just-in-time delivery of high-purity solvents to maintain uninterrupted fabrication schedules.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts and the reduction of acidic waste streams align perfectly with modern green chemistry principles and increasingly strict environmental regulations. The process generates fewer hazardous by-products, simplifying wastewater treatment and lowering the environmental compliance burden. This clean profile facilitates easier permitting for capacity expansion and supports the sustainability goals of multinational corporations seeking to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability, safety, and quality control aspects of this beta-alkoxypropionamide synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view into the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the separate reaction method superior to the one-pot method for beta-alkoxypropionamides?
A: The separate reaction method prevents the formation of azeotropes between unreacted amines and by-product alcohols (like methanol), allowing for easier purification and significantly higher selectivity (over 97%) compared to simultaneous reactions.
Q: What are the advantages of using a basic catalyst over an acid catalyst in this synthesis?
A: Basic catalysts, such as sodium methoxide, provide higher selectivity for the desired transesterified intermediate and minimize the formation of heavy by-products and decomposition products often seen with acid catalysts like sulfuric acid.
Q: How does this process address impurity concerns like DMAA formation?
A: By controlling the reaction temperature strictly below 120°C during amidation and utilizing the separate step approach, the thermal decomposition that leads to polymerizable impurities like dimethylacrylamide (DMAA) is effectively suppressed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Alkoxypropionamide Supplier
As the electronics and fine chemical industries continue to evolve, the need for efficient, scalable, and environmentally responsible synthesis routes becomes paramount. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging deep expertise in process chemistry to deliver high-value intermediates and solvents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced patents like CN102245561A are fully realized in practical, industrial applications. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for sensitive electronic applications.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific solvent requirements. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your supply chain resilience. Contact us today to discuss how we can support your transition to safer, high-performance chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
