Advanced Catalytic Hydrogenation for m-(β-Hydroxyethylsulfone) Aniline: Scaling High-Purity Dye Intermediates
Advanced Catalytic Hydrogenation for m-(β-Hydroxyethylsulfone) Aniline: Scaling High-Purity Dye Intermediates
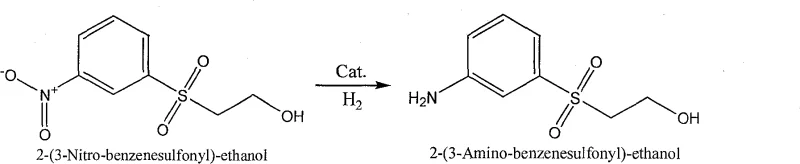
The global demand for high-performance reactive dyes continues to drive innovation in the synthesis of critical intermediates, specifically m-(β-hydroxyethylsulfone) aniline, a cornerstone molecule for vinyl sulfone dye chemistry. A pivotal advancement in this domain is detailed in patent CN100575339C, which introduces a robust method for preparing this key amine via the hydrogenation of m-(β-hydroxyethylsulfone) nitrobenzene using a specialized amorphous nickel alloy catalyst. This technology represents a significant departure from legacy manufacturing routes, addressing long-standing issues regarding environmental compliance, catalyst longevity, and process economics. By leveraging a modified amorphous nickel-aluminum alloy system, the process achieves exceptional selectivity and conversion under mild conditions, effectively bridging the gap between the high cost of noble metal catalysts and the severe pollution associated with traditional iron powder reduction methods. For industrial stakeholders, this patent outlines a pathway to a more sustainable and cost-effective supply chain for essential dye precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of m-(β-hydroxyethylsulfone) aniline has relied heavily on two primary methodologies, both of which present substantial operational and environmental drawbacks for modern chemical manufacturing. The most widespread traditional technique involves the reduction of the corresponding nitro compound using iron powder in an electrolyte solution. While chemically effective, this method is notoriously inefficient from a waste management perspective, generating voluminous quantities of iron sludge and wastewater contaminated with aromatic amines, posing severe disposal challenges and regulatory risks. Alternatively, catalytic hydrogenation using noble metals like palladium or platinum offers a cleaner profile but introduces prohibitive costs due to the price of the precious metals and the complexity of recovering them from the reaction mixture. Furthermore, conventional non-noble catalysts, such as standard skeletal nickel (Raney Nickel), have historically struggled with rapid deactivation and short service lives when applied to this specific substrate, leading to frequent catalyst replacement, increased downtime, and inconsistent batch quality.
The Novel Approach
The methodology disclosed in patent CN100575339C fundamentally reengineers the catalytic landscape by employing a modified amorphous nickel alloy that combines the economic benefits of base metals with the performance characteristics typically reserved for noble catalysts. This novel approach utilizes a unique catalyst precursor composed of nickel, aluminum, and specific additive metals (such as Fe, Mn, Mo, W, Co, Cu, or Cr), which is rapidly quenched to form an amorphous structure before being activated via alkali leaching. This specific structural arrangement creates a highly active surface with isotropic properties and a high density of active sites, granting the catalyst superior resistance to poisoning and thermal stability. Consequently, this new route not only eliminates the generation of hazardous iron sludge but also drastically extends the operational lifespan of the catalyst compared to traditional skeletal nickel, ensuring a more continuous and reliable production process with significantly reduced operational expenditures.
Mechanistic Insights into Amorphous Nickel Alloy Catalyzed Hydrogenation
The efficacy of this process lies in the unique atomic structure of the amorphous nickel alloy catalyst, which differs fundamentally from crystalline counterparts. Unlike crystalline catalysts where active sites are limited to specific lattice defects or edges, the amorphous structure possesses a disordered atomic arrangement that results in a uniform distribution of active centers across the entire surface. This "short-range order, long-range disorder" configuration facilitates the adsorption and activation of hydrogen molecules and the nitro substrate with remarkable efficiency. The addition of promoter metals (Component M in the NiAlM alloy) further modulates the electronic state of the nickel atoms, enhancing their ability to transfer hydrogen to the nitro group while suppressing side reactions that could lead to impurities. The activation process, involving the selective leaching of aluminum with sodium hydroxide, creates a highly porous "skeleton" structure that maximizes the specific surface area available for the reaction, thereby driving the kinetics forward even at moderate temperatures.
From an impurity control perspective, the mechanism ensures high selectivity for the primary amine, minimizing the formation of hydroxylamine intermediates or azo-coupling byproducts that often plague nitro reductions. The mild reaction conditions (50-90°C) prevent thermal degradation of the sensitive hydroxyethyl sulfone moiety, which can be prone to elimination reactions under harsher alkaline or acidic conditions found in other methods. The two-stage hydrogenation process mentioned in the patent abstract suggests a controlled addition of hydrogen or a staged temperature profile that allows for precise management of the exothermic reaction heat, further safeguarding product integrity. This mechanistic robustness translates directly to a cleaner crude product profile, reducing the burden on downstream purification units and ensuring that the final m-(β-hydroxyethylsulfone) aniline meets the stringent purity specifications required for high-quality vinyl sulfone dye synthesis.
How to Synthesize m-(β-Hydroxyethylsulfone) Aniline Efficiently
Implementing this advanced catalytic route requires precise adherence to the catalyst preparation and reaction parameters outlined in the intellectual property to ensure reproducibility and safety. The process begins with the synthesis of the amorphous alloy precursor, followed by its activation to generate the active catalytic species, and finally, the hydrogenation reaction itself in a pressurized reactor system. Operators must maintain strict control over the hydrogen pressure and temperature profiles to maximize the turnover number of the catalyst while preventing any potential safety incidents associated with high-pressure hydrogen handling. The following guide summarizes the critical operational phases derived from the patent examples, providing a framework for R&D teams to adapt this technology for pilot or commercial scale operations.
- Preparation of the amorphous NiAlM alloy precursor by melting Ni, Al, and additive metals (Fe, Mn, Mo, etc.) at 1000-1600°C followed by rapid quenching to form thin strips.
- Activation of the alloy powder by treating with 10-25wt% NaOH solution at 70-100°C to leach out aluminum, creating the active amorphous skeleton nickel structure.
- Hydrogenation of m-(β-hydroxyethylsulfone) nitrobenzene in methanol at 50-90°C and 0.5-1.2 MPa pressure using the activated catalyst to achieve >98% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this amorphous nickel catalytic technology offers a compelling value proposition centered on cost stability and operational continuity. By shifting away from the volatile pricing of noble metals and the waste disposal costs of iron powder reduction, manufacturers can achieve a more predictable cost structure for this critical dye intermediate. The elimination of heavy metal sludge not only reduces environmental compliance costs but also simplifies the logistics of waste transport and treatment, freeing up resources for core production activities. Furthermore, the enhanced durability of the catalyst means fewer interruptions for catalyst change-outs, directly translating to higher plant utilization rates and more reliable delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts (like Pd/C) with a non-noble amorphous nickel alloy results in a drastic reduction in raw material costs for the catalytic system. Since the catalyst life is significantly extended compared to traditional skeletal nickel, the frequency of catalyst purchase and regeneration is lowered, compounding the savings over time. Additionally, the high conversion rates (>98%) minimize the loss of valuable starting materials, improving the overall atom economy of the process and reducing the cost per kilogram of the final API or dye intermediate.
- Enhanced Supply Chain Reliability: The robustness of the amorphous nickel catalyst against poisoning ensures consistent batch-to-batch quality, reducing the risk of production delays caused by off-spec products. The use of readily available base metals (Nickel, Aluminum) for catalyst fabrication mitigates the supply chain risks associated with geopolitically sensitive precious metals. This stability allows for longer production runs without the need for frequent shutdowns, ensuring a steady flow of m-(β-hydroxyethylsulfone) aniline to meet the demands of the reactive dye market.
- Scalability and Environmental Compliance: The process operates under relatively mild pressures (0.5-1.2 MPa) and temperatures, making it highly scalable from pilot plants to multi-ton commercial reactors without requiring exotic high-pressure equipment. The absence of iron sludge and the use of methanol as a solvent (which can be recovered and recycled) align with increasingly strict global environmental regulations, future-proofing the manufacturing site against tightening emission standards and avoiding potential fines or shutdowns related to pollution.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amorphous nickel catalytic hydrogenation technology. These insights are derived directly from the experimental data and claims presented in patent CN100575339C, offering clarity on the practical application of this method for industrial synthesis. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing infrastructure.
Q: How does the amorphous nickel catalyst compare to traditional iron powder reduction?
A: Unlike the traditional iron powder method which generates massive amounts of toxic iron sludge and wastewater containing aromatic amines, the amorphous nickel catalytic process is environmentally friendly, produces no heavy metal sludge, and offers significantly higher atom economy and easier product isolation.
Q: What are the specific advantages of this catalyst over standard Raney Nickel?
A: Standard skeleton nickel catalysts often suffer from rapid deactivation and short service life when used for this specific substrate. The modified amorphous nickel alloy described in patent CN100575339C exhibits superior anti-poisoning capabilities and structural stability, resulting in a prolonged catalyst lifespan and consistent activity over multiple cycles.
Q: What are the optimal reaction conditions for this hydrogenation process?
A: The process operates under relatively mild conditions compared to many high-pressure hydrogenations. The optimal range identified is a temperature of 60-70°C and a hydrogen pressure of 0.8-1.0 MPa, with a reaction time of approximately 5-6 hours, achieving conversion rates of 100% and yields exceeding 98.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-(β-Hydroxyethylsulfone) Aniline Supplier
The technological breakthroughs detailed in patent CN100575339C underscore the immense potential for optimizing the production of m-(β-hydroxyethylsulfone) aniline, a vital building block for the global dye and specialty chemical industries. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot validation to full-scale supply is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of dye intermediate meets the exacting standards required by international textile and chemical manufacturers.
We invite you to collaborate with us to leverage this advanced catalytic technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your product quality while driving down your total cost of ownership.
