Advanced Macrocyclic Tyrosine Kinase Inhibitors: Overcoming Drug Resistance in Oncology
Advanced Macrocyclic Tyrosine Kinase Inhibitors: Overcoming Drug Resistance in Oncology
The landscape of oncology treatment is continuously evolving, driven by the urgent need to address drug resistance in targeted therapies. Patent CN111171049B introduces a groundbreaking class of macrocyclic tyrosine kinase inhibitors that demonstrate potent activity against TRK, ALK, and ROS1 receptors. These compounds are specifically engineered to overcome common resistance mutations, such as G595R and G623R, which often limit the efficacy of first-generation inhibitors. The disclosed technology represents a significant leap forward in medicinal chemistry, offering a robust platform for developing next-generation anticancer agents with improved pharmacokinetic profiles and bioavailability. For pharmaceutical developers and procurement specialists, understanding the synthetic accessibility and structural novelty of these intermediates is crucial for securing a reliable supply chain for future clinical programs.
![General Formula I of macrocyclic tyrosine kinase inhibitors showing the pyrazolo[1,5-a]pyrimidine core and variable ring systems](/insights/img/macrocyclic-kinase-inhibitors-pharma-supplier-20260309081533-01.png)
As a leading fine chemical manufacturer, we recognize the critical importance of high-purity intermediates in the development of these complex therapeutics. The structural versatility outlined in the patent allows for extensive SAR (Structure-Activity Relationship) exploration, enabling the optimization of potency against resistant tumor lines. By leveraging advanced catalytic techniques and precise process control, we ensure that every batch of these specialized intermediates meets the stringent quality standards required for GMP manufacturing. This commitment to excellence positions us as a reliable pharmaceutical intermediate supplier capable of supporting the rigorous demands of global drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional small molecule kinase inhibitors, while effective initially, often face the challenge of acquired resistance due to gatekeeper mutations in the ATP-binding pocket of the kinase domain. Conventional synthesis routes for kinase inhibitors frequently rely on linear architectures that may lack the conformational rigidity necessary to maintain binding affinity in the presence of steric hindrance caused by mutations. Furthermore, older manufacturing processes often involve harsh reaction conditions, expensive transition metal catalysts, and difficult purification steps that generate significant waste. These factors not only increase the cost of goods sold (COGS) but also introduce supply chain vulnerabilities related to raw material availability and environmental compliance. The inability of conventional acyclic inhibitors to effectively engage the mutated kinase domains necessitates a paradigm shift towards more sophisticated molecular architectures.
The Novel Approach
The novel approach detailed in patent CN111171049B utilizes a macrocyclic constraint to lock the inhibitor into a bioactive conformation that is less susceptible to resistance mechanisms. By bridging specific regions of the molecule, the macrocycle reduces the entropic penalty of binding, thereby enhancing affinity even against mutated targets like NTRK1-G595R. The synthesis strategy employs a convergent route where key fragments, such as the pyrazolo[1,5-a]pyrimidine core and the fused heterocyclic side chain, are assembled efficiently. This methodology avoids the use of prohibited substances and minimizes the reliance on scarce resources, aligning with green chemistry principles. The result is a streamlined process that yields high-purity compounds with excellent stability, facilitating cost reduction in API manufacturing and ensuring a consistent supply for clinical trials and commercial launch.
Mechanistic Insights into Macrocyclization and Kinase Inhibition
The mechanistic basis for the enhanced efficacy of these compounds lies in their ability to span the ATP-binding cleft of the tyrosine kinase while maintaining critical hydrogen bonding interactions. The pyrazolo[1,5-a]pyrimidine scaffold serves as a potent hinge binder, mimicking the adenine ring of ATP. The macrocyclic linker, often incorporating ether or amine functionalities, extends into the solvent-exposed region or the back pocket, providing additional binding energy that compensates for the loss of interactions caused by mutations. This dual-binding mode is crucial for overcoming the steric bulk introduced by residues like Glycine to Arginine substitutions. From a synthetic perspective, the formation of the macrocycle is a pivotal step, typically achieved through intramolecular amide coupling or nucleophilic substitution. Careful selection of coupling reagents and reaction conditions is essential to prevent oligomerization and ensure high cyclization efficiency.
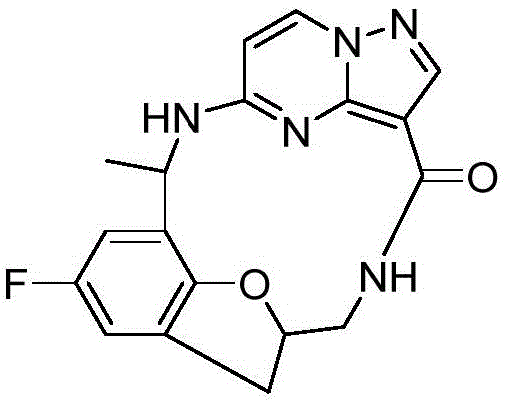
Impurity control is another critical aspect of the mechanism, as trace impurities can affect the safety profile of the final drug product. The synthetic route described in the patent incorporates specific purification steps, such as column chromatography and recrystallization, to remove side products like unreacted starting materials or hydrolysis byproducts. For instance, the use of protecting groups like phthalimide allows for the selective functionalization of amine sites without affecting other sensitive moieties. Subsequent deprotection steps are optimized to minimize degradation, ensuring that the final API intermediate possesses the required chemical integrity. Understanding these mechanistic nuances allows process chemists to troubleshoot potential scale-up issues and implement robust quality control measures that guarantee batch-to-batch consistency.
How to Synthesize Macrocyclic Tyrosine Kinase Inhibitors Efficiently
The synthesis of these complex macrocyclic inhibitors requires a meticulous multi-step approach that balances yield, purity, and operational simplicity. The process typically begins with the construction of the fused heterocyclic core, such as a benzofuran or cyclopenta[c]pyridine system, followed by the installation of the pyrazolo[1,5-a]pyrimidine warhead. Key transformations include halogenation, nucleophilic aromatic substitution, and reductive amination, each requiring precise control of temperature and stoichiometry. The final macrocyclization step is particularly sensitive, often requiring high dilution techniques or specialized coupling agents to favor intramolecular reaction over intermolecular polymerization. Detailed standardized synthesis steps are provided in the guide below to assist R&D teams in replicating these high-value intermediates.
- Preparation of key intermediates such as 2-bromo-4-fluorophenol and subsequent allylation to form the benzofuran core structure.
- Coupling of the benzofuran amine intermediate with ethyl 5-chloropyrazolo[1,5-a]pyrimidine-3-carboxylate under basic conditions.
- Final macrocyclization via amide bond formation using coupling reagents like FDPP to close the ring and yield the target inhibitor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere technical performance. The process design prioritizes the use of commercially available starting materials and avoids exotic reagents that are prone to supply disruptions. This inherent robustness translates into enhanced supply chain reliability, ensuring that production schedules are met without unexpected delays. Furthermore, the simplified workflow reduces the number of unit operations, which directly correlates to lower manufacturing costs and reduced capital expenditure on equipment. By eliminating the need for complex chiral resolution steps in certain embodiments through the use of chiral pool starting materials, the overall process economics are significantly improved, making these inhibitors viable candidates for widespread clinical use.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts often used in cross-coupling reactions, replacing them with more economical organic transformations. This shift not only lowers the direct material costs but also simplifies the downstream processing by removing heavy metal scavenging steps. The high atom economy of the macrocyclization step ensures that raw materials are utilized efficiently, minimizing waste disposal costs. Additionally, the ability to perform reactions at ambient or moderate temperatures reduces energy consumption, contributing to a lower carbon footprint and further operational savings.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals such as substituted phenols, allyl bromides, and common heterocycles ensures that the supply chain is not dependent on single-source suppliers for niche intermediates. The modular nature of the synthesis allows for flexible manufacturing, where different analogues can be produced on the same production line with minimal changeover time. This flexibility is crucial for responding to fluctuating demand in the pharmaceutical market. Moreover, the stability of the intermediates allows for longer storage periods, enabling manufacturers to build strategic stockpiles that buffer against global logistics volatility.
- Scalability and Environmental Compliance: The process has been designed with scalability in mind, utilizing solvents and reagents that are compatible with large-scale reactor systems. The avoidance of highly toxic reagents and the generation of benign byproducts facilitate easier waste treatment and regulatory compliance. This alignment with environmental, social, and governance (ESG) goals is increasingly important for pharmaceutical companies aiming to reduce their environmental impact. The robust nature of the chemistry ensures that yields remain consistent when scaling from laboratory grams to commercial metric tons, reducing the risk of batch failures and ensuring a steady flow of high-quality material.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these macrocyclic tyrosine kinase inhibitors. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. They serve to clarify the capabilities of the manufacturing process and the therapeutic potential of the compounds. Understanding these details helps stakeholders make informed decisions regarding partnership and procurement strategies.
Q: What specific mutations do these macrocyclic inhibitors target?
A: These compounds are designed to inhibit TRK, ALK, and ROS1 kinases, specifically addressing resistance mutations such as NTRK1-G595R and NTRK3-G623R found in various solid tumors.
Q: Is the synthesis process scalable for commercial production?
A: Yes, the patent describes a preparation process characterized by simple steps, high purity, and stable quality, making it suitable for large-scale industrial production from kilograms to metric tons.
Q: What is the core structural feature of these inhibitors?
A: The core structure features a pyrazolo[1,5-a]pyrimidine moiety linked via a macrocyclic bridge to a fused heterocyclic system, such as a benzofuran or cyclopenta[c]pyridine derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Macrocyclic Tyrosine Kinase Inhibitors Supplier
At NINGBO INNO PHARMCHEM, we combine deep chemical expertise with state-of-the-art manufacturing capabilities to deliver high-performance pharmaceutical intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every shipment meets the highest industry standards. Our commitment to quality and reliability makes us the preferred partner for global pharmaceutical companies seeking to advance their oncology pipelines with cutting-edge kinase inhibitors.
We invite you to contact our technical procurement team to discuss your specific requirements. We offer a Customized Cost-Saving Analysis to help you optimize your supply chain and reduce overall project costs. Reach out today to request specific COA data and route feasibility assessments for your next development program. Let us support your mission to bring life-saving therapies to patients worldwide with our superior chemical solutions and dedicated service.
