Advanced Manufacturing of Betrixaban Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for novel anticoagulants, and the synthesis of Betrixaban represents a critical area of innovation for supply chain stability. Patent CN108017576B introduces a transformative preparation method for Betrixaban and its hydrochloride salt that addresses long-standing inefficiencies in prior art. This technical disclosure outlines a streamlined approach utilizing thionyl chloride activation to generate an acid chloride intermediate, which subsequently undergoes acylation to form the target molecule. By shifting away from complex coupling agents and sensitive organometallic reagents, this methodology offers a pathway to high-purity intermediates suitable for large-scale commercialization. The strategic implementation of this chemistry allows manufacturers to bypass tedious purification steps, directly crystallizing the product from the reaction matrix. For global procurement teams and R&D directors, understanding this shift is vital for securing reliable sources of high-quality API intermediates.
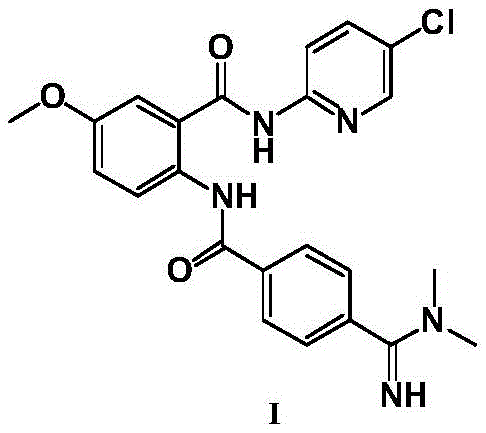
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Betrixaban has been plagued by significant operational hurdles that impede efficient industrial scale-up. Early methodologies, such as those disclosed in WO2012031017, relied heavily on carbodiimide coupling agents like EDC to facilitate amide bond formation. While chemically feasible, this approach generates urea derivatives as byproducts which possess strong polarity and high water solubility, making them notoriously difficult to separate from the target compound through standard phase extraction. Furthermore, alternative routes described in documents like WO2001019788 necessitated the use of corrosive hydrogen chloride gas in methanol, posing severe safety risks and environmental challenges regarding waste disposal. Perhaps most critically, processes utilizing active metal reagents such as n-hexyllithium or Grignard reagents, as seen in WO2007056517 and CN104693114, impose rigorous demands for strictly anhydrous conditions and cryogenic temperatures. These legacy methods not only inflate production costs due to the expense of specialized reagents but also introduce substantial variability in yield and purity, often requiring column chromatography for final purification.
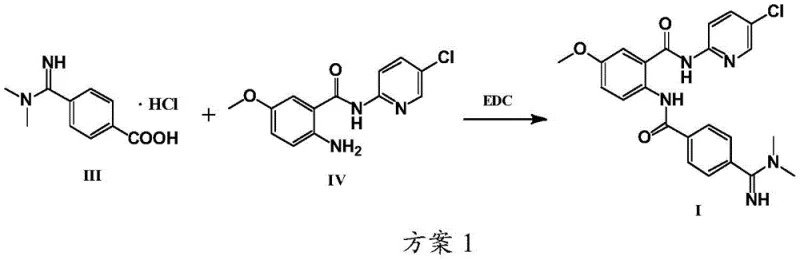
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the novel process detailed in CN108017576B leverages a classic yet optimized acylation strategy that prioritizes operational simplicity and economic efficiency. The core innovation lies in the in situ generation of an acid chloride intermediate (Compound VIII) from Compound III using thionyl chloride, followed by immediate reaction with the amine component (Compound IV). This sequence effectively circumvents the formation of hard-to-remove urea byproducts associated with EDC coupling. Moreover, the reaction conditions are remarkably mild, operating effectively within a temperature range of 0°C to 80°C, thereby eliminating the need for energy-intensive cryogenic cooling required by organolithium routes. A distinct advantage of this methodology is the ability to select solvents, such as dichloromethane or acetone, in which the final Betrixaban Hydrochloride product has low solubility. This physicochemical property enables the product to precipitate directly from the reaction system upon completion, allowing for isolation via simple filtration rather than complex chromatographic separation. This fundamental shift in process design drastically reduces solvent consumption and processing time.
Mechanistic Insights into Thionyl Chloride-Mediated Acylation
The mechanistic elegance of this synthesis rests on the efficient activation of the carboxylic acid moiety in Compound III. In the first stage, thionyl chloride acts as a chlorinating agent, converting the carboxylic acid into the highly reactive acid chloride (Compound VIII). The addition of a catalytic amount of N,N-dimethylformamide (DMF) serves to accelerate this transformation by forming a reactive Vilsmeier-Haack type intermediate, ensuring rapid and complete conversion even at moderate temperatures. This activation step is crucial as it primes the molecule for nucleophilic attack in the subsequent acylation phase. The reaction stoichiometry is carefully balanced, typically employing a molar ratio of thionyl chloride to Compound III ranging from 1:1 to 10:1, with a preference for 1:1 to 3:1 to minimize equipment corrosion while ensuring full conversion. The resulting acid chloride is sufficiently stable under the reaction conditions to proceed to the next step without isolation, streamlining the workflow.
Following activation, the introduction of Compound IV in the presence of a base initiates the acylation. The base, which can range from weak organic bases like triethylamine to inorganic carbonates like sodium bicarbonate, plays a dual role: it neutralizes the hydrogen chloride generated during the amide bond formation and scavenges any decomposition products from the thionyl chloride. Crucially, the selection of the base impacts the stability of the amidino group present in the molecule. While strong bases like sodium hydroxide can lead to partial hydrolysis of the amidino functionality, medium-strength or weak bases preserve this sensitive group, resulting in higher yields exceeding 85%. The solvent system is equally pivotal; by utilizing a solvent that is a poor solver for the product but a good solver for the reactants, the equilibrium is driven towards precipitation. This crystallization-induced driving force not only pushes the reaction to completion but also inherently purifies the product, as impurities remain dissolved in the mother liquor, yielding a solid with purity greater than 99.5%.
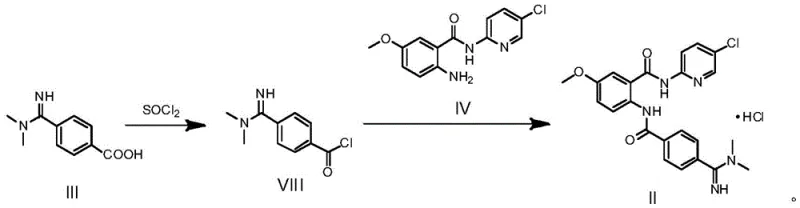
How to Synthesize Betrixaban Hydrochloride Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and ensure consistent crystal form production. The process begins with the activation of the benzoic acid derivative using thionyl chloride in an aprotic solvent, followed by the controlled addition of the amine partner and a suitable acid scavenger. Detailed operational guidelines regarding temperature ramps, addition rates, and workup procedures are essential for reproducibility. The following section outlines the standardized protocol derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale implementation.
- Activate Compound III by reacting with thionyl chloride (SOCl2) and a catalytic amount of DMF in an aprotic solvent like dichloromethane to form the acid chloride intermediate (Compound VIII).
- Perform acylation by adding Compound IV and a base (such as triethylamine or sodium bicarbonate) to the reaction mixture, maintaining temperatures between 0°C and 80°C.
- Isolate the final Betrixaban Hydrochloride product directly via filtration from the reaction system, achieving purity greater than 99.5% without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this novel synthetic route offers profound implications for cost structure and supply reliability. By transitioning away from proprietary coupling agents and hazardous organometallic reagents, manufacturers can significantly reduce the raw material cost basis associated with Betrixaban production. The elimination of column chromatography, a resource-intensive unit operation, further drives down processing costs and increases throughput capacity. Additionally, the use of common, commercially available solvents and reagents mitigates supply risk, ensuring that production schedules are not disrupted by the scarcity of specialized chemicals. The robustness of the process also translates to reduced batch failure rates, enhancing overall supply chain continuity.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive coupling reagents like EDC and sensitive organolithium compounds with cost-effective thionyl chloride. Furthermore, the ability to isolate the product via direct filtration eliminates the need for expensive silica gel and large volumes of elution solvents required for chromatography. This simplification of the downstream processing workflow results in substantial savings in both material costs and labor hours. The potential for solvent recycling, particularly when the same solvent is used for both reaction steps, further amplifies these cost efficiencies by reducing waste disposal fees and fresh solvent procurement needs.
- Enhanced Supply Chain Reliability: Supply security is markedly improved as the key reagents, including thionyl chloride and various amines, are commodity chemicals with stable global supply chains. Unlike methods relying on custom-synthesized catalysts or reagents with short shelf-lives, the inputs for this process are readily accessible from multiple vendors. This diversification of the supply base reduces dependency on single-source suppliers and minimizes the risk of production stoppages due to raw material shortages. The mild reaction conditions also reduce the burden on facility infrastructure, allowing for production in a wider range of manufacturing sites without the need for specialized cryogenic or high-pressure equipment.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is superior due to its atom economy and reduced waste generation. The avoidance of heavy metal catalysts and the minimization of solvent usage align with green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing site. The direct crystallization process generates a solid product that is easy to handle and transport, simplifying logistics. The high purity achieved directly from the reactor means fewer recrystallization cycles are needed, which in turn reduces the volume of wastewater and organic waste streams that require treatment, ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and quality attributes of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on critical process parameters and product specifications.
Q: What is the primary advantage of the thionyl chloride route over EDC coupling for Betrixaban?
A: The thionyl chloride route eliminates the formation of polar urea derivatives associated with EDC coupling, which are difficult to remove via extraction. This new method allows for direct crystallization of the product from the reaction system, significantly simplifying purification.
Q: Does this process require harsh anhydrous conditions like organolithium methods?
A: No. Unlike prior art methods utilizing n-hexyllithium or Grignard reagents which demand strict anhydrous environments and cryogenic temperatures, this novel process operates under mild conditions (0°C to 80°C) and tolerates a broader range of solvents, enhancing operational safety.
Q: What purity levels can be achieved with this manufacturing method?
A: The patented method consistently yields Betrixaban Hydrochloride with an HPLC purity of not less than 99.5%, with all single impurities controlled below 0.1%, meeting stringent regulatory requirements for API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Betrixaban Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering Betrixaban intermediates with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the >99.5% purity benchmark established by the latest patent innovations. Our facility is equipped to handle the specific solvent systems and crystallization protocols required for this process, guaranteeing consistent quality and supply continuity for our global partners.
We invite you to collaborate with us to optimize your supply chain for this vital anticoagulant intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's efficiency and profitability.
