Advanced Synthesis of Dioxopromethazine Hydrochloride: A Safe and Scalable Commercial Route
Introduction to Next-Generation Dioxopromethazine Hydrochloride Manufacturing
The pharmaceutical industry continuously seeks robust, safe, and economically viable synthetic routes for critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is documented in patent CN111943910B, which outlines a novel preparation method for Dioxopromethazine Hydrochloride, a potent antitussive agent with antihistamine and anti-inflammatory properties. This technology addresses long-standing safety and efficiency bottlenecks associated with traditional oxidation protocols by leveraging potassium hydrogen persulfate (KHSO5) as a selective oxidant. For R&D directors and procurement strategists, this patent represents a pivotal shift away from hazardous peroxide systems and expensive noble metal catalysts toward a streamlined, ambient-pressure process. The methodology not only enhances operational safety by eliminating the need for high-temperature reflux but also ensures a high-purity profile essential for regulatory compliance. By adopting this advanced synthetic strategy, manufacturers can secure a more reliable supply chain for this valuable pharmaceutical intermediate while simultaneously driving down production costs through simplified unit operations and reduced energy expenditure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dioxopromethazine Hydrochloride has been plagued by significant technical and safety challenges that hinder scalable manufacturing. Traditional methods predominantly rely on hydrogen peroxide oxidation systems, which necessitate rigorous heating or reflux conditions to drive the reaction to completion. These thermal requirements introduce substantial safety risks, including the potential for runaway exothermic reactions and the handling of unstable peroxide concentrations at elevated temperatures. Furthermore, alternative approaches disclosed in prior art, such as CN106749098B, utilize oxygen as an oxidant in conjunction with palladium acetate catalysts. While effective, this catalytic system imposes a heavy financial burden due to the high cost of palladium and the complexity of catalyst recovery. The susceptibility of palladium catalysts to poisoning further complicates the process, leading to inconsistent batch quality and increased waste generation. Additionally, oxygen-based systems often require pressurized reactors and extended reaction times, escalating both capital expenditure (CAPEX) for specialized equipment and operational expenditure (OPEX) for energy and maintenance. These cumulative inefficiencies create a fragile supply chain vulnerable to raw material price volatility and regulatory scrutiny regarding heavy metal residues.
The Novel Approach
The innovative process detailed in patent CN111943910B fundamentally reengineers the oxidation step to overcome these legacy deficiencies. By substituting hazardous peroxides and expensive palladium catalysts with potassium hydrogen persulfate (KHSO5), the new method achieves efficient oxidation of the promethazine sulfur atom under mild, non-pressurized conditions. This transition allows the reaction to proceed at low temperatures initially (0-20°C) followed by moderate heating (10-40°C), drastically reducing the thermal load and eliminating the need for high-pressure vessels. The operational simplicity of the KHSO5 system means that standard glass-lined or stainless steel reactors can be utilized without specialized pressure ratings, thereby lowering barrier-to-entry for manufacturing partners. Moreover, the absence of transition metal catalysts inherently guarantees a cleaner impurity profile, removing the necessity for complex metal scavenging steps that often reduce overall yield. This approach not only mitigates safety hazards associated with energetic materials but also aligns with green chemistry principles by reducing energy consumption and simplifying wastewater treatment protocols, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into KHSO5-Mediated Phenothiazine Oxidation
The core chemical transformation in this synthesis involves the selective oxidation of the sulfur atom within the phenothiazine ring of promethazine to form the corresponding sulfone, dioxopromethazine. Potassium hydrogen persulfate acts as a powerful yet controllable oxygen transfer agent in this mechanism. In the initial phase, the free base of promethazine is generated in situ or introduced into an aqueous medium, where it interacts with the bisulfate ions. Upon the dropwise addition of the KHSO5 solution at controlled low temperatures, the electrophilic oxygen species attack the nucleophilic sulfur center. This step is critical; maintaining the temperature between 0°C and 20°C during addition prevents over-oxidation or degradation of the sensitive amine side chain. Following the initial addition, the reaction mixture is gently warmed to 10-40°C to ensure complete conversion of the sulfide to the sulfone. This two-stage temperature profile optimizes the reaction kinetics, balancing the rate of oxidation with the stability of the intermediates. The use of an aqueous acidic medium (promethazine bisulfate solution) further stabilizes the amine functionality against unwanted side reactions, ensuring that the oxidation is highly regioselective for the sulfur atom.
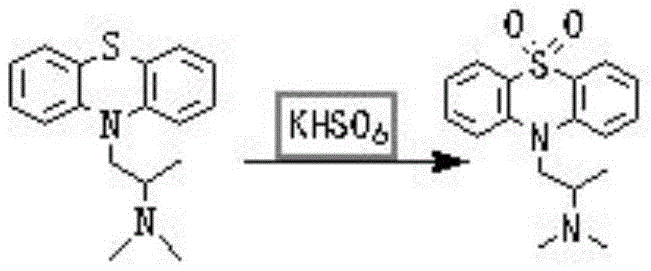
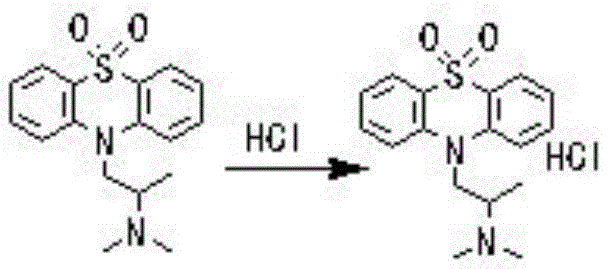
Following the oxidation, the process employs a sophisticated workup strategy to isolate the product with high fidelity. The reaction mixture is basified to liberate the free dioxopromethazine base, which is then extracted into an organic solvent such as dichloromethane. This liquid-liquid extraction effectively separates the organic product from inorganic sulfate byproducts and unreacted oxidant. The subsequent salt formation step involves dissolving the dried organic residue in acetone and introducing anhydrous hydrogen chloride gas. This acid-base reaction precipitates the hydrochloride salt, which is less soluble in acetone, facilitating easy filtration. To achieve the stringent purity standards required for pharmaceutical applications, a refining cycle is implemented. The crude salt is re-dissolved, basified to the free base, treated with activated carbon to adsorb colored impurities and trace organics, and then re-salted. This recrystallization-like purification ensures that the final Dioxopromethazine Hydrochloride meets purity specifications exceeding 98%, with minimal levels of related substances.
How to Synthesize Dioxopromethazine Hydrochloride Efficiently
The synthesis of high-purity Dioxopromethazine Hydrochloride via this patented route requires precise control over reaction parameters, particularly temperature and pH, to maximize yield and minimize impurity formation. The process begins with the preparation of a promethazine bisulfate solution, followed by the critical oxidation step using potassium hydrogen persulfate. Operators must adhere strictly to the temperature gradients specified, ensuring the exotherm during oxidant addition is managed effectively to prevent thermal degradation. Subsequent extraction and salt formation steps utilize common solvents like dichloromethane and acetone, which are easily recoverable, enhancing the economic viability of the process. The refining stage, involving activated carbon treatment, is essential for meeting visual and chromatographic purity standards. For a comprehensive understanding of the specific reagent quantities, stirring rates, and isolation techniques required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Oxidize free promethazine in an aqueous solution using potassium hydrogen persulfate (KHSO5) at controlled low temperatures (0-20°C) followed by mild heating to form dioxopromethazine.
- Extract the oxidized product into dichloromethane, wash, remove solvent, and dissolve in acetone before introducing hydrogen chloride gas to precipitate the crude hydrochloride salt.
- Purify the crude salt by liberating the free base, decolorizing with activated carbon in acetone, and re-forming the hydrochloride salt to achieve purity greater than 98%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the KHSO5-based synthesis route for Dioxopromethazine Hydrochloride offers transformative advantages in terms of cost structure and supply reliability. The elimination of palladium catalysts removes a major variable cost driver, as precious metal prices are subject to significant market fluctuations and geopolitical supply risks. Furthermore, the removal of heavy metals from the process flow negates the need for expensive scavenger resins and the associated validation testing for residual metals, streamlining the quality control workflow. The operational safety improvements also translate into tangible commercial benefits; by avoiding high-pressure oxygen systems and hazardous peroxide reflux, facilities can reduce insurance premiums and mitigate the risk of production shutdowns due to safety incidents. The use of commodity chemicals like potassium hydrogen persulfate and acetone ensures that raw material sourcing remains stable and competitive, shielding the supply chain from the bottlenecks often associated with specialized reagents.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free oxidation system is profound. By eradicating the need for palladium acetate, manufacturers avoid the substantial capital tie-up associated with precious metal catalysts and the complex recovery infrastructure required to make them economically feasible. Additionally, the ambient pressure operation significantly lowers energy consumption compared to pressurized oxygen oxidation or high-temperature reflux methods, resulting in reduced utility costs per kilogram of product. The simplified equipment requirements mean that existing multipurpose reactors can be utilized without costly modifications, accelerating time-to-market and maximizing asset utilization rates. These factors combine to deliver substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing strategies in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, bulk commodity reagents rather than specialized catalytic systems. Potassium hydrogen persulfate is a stable, commercially abundant oxidant with a robust global supply network, minimizing the risk of raw material shortages that can plague niche catalyst markets. The robustness of the reaction conditions—tolerant of minor variations and operable at mild temperatures—ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification results that disrupt inventory planning. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream API manufacturers, thereby strengthening the overall integrity of the pharmaceutical supply chain.
- Scalability and Environmental Compliance: From a scalability perspective, the process is inherently designed for large-scale commercialization. The absence of pressurization requirements simplifies the engineering controls needed for scale-up from pilot plant to multi-ton production, reducing the timeline for technology transfer. Environmentally, the process generates wastewater that is easier to treat compared to streams contaminated with heavy metals or high concentrations of organic peroxides. The ability to recycle solvents like acetone and dichloromethane further minimizes the environmental footprint, aligning with increasingly stringent global environmental regulations. This combination of easy scalability and improved environmental compliance makes the technology a sustainable choice for long-term commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Dioxopromethazine Hydrochloride using the patented KHSO5 oxidation method. These insights are derived directly from the experimental data and beneficial effects reported in patent CN111943910B, providing clarity on process safety, purity profiles, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the consensus on how this specific chemical innovation resolves historical pain points in phenothiazine derivative synthesis.
Q: Why is potassium hydrogen persulfate preferred over hydrogen peroxide for this synthesis?
A: Potassium hydrogen persulfate (KHSO5) offers superior operational safety compared to hydrogen peroxide systems which often require dangerous heating or reflux conditions. KHSO5 operates effectively at lower temperatures without pressurization, significantly reducing energy consumption and equipment stress while maintaining high oxidation efficiency.
Q: How does this method address the issue of heavy metal contamination?
A: Unlike alternative methods utilizing palladium acetate catalysts, this KHSO5-mediated oxidation is metal-free. This eliminates the risk of palladium poisoning in the final product and removes the costly and complex downstream processing steps required to scavenge trace heavy metals to meet pharmaceutical regulatory limits.
Q: What purity levels can be achieved with the described refining process?
A: The patented refining protocol, which includes a specific cycle of base liberation, activated carbon decolorization in acetone, and re-salification, consistently yields Dioxopromethazine Hydrochloride with purity exceeding 98%, ensuring suitability for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dioxopromethazine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, safe, and cost-effective synthetic routes for high-value pharmaceutical intermediates like Dioxopromethazine Hydrochloride. Our technical team has thoroughly analyzed the KHSO5-mediated oxidation pathway and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this patent-protected methodology to life. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the >98% purity benchmark essential for downstream API synthesis. Our facility is equipped to handle the specific solvent systems and temperature controls demanded by this process, ensuring a seamless transition from laboratory scale to full commercial supply.
We invite global pharmaceutical partners to collaborate with us to leverage this superior manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can optimize your total cost of ownership. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can secure your supply chain for Dioxopromethazine Hydrochloride and drive value for your organization.
