Advanced Synthesis of Dapagliflozin Intermediate via Copper-Catalyzed Grignard Coupling
Advanced Synthesis of Dapagliflozin Intermediate via Copper-Catalyzed Grignard Coupling
The global demand for SGLT2 inhibitors, particularly Dapagliflozin, has necessitated the development of robust, high-yielding synthetic routes for its key intermediates. Patent CN109232199B discloses a breakthrough methodology for synthesizing 1-chloro-2-(4-ethoxybenzyl)-4-iodobenzene, a critical building block in the Dapagliflozin value chain. This novel approach leverages a copper-catalyzed Grignard coupling strategy that fundamentally alters the economic and technical landscape of producing this complex aromatic structure. By shifting away from traditional electrophilic aromatic substitution methods, the process achieves exceptional purity levels exceeding 99.5% and yields approaching 96%, addressing long-standing bottlenecks in pharmaceutical intermediate manufacturing. For R&D directors and supply chain leaders, this technology represents a significant opportunity to optimize production costs while ensuring consistent quality for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-chloro-2-(4-ethoxybenzyl)-4-iodobenzene has relied heavily on Friedel-Crafts acylation strategies, as documented in prior art such as WO03099836. These conventional routes typically begin with 5-bromo-2-chlorobenzoic acid, which is converted to an acid chloride and subsequently reacted with phenetole. A major technical drawback of this pathway is the inherent lack of regioselectivity during the acylation step. The reaction inevitably produces a mixture of para- and ortho-substituted ketones, with the unwanted ortho-isomer content reaching upwards of 12%. Separating these isomers is notoriously difficult due to their similar physical properties, often requiring multiple recrystallizations or chromatographic steps that drastically reduce overall yield. Furthermore, the subsequent reduction of the ketone to the methylene bridge often requires harsh conditions or expensive hydride sources, compounding the environmental burden and cost.
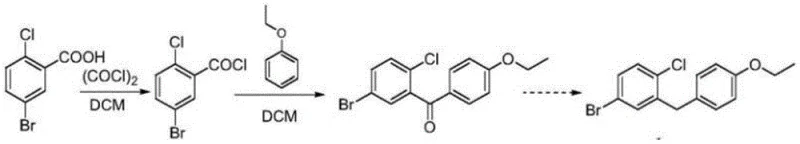
The Novel Approach
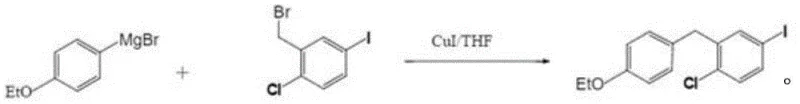
In stark contrast, the methodology described in CN109232199B circumvents these regiochemical challenges entirely by employing a nucleophilic substitution strategy. Instead of building the carbon skeleton through electrophilic attack on an aromatic ring, this route constructs the central C-C bond via the coupling of a Grignard reagent with a benzyl halide derivative. This strategic shift ensures that the substitution pattern is predetermined by the starting materials, effectively eliminating the formation of ortho-isomers. The process utilizes 4-bromophenyl ethyl ether and 2-(bromomethyl)-4-iodochlorobenzene as primary feedstocks. By reacting these precursors under mild conditions catalyzed by cuprous iodide, the synthesis achieves a direct and clean formation of the target diphenylmethane structure. This not only simplifies the purification workflow but also significantly enhances the atom economy of the overall transformation.
Mechanistic Insights into Copper-Catalyzed Grignard Coupling
The core of this innovative synthesis lies in the precise control of the organometallic species involved. The first stage involves the formation of the Grignard reagent from 4-bromophenyl ethyl ether and magnesium metal. Crucially, the patent specifies the use of elemental iodine as an initiator to activate the magnesium surface, ensuring rapid and complete formation of the organomagnesium species at temperatures between 40°C and 55°C. This activation step is vital for preventing the accumulation of unreacted magnesium, which can lead to inconsistent reaction kinetics in larger batches. Once formed, the Grignard reagent acts as a potent nucleophile, poised to attack the electrophilic benzylic carbon of the chloro-iodo-benzyl bromide derivative. The presence of the iodine atom on the benzyl ring is preserved throughout this sequence, demonstrating the chemoselectivity of the Grignard formation towards the bromide over the iodide in the specific context of the starting material design.
The second stage involves the actual coupling event, which is facilitated by the addition of cuprous iodide (CuI). The reaction temperature is critically controlled, dropping to between -15°C and -20°C before the addition of the electrophile. This low-temperature regime is essential for suppressing side reactions, particularly Wurtz-type homocoupling of the Grignard reagent or further metal-halogen exchange processes that could scramble the halogen pattern. The copper catalyst likely forms a transient organocuprate species (Gilman reagent analogue) in situ, which is softer and more selective than the bare Grignard reagent. This allows for efficient SN2-type displacement of the benzylic bromide without attacking the aryl chloride or iodide moieties. Following the addition, the reaction is allowed to warm to room temperature slowly, ensuring complete conversion while maintaining the integrity of the sensitive functional groups, resulting in the high purity observed in the final isolated product.
How to Synthesize 1-Chloro-2-(4-Ethoxybenzyl)-4-Iodobenzene Efficiently
Implementing this synthesis requires careful attention to solvent choice and addition rates to maximize safety and yield. The patent outlines a robust protocol using tetrahydrofuran (THF) or diethyl ether as the reaction medium, both of which stabilize the Grignard intermediate effectively. The molar ratio of the aryl bromide to magnesium is optimized between 1:1.05 and 1:1.2 to ensure full consumption of the halide without excessive waste of magnesium. The subsequent coupling step utilizes a slight excess of the Grignard reagent relative to the benzyl bromide (ratio 1:1 to 1:1.2) to drive the reaction to completion. Detailed standardized operating procedures regarding temperature ramps and quenching protocols are essential for reproducibility.
- Prepare the Grignard reagent by reacting 4-bromophenyl ethyl ether with magnesium chips in THF or ether, initiated by iodine at 40-55°C.
- Cool the Grignard solution to -15 to -20°C and add cuprous iodide catalyst.
- Slowly add 2-(bromomethyl)-4-iodochlorobenzene solution while maintaining low temperature, then warm to room temperature for final coupling.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly impact the bottom line for pharmaceutical manufacturers. The shift from acylation-based chemistry to Grignard coupling eliminates the need for corrosive and hazardous reagents such as oxalyl chloride and boron trifluoride etherate, which are common in the older Friedel-Crafts reduction pathways. This reduction in hazardous material handling translates to lower safety compliance costs and simplified waste management protocols. Furthermore, the starting materials, specifically 4-bromophenyl ethyl ether, are commodity chemicals that are readily available in the global market, reducing supply chain vulnerability compared to specialized benzoic acid derivatives required by alternative methods.
- Cost Reduction in Manufacturing: The elimination of complex purification steps required to remove ortho-isomers significantly lowers processing costs. In traditional routes, the 12% isomer impurity necessitates extensive chromatography or repeated crystallizations, which consume solvents, time, and labor. By achieving >99% purity directly from the reaction workup, this new method drastically reduces the operational expenditure associated with downstream processing. Additionally, the avoidance of expensive reducing agents like triethylsilane in favor of a direct coupling mechanism further contributes to substantial cost savings in raw material procurement.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as magnesium, iodine, and simple aryl bromides ensures a stable supply chain. Unlike routes dependent on custom-synthesized acid chlorides which may have long lead times, the feedstocks for this Grignard process are produced by multiple vendors globally. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate standard industrial solvents like THF and anisole, also means that the process can be easily transferred between different manufacturing sites without requiring specialized equipment modifications.
- Scalability and Environmental Compliance: The process is inherently scalable due to its straightforward exothermic profile and simple workup procedure involving acid quenching and organic extraction. The absence of heavy metal catalysts (other than trace copper which is easily removed) and carcinogenic solvents like 1,2-dichloroethane (used in some prior art) aligns with modern green chemistry principles. This facilitates easier regulatory approval for the manufacturing process and reduces the environmental footprint of the production facility. The high yield of nearly 96% maximizes the throughput of existing reactor volumes, allowing manufacturers to meet increasing market demand for Dapagliflozin intermediates without significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this route is superior for industrial application.
Q: How does this method improve purity compared to Friedel-Crafts acylation?
A: Traditional Friedel-Crafts routes generate difficult-to-separate ortho-isomers (up to 12%). This Grignard coupling method avoids positional isomerism entirely, achieving purities up to 99.5%.
Q: What are the cost advantages of this synthetic route?
A: The process utilizes readily available starting materials like 4-bromophenyl ethyl ether instead of expensive benzoic acid derivatives, and eliminates hazardous reagents like oxalyl chloride.
Q: Is this process scalable for industrial production?
A: Yes, the reaction uses standard solvents like THF and simple workup procedures (acid quench and extraction), making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Chloro-2-(4-Ethoxybenzyl)-4-Iodobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the synthesis of life-saving medications like Dapagliflozin. Our technical team has thoroughly evaluated the route described in CN109232199B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, such as ortho-isomers, guaranteeing that every batch supports your downstream API synthesis without failure.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this advanced Grignard coupling technology, we can help you optimize your supply chain for both cost and reliability. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
