Advanced Palladium-Catalyzed Synthesis of Multifunctional Pyrazolone Compounds for Commercial Scale-up
The pharmaceutical industry continuously seeks efficient methodologies to construct complex heterocyclic scaffolds, particularly those with proven biological relevance such as the pyrazolone core. A significant breakthrough in this domain is detailed in patent CN114478388A, which discloses a novel preparation method for multifunctional pyrazolone compounds. This technology leverages a sophisticated three-component decarboxylative cross-coupling reaction, merging pyrazolone exocyclic olefins, aryl allyl carbonates, and active methylene compounds under mild conditions. For R&D directors and process chemists, this represents a paradigm shift from traditional multi-step syntheses to a streamlined, atom-economical approach. The ability to generate highly functionalized drug-like skeletons in a single operational step not only accelerates lead generation but also offers a robust pathway for the commercial production of high-purity pharmaceutical intermediates. As a reliable pharmaceutical intermediate supplier, understanding such innovative synthetic routes is crucial for maintaining a competitive edge in the global supply chain.
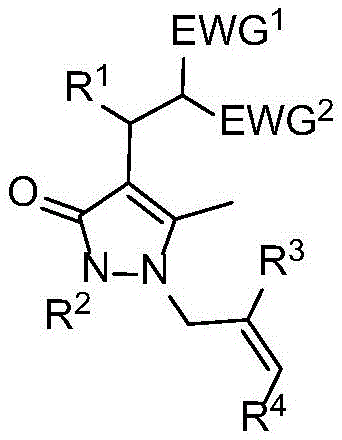
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polyfunctionalized pyrazolone derivatives has relied heavily on classical cyclization reactions or cycloadditions that often suffer from significant drawbacks. Traditional pathways frequently necessitate harsh reaction conditions, including elevated temperatures and strong acidic or basic environments, which can compromise the integrity of sensitive functional groups present in complex drug candidates. Furthermore, these conventional methods often lack regioselectivity and chemoselectivity, leading to the formation of difficult-to-separate isomeric mixtures that drastically reduce overall yield and purity. The requirement for pre-functionalized starting materials and multiple protection-deprotection sequences further inflates the cost and time associated with process development. For procurement managers, these inefficiencies translate into higher raw material costs and extended lead times, creating bottlenecks in the supply of critical API intermediates. Consequently, there is an urgent industrial need for methodologies that can bypass these limitations while maintaining high structural diversity.
The Novel Approach
The methodology described in the patent introduces a groundbreaking three-component decarboxylation cross-coupling reaction that effectively circumvents the pitfalls of legacy synthesis routes. By utilizing pyrazolone exocyclic olefins and aryl allyl carbonates as key building blocks, this novel approach enables the direct assembly of complex molecular architectures under exceptionally mild conditions, typically at room temperature. The reaction is driven by a synergistic catalytic system involving a transition metal and a phosphorus-containing ligand, which facilitates the in situ generation of reactive synthons without the need for external activating agents. This strategy not only simplifies the operational procedure but also ensures excellent chemical and regioselectivity, as evidenced by the high yields reported across a broad range of substrates. The visual representation of this transformative process highlights the convergence of three distinct components into a single, highly functionalized product, demonstrating the power of modern catalytic design in streamlining organic synthesis.
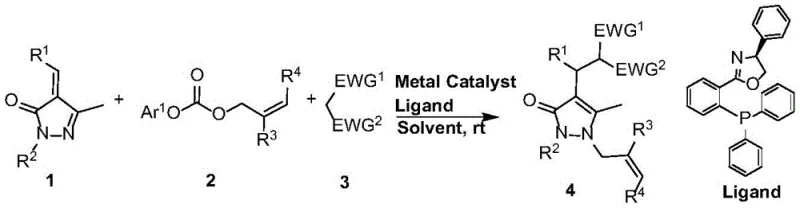
Mechanistic Insights into Pd-Catalyzed Decarboxylative Coupling
The success of this synthetic transformation hinges on the intricate interplay between the palladium catalyst and the specialized phosphorus-containing ligand. Mechanistically, the reaction initiates with the oxidative addition of the palladium species to the aryl allyl carbonate, followed by decarboxylation to generate a reactive pi-allyl palladium intermediate. This electrophilic species is then intercepted by the nucleophilic pyrazolone exocyclic olefin and the active methylene compound in a highly orchestrated sequence. The choice of ligand is paramount; chiral ligands, such as the Trost ligands or BINAP derivatives depicted in the patent data, create a specific chiral environment around the metal center. This steric and electronic tuning not only accelerates the reaction rate but also governs the stereochemical outcome, allowing for the precise construction of chiral centers which are often critical for biological activity. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal performance.
Furthermore, the impurity profile of the resulting pyrazolone derivatives is significantly improved due to the high selectivity of the catalytic cycle. Unlike non-catalytic thermal reactions that often produce polymeric byproducts or rearrangement products, this metal-catalyzed pathway channels the reactants through a defined transition state. The release of carbon dioxide as the sole stoichiometric byproduct further drives the reaction equilibrium forward and eliminates the need for extensive aqueous workups to remove salt waste. This clean reaction profile is particularly advantageous for the manufacture of high-purity OLED materials or pharmaceutical intermediates where trace impurities can have detrimental effects on downstream performance. The ability to control the reaction trajectory through ligand design ensures that the final product meets stringent quality specifications with minimal purification effort, thereby enhancing the overall sustainability of the manufacturing process.
How to Synthesize Multifunctional Pyrazolone Efficiently
Implementing this advanced synthesis route requires careful attention to reagent quality and reaction monitoring to ensure reproducibility on a larger scale. The patent outlines a straightforward protocol where the three components are combined in a polar organic solvent, such as tetrahydrofuran, in the presence of the palladium catalyst and ligand. The reaction proceeds efficiently at ambient temperature, eliminating the energy costs associated with heating or cooling. Detailed standard operating procedures for scaling this reaction from milligram to kilogram quantities are essential for technology transfer. For a comprehensive guide on the specific molar ratios, solvent choices, and purification techniques validated in the patent examples, please refer to the standardized synthesis steps provided below.
- Combine pyrazolone exocyclic olefin, aryl allyl carbonate, and nucleophilic active methylene compounds in a reaction vessel with a molar ratio of approximately 1: 1.5:2.
- Add a transition metal catalyst such as Pd2(dba)3·CHCl3 and a phosphorus-containing ligand, then dissolve in an organic solvent like tetrahydrofuran.
- Stir the mixture at room temperature for 1 to 48 hours, monitor by TLC, and purify the crude product via column chromatography to obtain the target pyrazolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this three-component coupling technology offers profound benefits for supply chain stability and cost management. The elimination of harsh reaction conditions and the reduction of synthetic steps directly correlate to a significant decrease in manufacturing overhead. By avoiding the use of expensive activating reagents and minimizing waste generation, the process aligns with green chemistry principles, which is increasingly becoming a requirement for regulatory compliance in major markets. For procurement managers, this translates into a more predictable cost structure and reduced dependency on specialized reagents that may be subject to supply volatility. The simplicity of the workup procedure, often requiring only standard column chromatography or crystallization, further reduces the burden on production facilities, allowing for faster turnaround times and increased throughput.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot reaction eliminates the need for intermediate isolation and purification, which are traditionally the most costly and time-consuming phases of chemical production. By consolidating multiple bond-forming events into a single operation, the consumption of solvents, labor, and energy is drastically reduced. Additionally, the high atom economy of the decarboxylative coupling ensures that a maximum proportion of the starting materials ends up in the final product, minimizing raw material waste. These factors collectively contribute to substantial cost savings in API manufacturing, making the final drug substance more economically viable to produce at a commercial scale.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including pyrazolone exocyclic olefins and aryl allyl carbonates, are structurally simple and can be sourced from a wide range of chemical suppliers. This abundance of raw materials mitigates the risk of supply chain disruptions that often plague processes relying on exotic or proprietary reagents. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch quality. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden challenges, but this methodology is inherently designed for scalability. The use of common solvents like THF and the absence of hazardous byproducts other than CO2 simplify the engineering controls required for large-scale reactors. The mild reaction conditions reduce the thermal load on the facility, lowering the risk of runaway reactions and enhancing operational safety. Moreover, the reduced environmental footprint supports corporate sustainability goals and facilitates easier permitting and regulatory approval, positioning manufacturers as responsible partners in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazolone synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Whether you are evaluating the feasibility of this route for a specific target molecule or assessing its potential impact on your supply chain, understanding these key aspects is essential. The answers below reflect the current state of the art as described in the intellectual property, offering a clear view of the capabilities and limitations of the method.
Q: What are the key advantages of this pyrazolone synthesis method over traditional cyclization?
A: Unlike traditional methods that often require harsh conditions or multi-step sequences, this patent describes a one-pot, three-component decarboxylative coupling. It operates at room temperature, utilizes readily available building blocks, and generates carbon dioxide as the only byproduct, significantly simplifying purification and reducing environmental impact.
Q: Is this method suitable for generating diverse chemical libraries for drug discovery?
A: Yes, the method exhibits a wide substrate scope. The patent demonstrates compatibility with various aryl, heteroaryl, and alkyl substituents on the pyrazolone olefin, allyl carbonate, and active methylene components. This modularity allows for the rapid construction of complex, drug-like molecular skeletons essential for lead optimization.
Q: What catalyst system is required for this transformation?
A: The reaction relies on a palladium catalyst, such as tris(dibenzylideneacetone)dipalladium-chloroform adduct, paired with a phosphorus-containing ligand. Chiral ligands like Trost ligands or BINAP derivatives can be employed to induce stereoselectivity, enabling the synthesis of enantiomerically enriched intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multifunctional Pyrazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN114478388A for accelerating drug development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in palladium-catalyzed cross-coupling reactions, we can help you optimize your synthesis routes for maximum efficiency and cost-effectiveness.
We invite you to collaborate with us to explore how this cutting-edge technology can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization. Together, we can overcome synthetic challenges and bring life-saving medicines to market faster.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
