Advanced N-Heterocyclic Benzophenone Derivatives for High-Performance OLED Display and Lighting Applications
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that combine high luminous efficiency with robust thermal and chemical stability. Patent CN110117283B introduces a groundbreaking class of N-heterocyclic benzophenone derivatives containing a hydrogenated phenothiazine group, specifically designed to address the critical limitations of conventional luminescent materials. These novel compounds exhibit a unique Aggregation Induced Emission (AIE) effect, which fundamentally alters how light is emitted in solid-state environments, preventing the efficiency roll-off often seen in traditional fluorophores. Furthermore, the structural integration of a bridged benzene ring between the N-containing aromatic anthryl and the carbonyl group creates a large conjugated plane that enhances fluorescence quantum yields. For R&D directors and procurement specialists in the display sector, this patent represents a significant opportunity to access high-purity OLED material precursors that promise superior device longevity and performance consistency across full-color display and solid-state lighting applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional luminescent materials used in the fabrication of organic light-emitting devices frequently suffer from inherent drawbacks that limit their commercial viability and operational lifespan. Many existing compounds demonstrate insufficient thermal stability, leading to degradation under the heat generated during prolonged device operation, which directly impacts the reliability of the final electronic product. Additionally, conventional fluorophores often exhibit poor solubility in common organic solvents, complicating the solution-processing steps required for thin-film deposition and uniform coating. This lack of solubility can result in uneven film morphology, causing defects in the display panel and reducing the overall yield of the manufacturing process. Moreover, standard materials often lack the specific spatial distortion required to inhibit intermolecular interactions that lead to fluorescence quenching, resulting in lower luminous intensity when the material is in a concentrated or solid state. These cumulative technical deficiencies necessitate frequent material replacements and complex engineering workarounds, driving up the total cost of ownership for display manufacturers.
The Novel Approach
The synthesis route disclosed in the patent offers a transformative solution by employing a precise two-step chemical strategy that ensures both structural integrity and functional performance. The process begins with a Michael cyclization reaction to construct the core imidazo-pyridine scaffold, followed by a palladium-catalyzed nucleophilic substitution to attach the hydrogenated phenothiazine group. 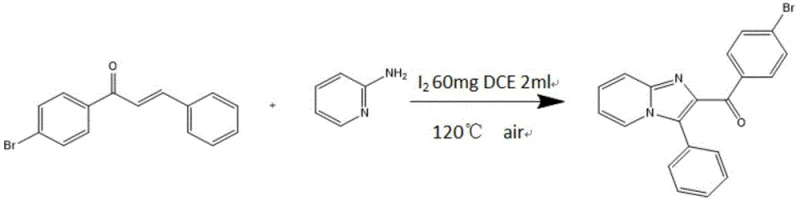 This specific pathway allows for the introduction of a highly twisted spatial structure that effectively suppresses concentration quenching. By utilizing readily available starting materials such as (E)-1-(4-bromophenyl)-3-phenylprop-2-en-1-one and 2-aminopyridine, the method ensures a stable supply chain foundation. The reaction conditions are optimized to operate at moderate temperatures, specifically around 120°C for the cyclization step, which minimizes energy consumption and reduces the risk of side reactions that could generate difficult-to-remove impurities. This novel approach not only streamlines the synthesis but also guarantees a product with the desired electronic properties for next-generation optoelectronic applications.
This specific pathway allows for the introduction of a highly twisted spatial structure that effectively suppresses concentration quenching. By utilizing readily available starting materials such as (E)-1-(4-bromophenyl)-3-phenylprop-2-en-1-one and 2-aminopyridine, the method ensures a stable supply chain foundation. The reaction conditions are optimized to operate at moderate temperatures, specifically around 120°C for the cyclization step, which minimizes energy consumption and reduces the risk of side reactions that could generate difficult-to-remove impurities. This novel approach not only streamlines the synthesis but also guarantees a product with the desired electronic properties for next-generation optoelectronic applications.
Mechanistic Insights into Pd-Catalyzed Nucleophilic Substitution
The core of the chemical innovation lies in the sophisticated molecular architecture that leverages C…H…π stacking interactions to favor luminescence. The presence of the hydrogenated phenothiazine group acts as a strong electron donor, while the benzophenone moiety serves as an electron acceptor, creating an intramolecular charge transfer (ICT) state that is crucial for efficient light emission. 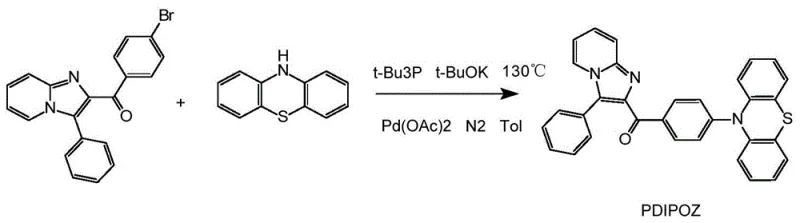 This donor-acceptor configuration is stabilized by the rigid conjugated plane formed by the bridged benzene ring, which enhances the thermal stability of the molecule significantly. The steric hindrance provided by the twisted structure prevents the molecules from packing too closely in the solid state, thereby maintaining high fluorescence intensity even at high concentrations. This mechanism is the key to the observed AIE effect, where the restriction of intramolecular motion in the aggregated state blocks non-radiative decay pathways. For technical teams, understanding this mechanistic nuance is vital for optimizing device layer thickness and doping concentrations to maximize external quantum efficiency without compromising the operational stability of the OLED stack.
This donor-acceptor configuration is stabilized by the rigid conjugated plane formed by the bridged benzene ring, which enhances the thermal stability of the molecule significantly. The steric hindrance provided by the twisted structure prevents the molecules from packing too closely in the solid state, thereby maintaining high fluorescence intensity even at high concentrations. This mechanism is the key to the observed AIE effect, where the restriction of intramolecular motion in the aggregated state blocks non-radiative decay pathways. For technical teams, understanding this mechanistic nuance is vital for optimizing device layer thickness and doping concentrations to maximize external quantum efficiency without compromising the operational stability of the OLED stack.
Impurity control is another critical aspect of this synthesis, achieved through the careful selection of catalysts and purification methods. The use of palladium acetate in conjunction with tri-tert-butylphosphine and potassium tert-butoxide in a toluene solvent system provides a highly selective environment for the coupling reaction. 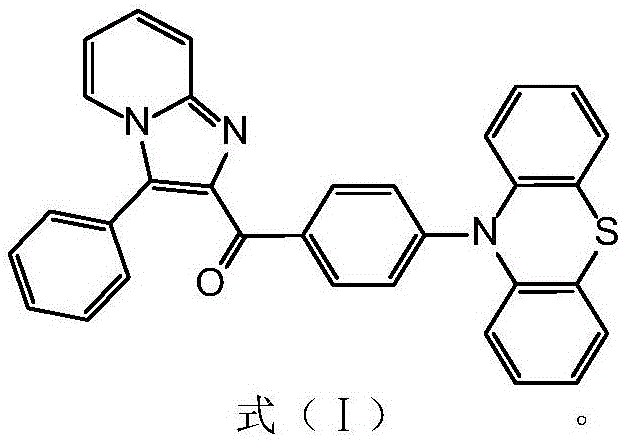 Post-reaction processing involves rigorous steps including distillation, extraction with dichloromethane, and drying with anhydrous magnesium sulfate to remove residual solvents and inorganic salts. The final purification via silica gel column chromatography using ethyl acetate and petroleum ether ensures that the resulting compound achieves a purity level of 99%. This high level of purity is essential for preventing trap states in the emissive layer of an OLED device, which can otherwise lead to efficiency loss and premature device failure. The robust nature of this purification protocol ensures that the material meets the stringent quality standards required by top-tier display manufacturers.
Post-reaction processing involves rigorous steps including distillation, extraction with dichloromethane, and drying with anhydrous magnesium sulfate to remove residual solvents and inorganic salts. The final purification via silica gel column chromatography using ethyl acetate and petroleum ether ensures that the resulting compound achieves a purity level of 99%. This high level of purity is essential for preventing trap states in the emissive layer of an OLED device, which can otherwise lead to efficiency loss and premature device failure. The robust nature of this purification protocol ensures that the material meets the stringent quality standards required by top-tier display manufacturers.
How to Synthesize N-Heterocyclic Benzophenone Efficiently
The synthesis of this high-performance luminescent molecule is designed to be reproducible and scalable, adhering to strict process parameters to ensure batch-to-batch consistency. The procedure involves precise molar ratios, such as 1:1.1 for the coupling reactants, and controlled atmospheric conditions using nitrogen protection to prevent oxidation of sensitive intermediates.
- Perform Michael cyclization of (E)-1-(4-bromophenyl)-3-phenylprop-2-en-1-one with 2-aminopyridine using iodine catalyst in dichloroethane at 120°C.
- Execute nucleophilic substitution between the intermediate and 10H-phenothiazine using palladium acetate and potassium tert-butoxide in toluene at 130°C.
- Purify the final crude product via silica gel column chromatography using ethyl acetate and petroleum ether to achieve 99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial advantages that directly impact the bottom line and operational reliability of manufacturing facilities. The synthesis route relies on raw materials that are commercially available and widely sourced, reducing the risk of supply chain disruptions caused by scarce or proprietary reagents. This accessibility ensures a continuous flow of materials necessary for sustained production schedules, which is critical for meeting the high-volume demands of the consumer electronics market. Furthermore, the process eliminates the need for exotic or extremely hazardous reagents that would require specialized handling and disposal infrastructure, thereby simplifying compliance with environmental and safety regulations. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in operational parameters, leading to higher overall process reliability and reduced waste generation.
- Cost Reduction in Manufacturing: The streamlined two-step synthesis significantly reduces the complexity of the production process compared to multi-step routes often required for similar high-performance materials. By avoiding the use of expensive transition metal removal steps typically associated with less selective catalysts, the overall processing cost is drastically simplified. The high yield and purity achieved reduce the need for extensive re-processing or recycling of off-spec material, leading to substantial cost savings in raw material utilization. Additionally, the use of common solvents like toluene and dichloroethane allows for efficient solvent recovery and reuse systems, further driving down the variable costs associated with large-scale chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable and widely available chemical precursors ensures that the supply chain is resilient against market volatility. Unlike materials dependent on rare earth elements or highly specialized custom synthesis, this derivative can be produced by multiple qualified chemical manufacturers, fostering a competitive supply environment. This redundancy is crucial for procurement managers seeking to mitigate risks associated with single-source dependencies. The scalability of the process from gram to tonnage scale ensures that supply can be ramped up quickly to meet sudden increases in demand from downstream display panel producers without long lead times for capacity expansion.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are manageable in standard industrial reactors. The waste profile is well-defined, consisting primarily of organic solvents and inorganic salts that can be treated using standard industrial waste management protocols. This facilitates easier permitting and compliance with increasingly strict environmental regulations in major manufacturing hubs. The high thermal stability of the final product also reduces the risk of degradation during storage and transport, minimizing losses due to spoilage and ensuring that the material arrives at the customer's site in optimal condition for immediate use in device fabrication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this N-heterocyclic benzophenone derivative in industrial applications. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a reliable basis for decision-making.
Q: What are the thermal stability characteristics of this N-heterocyclic benzophenone derivative?
A: The derivative exhibits exceptional thermal stability with a 5% weight loss temperature of 404.03°C and a glass transition temperature of 211.6°C, making it highly suitable for demanding OLED operational environments.
Q: How does the Aggregation Induced Emission (AIE) effect benefit device performance?
A: The AIE effect ensures that fluorescence intensity is significantly enhanced in high-concentration aggregation states, effectively inhibiting exciton annihilation and maintaining high luminous intensity in solid-state devices.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes commercially available raw materials and standard solvent systems like toluene and dichloroethane, facilitating straightforward scale-up from laboratory to commercial tonnage without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Heterocyclic Benzophenone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to handle the synthesis of complex OLED materials like the N-heterocyclic benzophenone derivative, ensuring that stringent purity specifications are met through our rigorous QC labs. We understand the critical nature of supply continuity in the electronics sector and have established robust logistics networks to guarantee timely delivery. Our commitment to quality means that every batch is thoroughly characterized to match the performance metrics outlined in the patent, providing our partners with the confidence needed to integrate these materials into high-value display technologies.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By contacting our technical procurement team, you can request specific COA data and route feasibility assessments to verify how this material can enhance your product portfolio. Let us help you optimize your supply chain with high-performance materials that drive innovation in the global display market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
