Scalable Synthesis of Thiencarbazone-methyl via N-Alkylimidazole Catalysis for Global Agrochemical Supply
Scalable Synthesis of Thiencarbazone-methyl via N-Alkylimidazole Catalysis for Global Agrochemical Supply
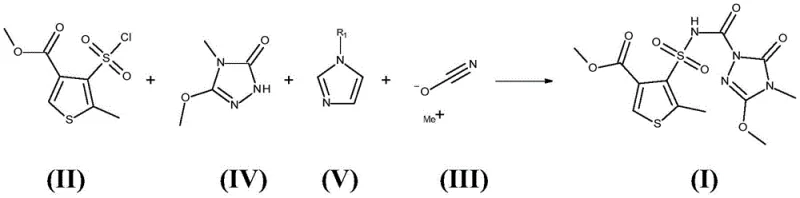
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates. A pivotal advancement in this sector is detailed in patent CN110352193B, which discloses a robust and highly selective method for preparing thiencarbazone-methyl. This compound, chemically known as 4-[(4,5-dihydro-3-methoxy-4-methyl-5-oxo-1H-1,2,4-triazol-1-yl)carbonyl)sulfamoyl]-5-methylthiophene-3-carboxylate, serves as a critical active ingredient in modern weed control formulations. The patent highlights a transformative shift from traditional, hazardous synthetic routes to a streamlined process utilizing N-alkylimidazole bases. For R&D directors and procurement specialists, this technology represents a significant opportunity to enhance process safety while securing a reliable supply of high-purity intermediates. By leveraging the unique catalytic properties of N-methylimidazole, manufacturers can achieve exceptional yields without the logistical burdens associated with toxic reagents like phosgene.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted thiophen-3-ylsulfonylaminocarbonyltriazolinones has been fraught with significant chemical and operational challenges. Prior art, such as that described in DE 10 2004 063192 A1, often relied on the use of phosgene to generate sulfonyl isocyanate intermediates from sulfonamides. Phosgene is a highly toxic gas that requires specialized containment infrastructure, rigorous safety protocols, and complex waste treatment systems, all of which drastically inflate capital and operational expenditures. Furthermore, alternative methods involving the reaction of sulfonyl chlorides with metal cyanates in the presence of conventional bases like pyridine, tributylamine, or inorganic alkoxides have demonstrated poor efficiency. Experimental comparisons reveal that using bases such as potassium tert-butoxide results in negligible product formation, while pyridine derivatives yield only modest conversions. These inefficiencies lead to complex purification workflows, increased solvent consumption, and inconsistent batch quality, creating bottlenecks for commercial scale-up of complex agrochemical intermediates.
The Novel Approach
The methodology outlined in CN110352193B introduces a paradigm shift by employing 1-substituted imidazole bases, specifically N-alkylimidazoles, as the primary reaction auxiliaries. This innovative approach enables the direct conversion of 4-methoxycarbonyl-2-methylthiophene-3-sulfonyl chloride into the target thiencarbazone-methyl in a highly efficient manner. The process can be operated as a convenient one-pot synthesis or a controlled two-step sequence, offering flexibility depending on production requirements. Crucially, the use of N-methylimidazole or N-butylimidazole facilitates the in situ generation of the reactive sulfonyl isocyanate species without the need for toxic phosgene. This not only mitigates severe safety risks but also simplifies the reaction profile, allowing for high selectivity even in the presence of competing nucleophiles. The result is a streamlined manufacturing pathway that significantly reduces lead time for high-purity agrochemical intermediates while ensuring environmental compliance.
Mechanistic Insights into N-Alkylimidazole Catalyzed Sulfonyl Isocyanate Formation
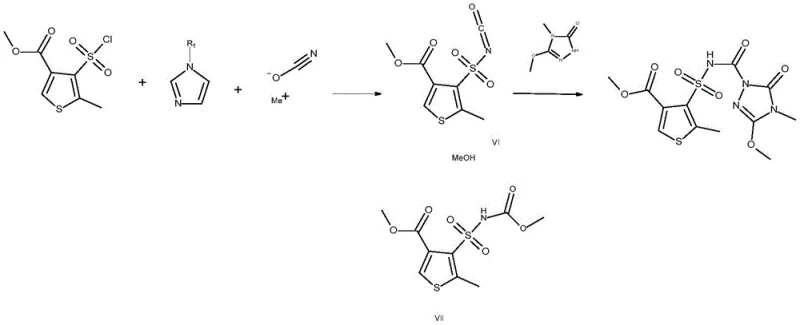
The core scientific breakthrough of this patent lies in the unexpected selectivity and activation capability of N-alkylimidazoles during the formation of the sulfonyl isocyanate intermediate. Mechanistically, the reaction involves the interaction of the sulfonyl chloride with a metal cyanate, typically sodium cyanate (NaOCN), which acts as an oxygen nucleophile. In conventional systems, the basicity of the reaction medium often leads to side reactions or decomposition. However, the presence of an N-alkylimidazole creates a unique microenvironment that stabilizes the transition state. It is hypothesized that the imidazole nitrogen coordinates with the sulfur center, enhancing the electrophilicity of the sulfonyl group and facilitating the displacement of the chloride ion by the cyanate. This generates the sulfonyl isocyanate intermediate (Formula VI) with remarkable efficiency. Unlike other nitrogen bases, N-alkylimidazoles do not interfere with the subsequent nucleophilic attack by the triazolinone ring, ensuring that the coupling reaction proceeds rapidly to form the final urea linkage.
Furthermore, the impurity profile of the reaction is tightly controlled by the specific choice of the N-alkyl substituent. The patent data indicates that unsubstituted or benzyl-substituted imidazoles are less effective than straight-chain alkyl variants like methyl or butyl. This suggests that steric and electronic factors play a critical role in preventing the hydrolysis of the isocyanate or the formation of symmetric sulfonamide byproducts. By maintaining the reaction temperature between 50°C and 80°C, preferably in polar aprotic solvents like acetonitrile or THF, the kinetic energy is sufficient to drive the reaction forward without triggering thermal degradation. This precise control over the reaction mechanism ensures that the final product precipitates cleanly from the solution, often as an insoluble salt upon the addition of a mild inorganic base, thereby simplifying downstream processing and maximizing overall recovery.
How to Synthesize Thiencarbazone-methyl Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and addition sequences to maximize the benefits of the N-alkylimidazole catalysis. The process is designed to be robust, tolerating slight variations in conditions while maintaining high purity standards. Operators can choose between a direct one-pot method, where all components are mixed and heated, or a sequential addition strategy to better manage exotherms. The detailed standard operating procedures for scaling this reaction from laboratory to pilot plant are critical for ensuring reproducibility and safety. For a comprehensive breakdown of the specific molar ratios, solvent choices, and workup protocols validated in the patent examples, please refer to the technical guide below.
- Mix 4-(chlorosulfonyl)-5-methylthiophene-3-carboxylate, sodium cyanate, N-methylimidazole, and 5-methoxy-4-methyl-2,4-dihydro-3H-1,2,4-triazol-3-one in acetonitrile.
- Heat the reaction mixture to 70°C and stir for 12 to 18 hours to facilitate the formation of the sulfonyl isocyanate intermediate and subsequent coupling.
- Cool the mixture, add sodium bicarbonate to precipitate the product salt, filter, wash with acid and water, and dry to obtain high-purity thiencarbazone-methyl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this N-alkylimidazole mediated synthesis offers profound strategic advantages beyond mere chemical yield. The elimination of phosgene from the supply chain removes a major regulatory and logistical hurdle, reducing the need for specialized storage and transport certifications. This simplification directly translates to cost reduction in herbicide manufacturing by lowering insurance premiums and safety compliance overheads. Additionally, the ability to use commercially available reagents like sodium cyanate and N-methylimidazole ensures a stable supply base, reducing the risk of raw material shortages that often plague specialty chemical production. The high selectivity of the reaction minimizes the generation of hazardous waste streams, aligning with increasingly stringent global environmental regulations and reducing disposal costs.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by eliminating the need for expensive and hazardous reagents like phosgene, which require complex handling infrastructure. The high reaction selectivity minimizes the formation of byproducts, thereby reducing the consumption of solvents and energy required for purification steps such as chromatography or extensive recrystallization. Furthermore, the ability to isolate the product via simple filtration of a precipitated salt streamlines the workflow, lowering labor costs and increasing throughput capacity per batch.
- Enhanced Supply Chain Reliability: By relying on widely available commodity chemicals such as sodium cyanate and N-methylimidazole, manufacturers can mitigate the risks associated with sourcing specialized or controlled precursors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, ensures consistent production schedules even when facing minor fluctuations in utility availability. This reliability is crucial for maintaining continuous supply to downstream formulators, preventing stockouts of critical agrochemical active ingredients during peak seasonal demand.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis significantly reduces the reactor occupancy time and the number of unit operations required, making it highly amenable to large-scale commercial production. The process generates fewer organic waste streams compared to multi-step alternatives, facilitating easier compliance with environmental discharge limits. The precipitation-based isolation method avoids the use of large volumes of extraction solvents, further enhancing the green chemistry profile of the manufacturing process and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on catalyst selection, safety profiles, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or process optimization.
Q: Why is N-methylimidazole preferred over traditional bases like pyridine for this synthesis?
A: N-methylimidazole acts as a superior activator and stabilizer for the sulfonyl isocyanate intermediate. Unlike pyridine or inorganic bases which result in low yields or no reaction, N-alkylimidazoles ensure high selectivity and prevent side reactions, achieving yields up to 84%.
Q: Does this process require the isolation of toxic sulfonyl isocyanate intermediates?
A: No, the patent describes a convenient one-pot procedure where the sulfonyl isocyanate is generated in situ and immediately reacted with the triazolinone. This eliminates the need to isolate hazardous intermediates, significantly improving operational safety.
Q: What are the purity specifications achievable with this new method?
A: The method allows for the direct precipitation of the product as a salt, which can be easily filtered and acidified. Experimental data demonstrates that this workflow consistently delivers thiencarbazone-methyl with purity levels exceeding 98%, often reaching 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiencarbazone-methyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes for high-value agrochemical intermediates like thiencarbazone-methyl. Our technical team has extensively analyzed the advancements presented in CN110352193B and possesses the expertise to implement this N-alkylimidazole catalyzed process at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in tangible supply. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for global herbicide registration and formulation.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing technology can optimize your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your production of thiencarbazone-methyl is both economically competitive and technically superior.
