Advanced Rare Earth Catalysis for Scalable Nitro-Aromatic Manufacturing Solutions
Introduction to Green Nitration Technology
The chemical industry is currently undergoing a significant paradigm shift towards sustainable manufacturing processes, particularly in the synthesis of high-value nitro-aromatic compounds which serve as critical building blocks for pharmaceuticals and agrochemicals. A pivotal development in this field is documented in Chinese Patent CN1166620C, which introduces a novel environmentally friendly synthesis method utilizing rare earth metal salts of perfluoroalkanesulfonic acid as catalysts. This technology represents a substantial departure from conventional methodologies by replacing corrosive mixed acid systems with water-tolerant Lewis acids that offer superior selectivity and reduced environmental impact. For R&D directors and process chemists, understanding the mechanistic nuances of this catalytic system is essential for designing next-generation synthetic routes that align with modern green chemistry principles. The patent elucidates how specific fluorocarbon chain lengths (C7-10) on the sulfonic acid ligand enhance the surface activity of the catalyst, facilitating efficient reactions even in biphasic systems where organic substrates and aqueous nitric acid typically struggle to interact effectively.
Furthermore, the economic implications of adopting this rare earth catalyzed nitration method are profound for procurement and supply chain stakeholders who are constantly seeking reliable pharmaceutical intermediates supplier partners capable of delivering cost-effective solutions. By eliminating the need for large quantities of sulfuric acid and mitigating the generation of hazardous waste streams, manufacturers can achieve significant operational savings while adhering to increasingly stringent environmental regulations. The versatility of this approach allows for the functionalization of diverse aromatic cores, including substituted benzenes and polycyclic hydrocarbons, making it a robust platform technology for the production of complex fine chemical intermediates. As we delve deeper into the technical specifics, it becomes clear that this innovation not only solves immediate processing challenges but also lays the groundwork for more sustainable and scalable industrial operations in the future.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of nitro-substituted aromatic compounds has relied heavily on the use of mixed acid technology, which combines concentrated nitric acid with sulfuric acid to generate the active nitronium ion species. While this method has served the industry for decades, it is plagued by inherent deficiencies that pose significant risks to both operational safety and environmental compliance. The primary drawback is the generation of massive quantities of spent sulfuric acid, which requires costly neutralization or regeneration processes, thereby inflating the overall production costs and creating a heavy burden on waste management infrastructure. Additionally, the highly corrosive nature of the mixed acid system necessitates the use of specialized, expensive reactor materials and rigorous safety protocols to prevent equipment failure and potential chemical leaks. From a chemical selectivity standpoint, traditional mixed acid nitration often suffers from poor regioselectivity, leading to complex mixtures of ortho, meta, and para isomers that require energy-intensive separation techniques to isolate the desired product. Furthermore, the harsh oxidative conditions frequently promote over-nitration or oxidation side reactions, resulting in lower yields and the formation of difficult-to-remove impurities that can compromise the quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes rare earth perfluoroalkanesulfonates, such as Yb(OPf)3 or La(OPf)3, to catalyze the nitration reaction under much milder and cleaner conditions. This innovative strategy leverages the unique Lewis acidity of lanthanide ions combined with the lipophilic character of long-chain perfluoroalkyl groups to create a highly efficient catalytic environment. The use of these catalysts allows for the direct use of 65% nitric acid without the need for sulfuric acid dehydration agents, fundamentally simplifying the reaction mixture and eliminating the associated waste acid problems. Experimental data indicates that this system can operate effectively in organic solvents or even under solvent-free conditions, providing flexibility in process design that is unavailable with traditional methods. The visual representation of this transformation highlights the clean conversion of aromatic substrates to their nitro-derivatives with improved control over substitution patterns.
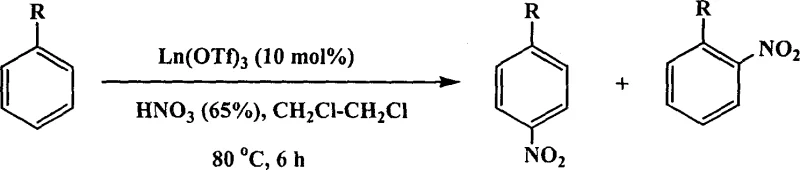
Moreover, the catalyst loading required is exceptionally low, often in the range of 0.00005 to 0.005 molar equivalents, which drastically reduces the raw material costs associated with the catalytic system. The ability of these rare earth salts to tolerate water is another critical advantage, as it permits the use of aqueous nitric acid directly without the need for rigorous drying of reagents, further streamlining the operational workflow. This combination of high efficiency, low toxicity, and ease of handling makes the rare earth catalyzed method a superior alternative for the modern manufacture of high-purity OLED material precursors and other specialty chemicals.
Mechanistic Insights into Rare Earth Lewis Acid Catalyzed Nitration
The mechanistic foundation of this technology rests on the ability of the rare earth metal center to act as a potent Lewis acid, coordinating with the oxygen atoms of the nitric acid molecule to facilitate the generation of the electrophilic nitronium ion or a highly activated nitric acid complex. Unlike traditional Lewis acids such as aluminum chloride or boron trifluoride, which are notoriously sensitive to moisture and decompose rapidly in the presence of water, the perfluoroalkanesulfonic acid rare earth salts exhibit remarkable hydrolytic stability. This stability is attributed to the strong electron-withdrawing nature of the perfluoroalkyl group, which stabilizes the metal-ligand bond and prevents the displacement of the sulfonate anion by water molecules. Consequently, the catalytic cycle can proceed efficiently in the presence of the water produced during the nitration reaction, maintaining high turnover numbers throughout the process. The specific choice of the rare earth element, such as Ytterbium, Lanthanum, or Scandium, influences the Lewis acidity strength and the steric environment around the active site, allowing for fine-tuning of the reaction rate and selectivity.
Impurity control is another area where this mechanistic approach offers distinct advantages over conventional pathways. The mild reaction conditions, typically conducted at temperatures between 50°C and 100°C, minimize the thermal energy available for side reactions such as oxidation of the aromatic ring or polymerization of reactive intermediates. The high regioselectivity observed, particularly the preference for para-substitution in alkylbenzenes like toluene, suggests that the bulky perfluoroalkyl chains on the catalyst may exert a steric influence that directs the incoming nitro group to the less hindered position. This intrinsic selectivity reduces the burden on downstream purification processes, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications. By understanding these mechanistic details, process engineers can optimize reaction parameters to maximize yield and minimize the formation of by-products, thereby enhancing the overall robustness of the manufacturing process.
How to Synthesize Nitro-Aromatic Compounds Efficiently
Implementing this advanced nitration protocol requires careful attention to reaction parameters to fully realize its potential for cost reduction in pharmaceutical intermediates manufacturing. The general procedure involves combining the aromatic substrate with 65% nitric acid and a catalytic amount of the rare earth salt, followed by heating under reflux conditions. Detailed standard operating procedures regarding specific stoichiometry, temperature profiles, and workup techniques are outlined in the structured guide below, which serves as a reference for scaling this technology from laboratory benchtop to pilot plant operations.
- Mix the aromatic substrate (e.g., toluene) with 65% concentrated nitric acid in a reactor.
- Add the rare earth catalyst, such as Yb(OPf)3 or La(OPf)3, at a molar ratio of 0.00005 to 0.005 relative to the substrate.
- Heat the mixture to 50-100°C (typically 70°C) and stir for 10 to 100 hours, followed by aqueous workup and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rare earth catalyzed nitration technology presents a compelling value proposition centered around risk mitigation and long-term cost efficiency. The elimination of sulfuric acid from the process equation removes a major logistical and safety hazard, simplifying the storage and handling requirements for raw materials and reducing the regulatory burden associated with hazardous waste disposal. This simplification translates directly into lower operational expenditures, as facilities no longer need to invest in extensive acid recovery units or pay for the transportation and treatment of large volumes of spent acid. Furthermore, the reusability of the catalyst means that the effective cost per kilogram of product decreases significantly over time, as the initial investment in the rare earth salt is amortized over multiple production batches. This economic model supports a more predictable pricing structure for customers seeking reliable agrochemical intermediate supplier partnerships, shielding them from the volatility associated with waste management fees.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in auxiliary material consumption and waste treatment expenses. By avoiding the use of sulfuric acid, manufacturers eliminate the need for neutralization bases and the associated sludge disposal costs, which can constitute a significant portion of the total production budget. Additionally, the low catalyst loading and potential for catalyst recovery mean that the expense of the rare earth metal is negligible compared to the savings gained from simplified processing. The ability to run reactions without additional organic solvents in certain configurations further reduces the cost of goods sold by minimizing solvent purchase, recovery, and loss. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable raw materials, specifically 65% nitric acid and common aromatic feedstocks, which are less subject to supply disruptions than specialized dehydrating agents. The robustness of the catalyst against moisture ensures that production schedules are not delayed by the need for rigorously anhydrous conditions, allowing for more flexible and continuous manufacturing operations. This reliability is crucial for maintaining consistent delivery timelines to downstream customers who depend on a steady flow of high-purity nitro-aromatic intermediates for their own synthesis campaigns. Moreover, the simplified process flow reduces the number of unit operations required, decreasing the likelihood of mechanical failures or bottlenecks that could interrupt supply continuity.
- Scalability and Environmental Compliance: Scaling this technology to commercial levels is facilitated by the mild reaction conditions and the absence of highly corrosive media, which allows for the use of standard stainless steel reactors rather than exotic alloys. This compatibility with existing infrastructure lowers the capital expenditure barrier for expanding production capacity to meet growing market demand. From an environmental perspective, the process aligns perfectly with green chemistry mandates by generating only water and the desired nitro product as by-products, with no toxic emissions or persistent organic pollutants. This clean profile ensures compliance with global environmental standards, reducing the risk of regulatory fines and enhancing the corporate sustainability image of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rare earth catalyzed nitration technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the advantages of using rare earth perfluoroalkanesulfonates over traditional mixed acids?
A: Unlike traditional mixed acid nitration which generates large amounts of waste sulfuric acid and suffers from corrosion issues, this method uses water-tolerant Lewis acids that can be recycled, significantly reducing environmental pollution and equipment maintenance costs.
Q: Is the catalyst reusable in this nitration process?
A: Yes, the patent highlights that the rare earth metal salts of perfluoroalkanesulfonic acid maintain catalytic activity after recovery, allowing for multiple cycles of use which enhances the economic viability of the process.
Q: What types of aromatic substrates are compatible with this method?
A: The technology demonstrates broad substrate scope, successfully nitrating toluene, anisole, chlorobenzene, bromobenzene, xylene, biphenyl, and polycyclic aromatics like naphthalene and anthracene derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitro-Aromatic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the rare earth perfluoroalkanesulfonate system in driving the next generation of chemical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards for pharmaceutical and agrochemical intermediates. We understand that transitioning to a new catalytic platform requires confidence in the supplier's ability to manage complexity, and our team is equipped to handle the nuances of rare earth chemistry to deliver consistent results.
We invite you to engage with our technical procurement team to discuss how this green nitration technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this catalytic route for your target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain efficiency and your environmental footprint.
