Advanced Synthesis of N-Acryloyl Succinimide for High-Performance Polymer Applications
Advanced Synthesis of N-Acryloyl Succinimide for High-Performance Polymer Applications
The chemical landscape for high-performance polymer additives is constantly evolving, driven by the need for materials that can withstand extreme thermal and chemical environments. A significant breakthrough in this domain is documented in Chinese Patent CN103086941A, which discloses a robust and efficient synthetic method for N-acryloyl succinimide. This compound represents a critical advancement over existing N-substituted imides, primarily due to the incorporation of a reactive acryl group that facilitates superior cross-linking capabilities. As a reliable polymer intermediate supplier, understanding the nuances of this synthesis is vital for developing next-generation materials that offer enhanced thermostability and resistance to acidic and alkaline degradation. The patent outlines a streamlined pathway that avoids the pitfalls of traditional methods, ensuring high yields and exceptional structural integrity of the final monomer.
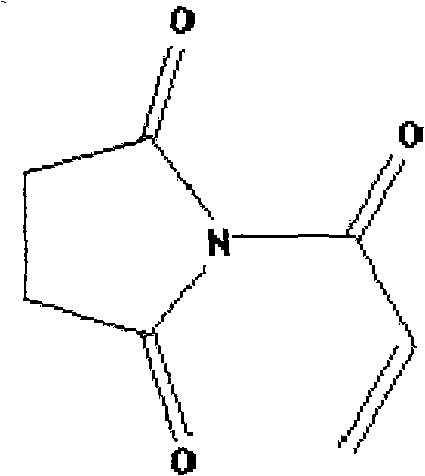
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of N-substituted imide compounds has been plagued by limitations regarding their functional versatility and thermal performance. Traditional N-alkyl or N-aryl imides, while useful, often lack the reactive sites necessary for creating complex copolymer networks that demand high durability. Furthermore, conventional synthesis routes frequently involve harsh conditions that can degrade sensitive functional groups or result in significant byproduct formation, complicating downstream purification. The absence of a polymerizable double bond in many standard imides restricts their application to simple blending rather than chemical integration into the polymer backbone. This limitation often results in phase separation issues and reduced mechanical strength in the final material, failing to meet the rigorous standards required for aerospace, automotive, and advanced electronic applications where thermal stability is non-negotiable.
The Novel Approach
The methodology presented in the patent introduces a sophisticated two-step strategy that effectively overcomes these historical barriers by utilizing succinic anhydride and acrylamide as primary feedstocks. This novel approach leverages a controlled acylation followed by a catalytic dehydration cyclization, allowing for the precise construction of the imide ring while preserving the critical vinyl functionality. By carefully selecting solvents and reaction parameters, the process minimizes side reactions and maximizes the retention of the unsaturated double bond, which is essential for subsequent polymerization. This route not only simplifies the operational workflow but also enhances the economic viability of producing high-purity monomers. The ability to tune reaction conditions, such as temperature and catalyst loading, provides manufacturers with the flexibility to optimize for either speed or purity, making it a highly adaptable solution for cost reduction in polymer additive manufacturing.
Mechanistic Insights into Acylation and Dehydration Cyclization
The core of this synthesis lies in the sequential transformation of raw materials into the target imide structure through well-defined mechanistic steps. The initial phase involves the nucleophilic attack of the amide nitrogen of acrylamide on one of the carbonyl carbons of succinic anhydride. This acylation reaction opens the anhydride ring, forming an intermediate amic acid derivative. The reaction is typically conducted in polar aprotic or aromatic solvents like dimethylformamide or toluene at moderate temperatures ranging from 35°C to 65°C. Maintaining this temperature window is crucial; it provides sufficient energy to overcome the activation barrier for ring opening without initiating premature polymerization of the acryl moiety. The stoichiometry is carefully balanced, often using a slight excess of succinic anhydride (ratio 1:1 to 1:1.3) to drive the equilibrium towards the formation of the open-chain intermediate.
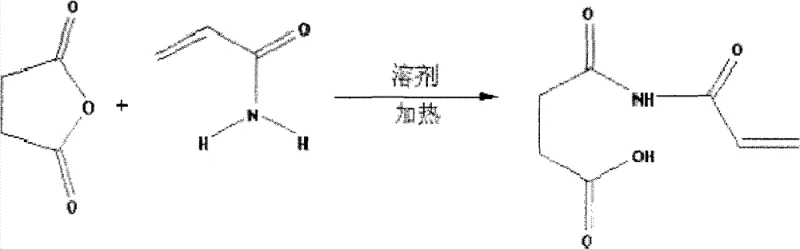
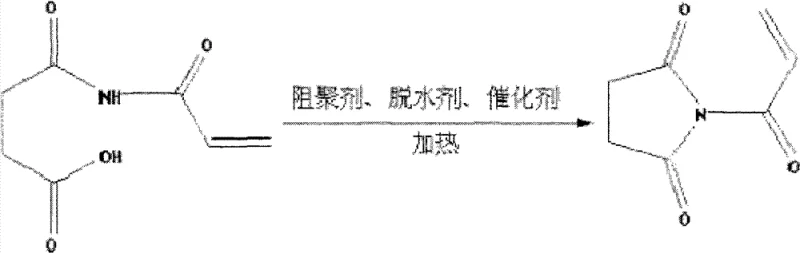
Following the acylation, the system undergoes a dehydration cyclization to close the five-membered imide ring. This step is chemically demanding as it requires the removal of a water molecule to form the stable imide bond, a process facilitated by the addition of specific dehydrating agents and catalysts. Crucially, this stage employs polymerization inhibitors such as hydroquinone or MEHQ to protect the reactive double bond from radical attack. The presence of catalysts like zinc acetate or triethylamine lowers the energy requirement for cyclization, allowing the reaction to proceed efficiently at temperatures between 40°C and 75°C. Simultaneous removal of generated water, often via azeotropic distillation or chemical dehydration, shifts the equilibrium towards the product, ensuring high conversion rates. This delicate balance of catalysis and inhibition is what allows for the high retention rate of the unsaturated bond, often exceeding 92%, which is a key quality metric for the monomer's downstream utility.
How to Synthesize N-Acryloyl Succinimide Efficiently
Executing this synthesis requires precise control over reaction parameters to ensure safety and product quality. The process begins with the uniform mixing of succinic anhydride and acrylamide in a selected solvent system, followed by a sustained heating period to complete the acylation. Once the intermediate is formed, the introduction of the inhibitor-dehydrator-catalyst cocktail triggers the ring-closing event. It is imperative to monitor the reaction progress closely to prevent overheating, which could lead to polymerization or decomposition. After the cyclization is complete, the solvent is removed under reduced pressure to isolate the crude solid. The final purification step involves recrystallization, typically using water or alcohols, which removes unreacted starting materials and byproducts, yielding a white crystalline powder of high purity. For detailed operational parameters and specific embodiment data, refer to the standardized guide below.
- Mix succinic anhydride and acrylamide in a suitable solvent (e.g., toluene, DMF) at 35-65°C for 4.5-6.5 hours to perform acylation.
- Add polymerization inhibitor, dehydrating agent, and catalyst, then heat to 40-75°C for 0.5-2 hours to effect dehydration ring-closure while removing water.
- Remove solvent under reduced pressure and purify the crude product via recrystallization in solvents like deionized water or alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this synthetic route offers distinct advantages that translate directly into operational efficiency and risk mitigation. The use of readily available commodity chemicals like succinic anhydride and acrylamide ensures a stable and resilient supply base, reducing the vulnerability to raw material shortages that often plague specialty chemical manufacturing. Furthermore, the reaction conditions are relatively mild, operating at temperatures well below 100°C, which significantly reduces energy consumption compared to high-temperature imidization processes. This energy efficiency, combined with the high atom economy of the cyclization step, contributes to substantial cost savings in large-scale production environments. The robustness of the process also implies fewer batch failures and more consistent output, which is critical for maintaining just-in-time delivery schedules for downstream polymer producers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reliance on common organic acids and bases for catalysis drastically lowers the input cost per kilogram. Additionally, the high yield and purity achieved reduce the burden on waste treatment facilities and minimize the loss of valuable material during purification. By avoiding complex multi-step protections and deprotections often seen in fine chemical synthesis, the overall process intensity is reduced, leading to lower capital expenditure requirements for reactor infrastructure and simplified maintenance protocols.
- Enhanced Supply Chain Reliability: The flexibility in solvent selection, allowing for the use of toluene, dioxane, or acetic acid, provides procurement teams with the agility to switch suppliers based on market availability and pricing without compromising the reaction outcome. This adaptability ensures continuity of supply even when specific solvent markets experience volatility. Moreover, the stability of the intermediates allows for potential semi-batch processing strategies, further smoothing out production bottlenecks and ensuring a steady flow of finished goods to meet customer demand fluctuations.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous streams from recrystallization and spent solvents that can be recovered and recycled. The absence of heavy metals in the catalyst system simplifies the regulatory compliance landscape, making it easier to obtain environmental permits for scale-up. The straightforward workup procedure, involving simple distillation and filtration, is inherently scalable from pilot plant to commercial tonnage, facilitating rapid technology transfer and reducing the time-to-market for new polymer formulations derived from this monomer.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the production and application of N-acryloyl succinimide, derived directly from the patented methodology. These insights are intended to clarify the operational feasibility and quality benchmarks associated with this specific synthetic route. Understanding these details is essential for technical teams evaluating the integration of this monomer into their existing polymer portfolios.
Q: What is the primary advantage of N-acryloyl succinimide over traditional N-substituted imides?
A: Unlike traditional N-substituted imides which often lack reactive functionality, N-acryloyl succinimide contains a carbon-carbon unsaturated double bond. This allows it to act as a reactive monomer for copolymerization, resulting in polymers with superior heat resistance and acid-alkali stability.
Q: How does the synthesis method prevent polymerization of the acryl group during production?
A: The process explicitly incorporates specific polymerization inhibitors such as hydroquinone, MEHQ, or tert-butyl catechol during the dehydration cyclization step. This ensures the unsaturated double bond remains intact (retention rates >90%) rather than prematurely polymerizing.
Q: What purity levels can be achieved with this synthetic route?
A: Through optimized recrystallization steps using solvents like deionized water or alcohols, the method consistently achieves purity levels exceeding 98%, with specific embodiments reaching up to 99.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acryloyl Succinimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of N-acryloyl succinimide in the development of advanced engineering plastics and coatings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial application is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for high-performance polymerization. Our commitment to quality assurance means that you receive a product with consistent double bond retention and minimal impurity profiles, essential for reproducible polymer properties.
We invite you to collaborate with us to leverage this innovative chemistry for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the specialty chemicals sector.
