Revolutionizing Hemiaminal Synthesis: Enzymatic Routes for High-Purity Pharmaceutical Intermediates
Revolutionizing Hemiaminal Synthesis: Enzymatic Routes for High-Purity Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more efficient synthetic methodologies, particularly for unstable structural motifs like hemiaminals. Patent CN101659971A introduces a groundbreaking approach for the enzymatic synthesis of nitrogen heterocyclic derivatives possessing hemiaminal structures, addressing critical challenges in stability and selectivity that have long plagued traditional organic synthesis. This technology leverages the specificity of lipases to facilitate a three-component one-pot reaction, effectively bypassing the decomposition pathways typical of hemiaminal intermediates. For R&D directors and procurement strategists, this represents a significant opportunity to access high-purity pharmaceutical intermediates through a route that minimizes environmental impact and operational complexity. By utilizing commercially available substrates and robust biocatalysts, this method offers a scalable solution for producing complex nitrogen-containing scaffolds essential for antifungal and antitumor drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hemiaminal-containing compounds has been fraught with difficulties due to the inherent instability of the hemiaminal functional group, which readily decomposes into starting materials or dehydrates to form imine structures under standard conditions. Conventional synthetic routes often rely on strong Lewis acid-base catalysts or transition metal catalysts, which necessitate rigorous anhydrous conditions and frequently require elevated temperatures that accelerate degradation. Furthermore, these traditional multi-step processes involve the isolation of reactive intermediates, leading to substantial product loss and low overall yields. The reliance on heavy metal catalysts also introduces significant downstream purification burdens, requiring expensive removal steps to meet stringent pharmaceutical purity specifications. Additionally, the use of harsh reagents generates considerable chemical waste, posing environmental compliance challenges and increasing the total cost of ownership for manufacturers seeking sustainable production capabilities.
The Novel Approach
In stark contrast, the novel enzymatic method disclosed in the patent utilizes lipase as a highly selective biocatalyst to drive the condensation of nitrogen heterocycles, aldehydes, and acylating agents in a single pot. This approach operates under remarkably mild conditions, typically between 10~70°C, which preserves the delicate hemiaminal structure while promoting efficient acylation. The one-pot nature of the reaction eliminates the need to isolate unstable intermediates, thereby preventing their decomposition and significantly boosting the final yield of the target nitrogen heterocyclic derivative. By employing immobilized lipases such as Candida antarctica lipase B (CAL-B), the process ensures catalyst reusability and simplifies product separation. This methodology not only streamlines the synthetic workflow but also aligns with green chemistry principles by reducing solvent usage and eliminating toxic metal residues, making it an ideal candidate for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Lipase-Catalyzed Hemiaminal Formation
The core of this technological advancement lies in the unique ability of lipases to function effectively in non-aqueous media, creating a microenvironment that stabilizes the transition state of the hemiaminal formation. The reaction mechanism involves the nucleophilic attack of the nitrogen heterocycle on the aldehyde to form the transient hemiaminal, which is immediately trapped by the acylating agent in the presence of the enzyme. This in-situ acylation is crucial, as it converts the labile hydroxyl group of the hemiaminal into a stable ester, locking the structure and preventing retro-aldol type decomposition. The enzyme acts not merely as a catalyst for acylation but also facilitates the initial condensation step through precise orientation of the substrates within its active site. This dual functionality allows for high regioselectivity and chemoselectivity, ensuring that side reactions such as polymerization or over-oxidation are minimized. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters for specific substrate combinations.
Substrate compatibility is a key factor in the success of this enzymatic route, with the patent specifying a broad scope for the nitrogen heterocyclic component. As illustrated in the structural framework below, the nitrogen heterocycle typically features an imidazole core with variable substituents that can influence electronic density and steric hindrance. 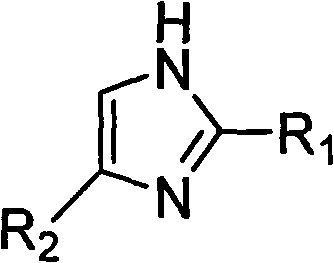 . The versatility of the system allows for R1 to be hydrogen or methyl, and R2 to be hydrogen, methyl, or nitro groups, enabling the synthesis of a diverse library of derivatives. Similarly, the aldehyde component, depicted in the following structure, accommodates a wide range of alkyl and aryl groups, from simple methyl chains to complex aromatic systems like pyridyl or nitrophenyl groups.
. The versatility of the system allows for R1 to be hydrogen or methyl, and R2 to be hydrogen, methyl, or nitro groups, enabling the synthesis of a diverse library of derivatives. Similarly, the aldehyde component, depicted in the following structure, accommodates a wide range of alkyl and aryl groups, from simple methyl chains to complex aromatic systems like pyridyl or nitrophenyl groups. 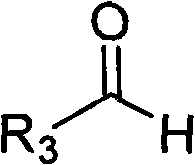 . This structural flexibility ensures that the process can be adapted for the commercial scale-up of complex pharmaceutical additives and active ingredients without requiring extensive re-engineering of the catalytic system.
. This structural flexibility ensures that the process can be adapted for the commercial scale-up of complex pharmaceutical additives and active ingredients without requiring extensive re-engineering of the catalytic system.
How to Synthesize Hemiaminal Nitrogen Heterocycles Efficiently
Implementing this enzymatic synthesis requires careful attention to solvent selection and enzyme loading to maximize catalytic efficiency. The process begins with the preparation of an anhydrous reaction medium, as water can compete with the nucleophile and hydrolyze the acylating agent, reducing yield. Operators must select solvents with a LogP value less than 6 to balance enzyme hydration needs with substrate solubility, with options ranging from isopropyl ether to tetrahydrofuran. The detailed standardized synthesis steps see the guide below outline the precise molar ratios and temperature profiles required to achieve optimal conversion rates while maintaining enzyme stability over extended reaction times.
- Prepare the reaction system by mixing the nitrogen heterocyclic compound, aldehyde, acylating reagent, and anhydrous organic solvent.
- Add immobilized lipase (such as CAL-B) to the mixture to achieve a concentration of 100-200 IU/ml.
- Maintain the reaction at 10-70°C for 4-72 hours, then purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic protocol offers tangible strategic advantages beyond mere technical feasibility. The elimination of transition metal catalysts removes the need for costly scavenging resins and extensive purification workflows, directly translating to reduced processing time and lower operational expenditures. Furthermore, the mild reaction conditions significantly decrease energy consumption compared to high-temperature thermal processes, contributing to a smaller carbon footprint and lower utility costs. The use of commercially available, off-the-shelf raw materials ensures a robust supply chain with minimal risk of bottlenecking, while the high selectivity of the enzyme reduces the formation of difficult-to-remove impurities. These factors collectively enhance the reliability of the supply chain for high-purity intermediates, ensuring consistent quality and delivery performance for downstream pharmaceutical applications.
- Cost Reduction in Manufacturing: The enzymatic route drastically simplifies the production process by combining multiple synthetic steps into a single operation, thereby reducing labor costs and equipment utilization time. By avoiding the use of expensive and toxic metal catalysts, manufacturers eliminate the regulatory and financial burdens associated with heavy metal testing and removal, leading to substantial cost savings in quality control and waste disposal. The high atom economy of the one-pot reaction ensures that raw materials are converted efficiently into the desired product, minimizing waste generation and maximizing the value derived from each batch of inputs.
- Enhanced Supply Chain Reliability: The reliance on stable, immobilized enzymes and common organic solvents mitigates the risk of supply disruptions often associated with specialized reagents or custom catalysts. Since the substrates such as imidazoles and aldehydes are commodity chemicals with established global supply networks, procurement teams can secure long-term contracts with favorable pricing structures. The robustness of the biocatalyst allows for consistent batch-to-batch reproducibility, reducing the likelihood of failed runs and ensuring a steady flow of material to meet production schedules without unexpected delays.
- Scalability and Environmental Compliance: This process is inherently scalable, moving seamlessly from laboratory benchtop experiments to industrial-scale reactors without significant changes in reaction dynamics or yield profiles. The absence of hazardous reagents and the use of biodegradable enzymes align with increasingly strict environmental regulations, facilitating easier permitting and reducing the risk of compliance violations. The simplified workup procedure, often requiring only filtration to remove the immobilized enzyme followed by standard chromatography, reduces the volume of solvent waste, supporting corporate sustainability goals and reducing disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this enzymatic synthesis route. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering practical guidance for process development teams. Understanding these nuances is essential for successfully transferring this technology from the lab to commercial production environments.
Q: Why is enzymatic catalysis preferred over traditional metal catalysts for hemiaminal synthesis?
A: Traditional methods often require harsh Lewis acids or high temperatures which degrade unstable hemiaminal structures. Enzymatic catalysis operates under mild conditions (10-70°C), preserving the integrity of the sensitive hemiaminal unit while avoiding toxic metal residues.
Q: What represents the optimal solvent system for this lipase-catalyzed reaction?
A: Anhydrous organic solvents with a LogP value less than 6 are preferred. Solvents like isopropyl ether, tetrahydrofuran, or mixed systems balance enzyme activity maintenance with sufficient substrate solubility, preventing enzyme deactivation.
Q: How does this one-pot method improve yield compared to multi-step synthesis?
A: By performing the reaction in a one-pot system, the unstable hemiaminal intermediate is generated and immediately stabilized via acylation without isolation. This prevents decomposition into imines or starting materials, significantly enhancing overall process efficiency and yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hemiaminal Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of enzymatic catalysis in modern pharmaceutical manufacturing and are committed to delivering these advanced solutions to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of hemiaminal derivatives meets the highest industry standards for potency and impurity profiles. Our infrastructure is designed to handle complex biocatalytic processes with precision, providing a secure and reliable source for your critical intermediate needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our enzymatic synthesis capabilities can optimize your supply chain and reduce your overall cost of goods sold.
