Advanced Pd-Catalyzed Synthesis of Alpha-Amino Ketone Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex structural motifs, particularly alpha-amino ketone derivatives which serve as pivotal intermediates in the synthesis of various heterocycles and bioactive molecules. Patent CN109096138B introduces a groundbreaking methodology that leverages palladium catalysis to drive a tandem nucleophilic addition and ring-opening rearrangement reaction between 2H-aziridines and carboxylic acids. This innovation represents a significant leap forward in synthetic organic chemistry, addressing long-standing challenges regarding substrate availability and reaction conditions. By enabling the direct construction of alpha-amino ketone scaffolds in a single step, this technology offers a streamlined alternative to multi-step sequences, thereby enhancing overall process efficiency. The robustness of this catalytic system, which tolerates ambient conditions without the need for rigorous exclusion of moisture or oxygen, positions it as a highly attractive candidate for industrial adoption. For procurement and supply chain leaders, understanding the implications of such a versatile synthetic tool is crucial for securing reliable sources of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-amino ketone derivatives has been plagued by significant synthetic hurdles that impede scalable manufacturing and cost-effective production. Traditional approaches often rely on the use of protected aziridines, such as those bearing p-toluenesulfonyl groups, which necessitate additional synthetic steps for both installation and subsequent removal of the protecting group. As illustrated in prior art methodologies, these protected substrates frequently suffer from poor regioselectivity during ring-opening events, leading to complex mixtures of isomers that are difficult and expensive to separate. Furthermore, alternative strategies involving methylene azetidines have demonstrated extremely narrow substrate scopes and require the use of sensitive phosphine ligands that mandate strict anaerobic conditions. These constraints not only increase the capital expenditure for specialized reactor equipment but also introduce substantial safety risks and operational delays associated with handling air-sensitive reagents. Consequently, the existing landscape of synthetic methods fails to meet the demands of modern process chemistry, where simplicity, safety, and atom economy are paramount.
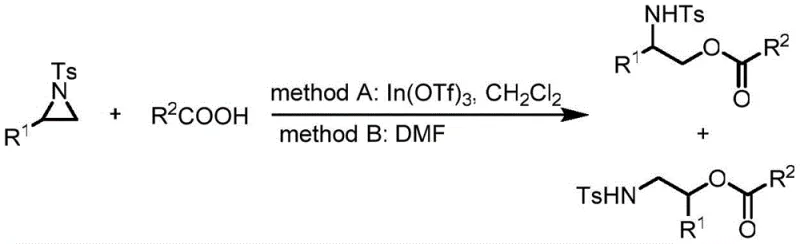
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN109096138B utilizes unprotected 2H-aziridines as highly reactive synthons that undergo direct transformation under mild palladium catalysis. This methodology eliminates the need for protecting group manipulation entirely, thereby reducing the total number of synthetic steps and improving the overall atom economy of the process. The reaction proceeds through a unique mechanism where the carboxylic acid acts as a nucleophile, attacking the imine bond of the 2H-aziridine followed by a spontaneous ring-opening rearrangement to yield the desired alpha-amino ketone structure. Crucially, this transformation is insensitive to air and moisture, allowing reactions to be conducted in standard laboratory glassware without the need for gloveboxes or Schlenk lines. The broad substrate compatibility extends to various substituted aryl and heteroaryl carboxylic acids, enabling the rapid generation of diverse chemical libraries. This paradigm shift from protected, sensitive substrates to robust, unprotected synthons fundamentally alters the economic and logistical feasibility of producing these valuable intermediates on a commercial scale.
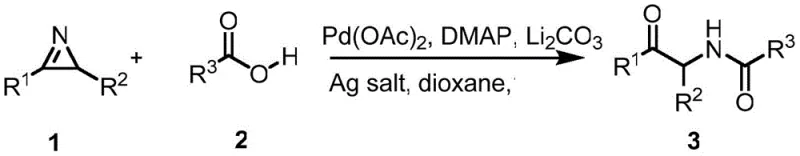
Mechanistic Insights into Pd-Catalyzed Nucleophilic Addition and Ring-Opening
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst and the unique electronic properties of the 2H-aziridine ring. The catalytic cycle initiates with the activation of the carboxylic acid species, likely facilitated by the base and the silver salt additive, which enhances the nucleophilicity of the carboxylate oxygen. This activated nucleophile then attacks the electrophilic imine carbon within the strained three-membered ring of the 2H-aziridine. The inherent ring strain of the aziridine, combined with the electron-withdrawing nature of the imine nitrogen, lowers the activation energy for this addition step, driving the reaction forward efficiently. Following the initial nucleophilic attack, the intermediate undergoes a critical intramolecular rearrangement where the carbon-nitrogen bond cleaves, resulting in the formation of the alpha-amino ketone backbone. The presence of the palladium species is essential not only for facilitating the initial activation but also for stabilizing the transition states throughout this tandem sequence. This mechanistic pathway avoids the formation of unstable carbene or nitrile ylide intermediates that are common in other aziridine transformations, thereby ensuring high chemical selectivity and minimizing the generation of unwanted byproducts.
From an impurity control perspective, the high regioselectivity of this reaction is a major advantage for R&D teams focused on process purity. Unlike traditional acid-catalyzed ring openings which can occur at either carbon of the aziridine ring leading to regioisomeric mixtures, this palladium-mediated process directs the nucleophilic attack specifically to the imine carbon. This specificity ensures that the resulting alpha-amino ketone derivative is formed with high structural fidelity, significantly reducing the burden on downstream purification processes. The tolerance for various functional groups, including halogens, nitro groups, and trifluoromethyl substituents, further demonstrates the robustness of the catalytic system against potential side reactions such as oxidative addition into aryl halides or decomposition of sensitive moieties. By maintaining a clean reaction profile with minimal byproduct formation, this method supports the production of high-purity intermediates that meet the stringent quality standards required for pharmaceutical applications. The ability to achieve yields ranging from 19% to 92% depending on the substrate electronic properties provides a predictable framework for optimizing reaction conditions for specific target molecules.
How to Synthesize Alpha-Amino Ketone Derivatives Efficiently
Implementing this synthesis in a practical setting involves a straightforward protocol that balances reagent stoichiometry with thermal energy to drive the tandem transformation to completion. The process begins by combining the 2H-aziridine substrate with the chosen carboxylic acid in a reaction vessel containing a polar aprotic solvent such as 1,4-dioxane. To this mixture, a catalytic amount of palladium acetate is added along with a specialized silver salt additive and DMAP as a nucleophilic catalyst promoter, while lithium carbonate serves as the base to neutralize the acid and generate the reactive carboxylate species. The detailed standardized synthesis steps see the guide below.
- Combine 2H-aziridine, carboxylic acid, Pd(OAc)2 catalyst, silver salt additive, DMAP, and Li2CO3 base in a suitable solvent like dioxane.
- Heat the reaction mixture to a temperature range of 80-110°C and maintain stirring for 12 to 18 hours to facilitate nucleophilic addition and ring-opening.
- Cool the reaction to room temperature, remove the solvent under reduced pressure, and purify the crude product via column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The elimination of protecting group steps translates directly into a reduction in raw material consumption and waste generation, which are key drivers of manufacturing costs in the fine chemical sector. By removing the need for tosyl chloride or similar protecting agents, companies can simplify their supply chains and reduce dependency on multiple vendors for specialized reagents. Furthermore, the operational simplicity of running reactions under ambient atmospheric conditions removes the requirement for expensive inert gas infrastructure and the associated maintenance costs, leading to substantial overhead savings. The robustness of the reaction conditions also implies a lower risk of batch failure due to environmental factors, thereby enhancing supply continuity and reliability for critical intermediate production. These factors collectively contribute to a more resilient and cost-effective manufacturing ecosystem.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis significantly lowers the cost of goods sold by consolidating multiple synthetic operations into a single unit operation. By avoiding the isolation and purification of protected intermediates, manufacturers save on solvent usage, energy consumption for heating and cooling cycles, and labor hours associated with extended processing times. The high atom economy ensures that a larger proportion of the starting mass ends up in the final product, minimizing waste disposal fees and maximizing resource utilization. Additionally, the use of commercially available and relatively inexpensive catalysts like palladium acetate, which can potentially be recovered or used in low loadings, further optimizes the cost structure. This economic efficiency makes the production of complex alpha-amino ketone derivatives viable even for price-sensitive markets.
- Enhanced Supply Chain Reliability: The insensitivity of this reaction to air and moisture drastically reduces the logistical complexities associated with transporting and storing air-sensitive reagents. Suppliers can utilize standard packaging and shipping methods rather than specialized containers required for pyrophoric or moisture-sensitive materials, leading to faster lead times and lower freight costs. The broad substrate scope allows for flexibility in sourcing raw materials; if a specific substituted benzoic acid is unavailable, alternative analogs can often be substituted without re-optimizing the entire process. This flexibility mitigates the risk of supply disruptions caused by raw material shortages. Moreover, the mild reaction conditions reduce wear and tear on reactor vessels, extending equipment lifespan and ensuring consistent production capacity over time.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne levels is facilitated by the absence of hazardous exotherms typically associated with highly reactive organometallic reagents. The use of dioxane as a solvent, while requiring proper handling, is well-established in industrial settings with existing recovery and recycling protocols. The high selectivity of the reaction minimizes the formation of toxic byproducts, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations. The ability to operate at moderate temperatures (80-110°C) reduces the energy footprint of the manufacturing process compared to high-temperature alternatives. These environmental and safety advantages align with the sustainability goals of modern chemical enterprises, making this technology a future-proof choice for long-term production planning.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this palladium-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production workflows. The responses cover aspects ranging from reaction conditions to substrate limitations, offering a comprehensive overview of the method's capabilities.
Q: What are the key advantages of using 2H-aziridines over traditional aziridines in this synthesis?
A: Unlike traditional aziridines that often require cumbersome protecting groups like tosyl which limit regioselectivity, 2H-aziridines possess inherent ring strain and an imine group that allow for direct nucleophilic addition and ring-opening rearrangement without protection, simplifying the synthetic route significantly.
Q: Does this Pd-catalyzed method require strict anaerobic conditions?
A: No, one of the major breakthroughs of this technology is its insensitivity to water and air. Unlike previous methods utilizing phosphine ligands that demanded strict inert gas protection, this protocol operates efficiently under ambient atmospheric conditions, greatly reducing operational complexity and equipment costs.
Q: What is the substrate scope for the carboxylic acid component in this reaction?
A: The method demonstrates excellent functional group tolerance, accommodating a wide range of carboxylic acids including those with substituted aryl groups, heteroaryl moieties like thiophene, and even aliphatic chains, allowing for the diverse generation of alpha-amino ketone libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino Ketone Derivatives Supplier
As the demand for high-quality pharmaceutical intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-amino ketone derivatives meets the highest industry standards. Our commitment to technical excellence means we can navigate the complexities of palladium-catalyzed reactions with precision, delivering products that support your drug development timelines effectively.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic benefits of switching to this more efficient methodology. We encourage you to contact us to obtain specific COA data for relevant intermediates and to receive detailed route feasibility assessments. Let us help you optimize your supply chain and accelerate your path to market with our reliable Alpha-Amino Ketone Derivatives supply solutions.
