Scalable Enzymatic Resolution of Chiral Pyrazole Intermediates for Analgesic Manufacturing
Scalable Enzymatic Resolution of Chiral Pyrazole Intermediates for Analgesic Manufacturing
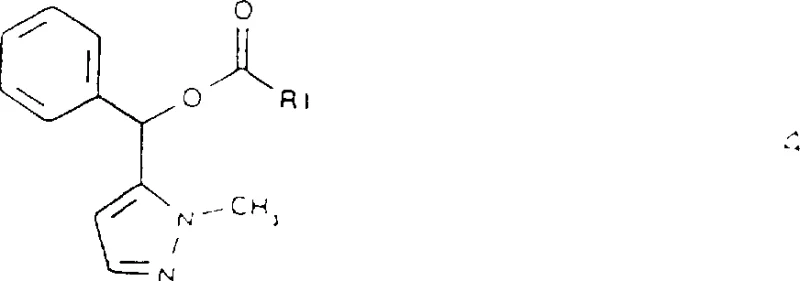
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant advancements in biocatalytic processes, as exemplified by the groundbreaking technology disclosed in patent CN1177958A. This patent outlines a sophisticated method for the resolution of racemic 5-(phenyl)hydroxymethyl-1-methyl-1H-pyrazole, a critical precursor in the synthesis of potent analgesic compounds. By leveraging the stereoselective power of lipases, this process overcomes the inherent limitations of traditional chemical resolution, offering a pathway to obtain the biologically active (R)-(+)-enantiomer with exceptional optical purity. For R&D directors and process chemists, understanding the nuances of this enzymatic transformation is vital for developing robust, scalable manufacturing routes that meet stringent regulatory standards for chiral drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of enantiomers for pyrazole-based pharmaceutical intermediates relied heavily on classical resolution techniques, such as fractional crystallization of diastereomeric salts or complex column chromatography. These conventional methods are often plagued by significant inefficiencies, including low theoretical yields capped at 50% for a single resolution cycle and the requirement for stoichiometric amounts of expensive chiral resolving agents. Furthermore, the purification steps involved in removing these resolving agents can be tedious and environmentally burdensome, generating substantial chemical waste. The reliance on harsh chemical conditions for derivatization and subsequent de-protection also poses risks to the structural integrity of sensitive heterocyclic cores, potentially leading to impurity profiles that are difficult to control during scale-up.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a dynamic kinetic resolution strategy driven by biocatalysis. By employing a lipase, specifically Lipase PS, the process selectively catalyzes the transesterification of one enantiomer of the racemic alcohol while leaving the other untouched. This kinetic differentiation allows for the efficient separation of the desired unreacted alcohol from the esterified byproduct. The use of vinyl esters as acylating agents drives the reaction to completion through the irreversible tautomerization of the leaving group, ensuring high conversion rates. This method not only simplifies the downstream processing but also enables the recycling of the unwanted enantiomer through hydrolysis, effectively doubling the potential yield from a single batch of racemic starting material.
Mechanistic Insights into Lipase-Catalyzed Transesterification
The core of this technology lies in the precise mechanistic action of hydrolases, particularly lipases, within an organic medium. Unlike aqueous environments where lipases typically catalyze hydrolysis, in low-water organic solvents, these enzymes facilitate transesterification. The enzyme's active site possesses a chiral pocket that sterically accommodates only one specific enantiomer of the racemic 5-(phenyl)hydroxymethyl-1-methyl-1H-pyrazole. Once bound, the catalytic triad within the enzyme promotes the nucleophilic attack of the alcohol hydroxyl group on the carbonyl carbon of the vinyl ester. This results in the formation of an acyl-enzyme intermediate and the release of vinyl alcohol, which rapidly tautomerizes to acetaldehyde, rendering the reaction irreversible. This thermodynamic sink is crucial for driving the equilibrium towards the ester product, ensuring that the reaction proceeds efficiently even with limited enzyme loading.
Controlling the impurity profile in such biocatalytic processes is inherently superior to chemical methods due to the enzyme's high substrate specificity. The lipase discriminates not only between enantiomers but also rejects structurally similar impurities that might be present in the crude racemate, acting as a built-in purification step. Furthermore, the reaction conditions—typically ranging from 20°C to 60°C—are mild enough to prevent thermal degradation of the pyrazole ring or racemization of the chiral center. The addition of molecular sieves to the reaction medium plays a critical role in scavenging trace water, which could otherwise compete with the alcohol for the acyl-enzyme intermediate, leading to hydrolysis of the ester product and reduced optical purity. This careful management of water activity ensures that the enantiomeric excess (ee) remains consistently above 94%, meeting the rigorous demands of modern pharmaceutical synthesis.
How to Synthesize 5-(phenyl)hydroxymethyl-1-methyl-1H-pyrazole Efficiently
Implementing this enzymatic resolution requires a systematic approach to reaction optimization and workup procedures to maximize both yield and optical purity. The process begins with the suspension of the racemic alcohol and the lipase catalyst in a suitable organic solvent or under solvent-free conditions, followed by the slow addition of the vinyl ester acylating agent. Monitoring the reaction progress via proton NMR or chiral HPLC is essential to determine the optimal endpoint, typically around 50-55% conversion, to ensure the highest enantiomeric purity of the remaining alcohol. Detailed standardized synthetic steps for this specific transformation are provided in the guide below.
- Perform enzymatic transesterification of the racemic alcohol using Lipase PS and vinyl acetate in an organic solvent or solvent-free condition at 20-60°C.
- Separate the unreacted enantiomer from the esterified enantiomer using silica gel chromatography or crystallization techniques.
- Hydrolyze the esterified enantiomer under acidic or basic conditions to recover the opposite enantiomer or racemate for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chemical to enzymatic resolution represents a strategic opportunity to optimize cost structures and enhance supply reliability. The elimination of stoichiometric chiral resolving agents significantly reduces the raw material costs associated with each production batch. Moreover, the ability to recycle the unwanted enantiomer back into the process loop drastically improves the overall atom economy, reducing the volume of raw materials required per kilogram of final product. This efficiency translates directly into a more competitive pricing model for the final API intermediate, providing a distinct market advantage in cost-sensitive therapeutic areas.
- Cost Reduction in Manufacturing: The implementation of this biocatalytic route eliminates the need for expensive heavy metal catalysts or complex chiral ligands often required in asymmetric synthesis. By utilizing commercially available lipases that can be recovered and reused, the operational expenditure related to catalyst consumption is substantially lowered. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further contributing to a leaner manufacturing cost profile without compromising on the quality or purity of the intermediate.
- Enhanced Supply Chain Reliability: Reliance on enzymatic processes mitigates the risks associated with the supply volatility of specialized chemical reagents. Lipases are robust, commercially available biocatalysts with stable supply chains, ensuring consistent production schedules. The simplified workup procedure, involving basic filtration and crystallization rather than complex chromatographic separations, shortens the production cycle time. This agility allows for faster response to market demand fluctuations and reduces the lead time for delivering high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling biocatalytic reactions is generally more straightforward than scaling complex asymmetric chemical syntheses, as the reaction kinetics remain predictable from gram to ton scale. The process generates significantly less hazardous waste compared to traditional methods, as it avoids the use of toxic solvents and heavy metals. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with waste disposal and environmental compliance, making it a sustainable choice for long-term commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic resolution of pyrazole carbinols. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for potential partners evaluating this technology for their supply chain.
Q: What is the primary advantage of using lipase for pyrazole resolution?
A: Lipase-catalyzed resolution offers high enantioselectivity under mild conditions, avoiding the harsh reagents and complex purification steps associated with traditional chemical resolution methods.
Q: Can the unwanted enantiomer be recycled in this process?
A: Yes, the esterified enantiomer can be hydrolyzed to regenerate the racemic alcohol, which can then be re-subjected to the enzymatic resolution, significantly improving overall atom economy.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The use of immobilized or free lipases in organic solvents allows for easy catalyst recovery and reuse, making the process highly scalable and cost-effective for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(phenyl)hydroxymethyl-1-methyl-1H-pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust chiral synthesis platforms in the development of next-generation analgesics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering intermediates with stringent purity specifications, supported by our rigorous QC labs that utilize advanced chiral HPLC and NMR spectroscopy to verify enantiomeric excess and chemical identity at every stage of production.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your drug development timeline while optimizing your overall production costs.
