Advanced Enzymatic Synthesis of Cyclohexenone Derivatives for Commercial Scale-Up
Advanced Enzymatic Synthesis of Cyclohexenone Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking sustainable, efficient pathways to construct complex molecular scaffolds, particularly six-membered ring systems which serve as critical backbones for numerous bioactive compounds. A significant technological breakthrough in this domain is documented in Chinese Patent CN102162002B, which discloses a novel method for the acylase-catalyzed multi-step tandem synthesis of cyclohexenone derivatives. This innovation represents a paradigm shift from traditional stoichiometric chemical synthesis to a more elegant, biocatalytic approach that leverages the multifunctionality of enzymes to drive complex cascade reactions. By utilizing a combination of acylase and specific organic additives, this method enables the direct conversion of simple aldehydes and ketones into highly functionalized cyclohexenone structures under remarkably mild conditions. For R&D directors and procurement specialists alike, this technology offers a compelling value proposition by merging the precision of enzymatic catalysis with the operational simplicity required for cost reduction in pharmaceutical intermediate manufacturing.
The core of this invention lies in its ability to orchestrate a sequence of bond-forming events—specifically aldol condensation, elimination, Michael addition, and intramolecular aldol reactions—within a single reaction vessel. Unlike conventional methods that often require distinct steps, different catalysts, and rigorous isolation of unstable intermediates, this enzymatic tandem process streamlines the entire synthetic trajectory. The patent specifies that the reaction can be conducted at temperatures ranging from 10°C to 70°C over a period of 4 to 72 hours, utilizing solvents that range from aqueous systems to organic media like THF or dioxane. This flexibility not only broadens the substrate scope but also aligns perfectly with green chemistry principles, addressing the growing regulatory pressure to minimize hazardous waste and energy consumption in chemical production facilities globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of cyclohexenone rings has relied heavily on transition metal catalysis or strong base-mediated aldol condensations, which present substantial challenges for large-scale operations. Traditional chemical routes often necessitate the use of expensive and toxic heavy metal catalysts, which subsequently require rigorous and costly purification steps to ensure the final product meets stringent pharmaceutical purity specifications regarding residual metals. Furthermore, stepwise synthetic approaches typically involve the isolation of reactive intermediates, which can be thermally unstable or prone to polymerization, leading to significant yield losses and safety hazards during handling. The accumulation of waste solvents and the need for extreme reaction conditions, such as high temperatures or cryogenic cooling, further exacerbate the environmental footprint and operational expenditures associated with these legacy processes, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the acylase-catalyzed method described in the patent introduces a biomimetic strategy that circumvents these industrial pain points by employing a single enzyme system capable of promoting multiple reaction types sequentially. This one-pot methodology eliminates the need for intermediate isolation, thereby drastically reducing solvent usage and processing time while enhancing the overall atom economy of the transformation. The use of acylase, an enzyme traditionally known for hydrolysis, in a non-natural carbon-carbon bond-forming capacity highlights the versatility of biocatalysts when paired with appropriate co-catalysts. This approach not only simplifies the operational workflow for plant managers but also ensures a cleaner impurity profile, as the high specificity of the enzyme minimizes the formation of side products that are common in non-selective chemical catalysis, ultimately facilitating easier downstream processing and purification.
Mechanistic Insights into Acylase-Catalyzed Tandem Cyclization
The mechanistic elegance of this process stems from the synergistic interaction between the acylase enzyme and small molecule additives, which together create a catalytic environment conducive to complex cascade reactions. The patent elucidates that the transformation proceeds through a sophisticated sequence beginning with an initial aldol reaction between the aldehyde and the ketone, followed by dehydration to form an alpha,beta-unsaturated intermediate. This intermediate then undergoes a Michael addition with another equivalent of the ketone enolate, setting the stage for an intramolecular aldol cyclization that closes the six-membered ring. The final elimination step yields the conjugated cyclohexenone system. The enzyme likely facilitates these steps by stabilizing transition states through hydrogen bonding networks within its active site, while simultaneously activating the nucleophilic species, effectively lowering the activation energy barriers for each consecutive step in the cascade without the need for external thermal input.
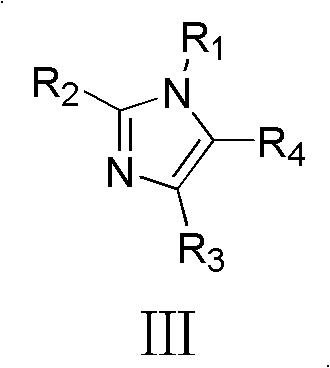
A critical component of this catalytic system is the inclusion of specific additives, such as imidazole derivatives or amino acids, which act as co-catalysts to modulate the reaction kinetics and selectivity. As illustrated in the structural formulas provided in the patent, additives like N-methylimidazole or nitro-substituted imidazoles play a pivotal role in proton transfer processes, potentially acting as general bases to facilitate enolization or stabilizing charged intermediates during the Michael addition phase. The presence of these additives allows the acylase to function efficiently in organic solvent mixtures, expanding the solubility of hydrophobic substrates that would otherwise be inaccessible in purely aqueous enzymatic reactions. This dual-catalyst system ensures that the reaction proceeds with high fidelity, minimizing the formation of oligomeric byproducts and ensuring that the desired cyclohexenone scaffold is generated with high regioselectivity, a crucial factor for maintaining the quality standards required for reliable pharmaceutical intermediate suppliers.
How to Synthesize 3-Methyl-5-phenyl-2-cyclohexen-1-one Efficiently
Implementing this synthesis route requires careful optimization of reaction parameters to maximize yield and purity, leveraging the specific conditions outlined in the patent examples. The process begins with the preparation of a homogeneous reaction mixture containing the aldehyde substrate, such as benzaldehyde or its substituted derivatives, and acetone as the ketone source in a selected solvent system. The choice of solvent is flexible, encompassing polar aprotic solvents like DMF or DMSO, as well as greener options like water or ethanol, depending on the solubility profile of the specific starting materials. Once the substrate solution is prepared, the biocatalytic system is introduced by adding the acylase enzyme along with the requisite amount of additive, initiating the tandem cascade that constructs the cyclic framework in a single operational step.
- Prepare a reaction system by mixing a specific aldehyde (such as substituted benzaldehyde) and a ketone (preferably acetone) in a suitable solvent like water, THF, or dioxane.
- Introduce the biocatalyst system by adding D-amino acid acylase (50mu-2000mu per liter) and a specific additive such as imidazole, substituted imidazole, or an amino acid (4g-128g per liter).
- Maintain the reaction mixture at a mild temperature between 10°C and 70°C for a duration of 4 to 72 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced capital expenditure and lower operating costs. By consolidating multiple synthetic steps into a single pot, manufacturers can significantly reduce the consumption of solvents and reagents, while also minimizing the labor and equipment time associated with intermediate workups and isolations. This streamlined workflow enhances the overall throughput of the production facility, allowing for faster turnaround times and improved responsiveness to market demand fluctuations for critical pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive and specialized metal scavenging resins or complex filtration protocols, which are often major cost drivers in traditional synthesis. Furthermore, the mild reaction temperatures (ranging from ambient to moderately elevated) result in substantial energy savings compared to processes requiring reflux or cryogenic conditions. The high selectivity of the enzymatic route also implies a reduction in waste disposal costs, as fewer byproducts are generated, and the use of biodegradable enzymes aligns with increasingly strict environmental regulations, avoiding potential fines and remediation expenses associated with hazardous chemical waste.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically acetone and various substituted benzaldehydes, are commodity chemicals with robust and stable global supply chains, mitigating the risk of raw material shortages that can plague specialty reagent-dependent processes. The versatility of the solvent system allows manufacturers to adapt to local availability and pricing of solvents, further insulating the supply chain from regional disruptions. Additionally, the stability of the enzyme-additive system under the specified reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term supply contracts with multinational pharmaceutical clients who demand unwavering quality and continuity.
- Scalability and Environmental Compliance: The one-pot nature of this reaction is inherently scalable, as it avoids the engineering complexities associated with multi-step flow chemistry or the handling of unstable intermediates on a large scale. The process generates water as a primary byproduct alongside the organic product, significantly reducing the load on wastewater treatment facilities compared to processes generating heavy metal sludge or halogenated waste. This environmental compatibility not only facilitates easier permitting for new production lines but also enhances the corporate sustainability profile of the manufacturer, a metric that is becoming increasingly important in vendor selection criteria for major global pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acylase-catalyzed technology, derived from the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the operational parameters and scope of the method for potential partners evaluating its integration into their existing manufacturing portfolios. Understanding these nuances is essential for assessing the feasibility of scaling this route for specific target molecules within the cyclohexenone class.
Q: What are the key advantages of using acylase for cyclohexenone synthesis compared to traditional chemical catalysts?
A: The acylase-catalyzed method operates under significantly milder conditions (10°C-70°C) compared to harsh chemical methods, eliminates the need for toxic heavy metal catalysts, and simplifies the workflow through a one-pot tandem reaction strategy that reduces waste generation.
Q: Which additives are compatible with the acylase catalyst in this tandem reaction?
A: The process utilizes specific additives to enhance catalytic efficiency, including imidazole, substituted imidazoles (such as N-methylimidazole or nitroimidazoles), benzimidazole, or various amino acids like L-proline and L-lysine.
Q: Is this enzymatic process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method demonstrates high scalability potential due to the use of commercially available raw materials, robust reaction conditions that tolerate various solvents, and the elimination of complex intermediate isolation steps, making it suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexenone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic methods like the one described in CN102162002B for producing high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications and deliver consistent quality for complex cyclic ketones and other fine chemical intermediates required by the global healthcare industry.
We invite you to collaborate with our technical team to explore how this enzymatic route can optimize your specific supply chain needs. By engaging with us, you gain access to a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this technology can reduce your total cost of ownership. We encourage you to contact our technical procurement team today to request specific COA data for similar structures and comprehensive route feasibility assessments, ensuring your project moves forward with the highest level of technical confidence and commercial viability.
