Advanced Stereoselective Synthesis of Oxooxazoline Derivatives for Commercial Scale-Up
Advanced Stereoselective Synthesis of Oxooxazoline Derivatives for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust, scalable, and economically viable pathways for synthesizing complex heterocyclic intermediates. Patent CN1138766C presents a breakthrough methodology for the preparation of oxooxazoline and alloamino acid derivatives, which serve as critical building blocks for Thyrotropin-Releasing Hormone (TRH) analogs and other bioactive peptides. This technology addresses long-standing challenges in stereocontrol and reagent toxicity, offering a streamlined route that transforms inexpensive, naturally occurring amino acids into high-value chiral scaffolds. By leveraging a unique thionyl chloride-mediated cyclization, this process achieves high stereoselectivity without relying on prohibitively expensive starting materials or hazardous phosgene chemistry.
For R&D directors and process chemists, the significance of this patent lies in its ability to access the 'allo' configuration of amino acids through a controlled inversion mechanism. Traditionally, accessing these specific stereoisomers required sourcing rare natural enantiomers or employing complex resolution techniques. The disclosed method utilizes common L-threonine or D-threonine as the chiral pool source, effectively flipping the stereochemistry during the ring-closing step. This not only simplifies the supply chain by relying on commodity chemicals but also enhances the overall atom economy of the synthesis. The resulting oxooxazoline derivatives are versatile tools for combinatorial chemistry and peptide synthesis, enabling the rapid development of new therapeutic candidates with precise spatial arrangements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing oxooxazoline derivatives were fraught with significant industrial and safety drawbacks. One established approach involved the use of phosgene gas to effect cyclization while maintaining the stereoconfiguration of the starting material. While chemically effective, phosgene is an extremely toxic and regulated substance, necessitating specialized containment equipment and rigorous safety protocols that drastically inflate capital expenditure and operational costs. Furthermore, this conventional route typically required L-allothreonine as the starting material to achieve the desired cis-configuration in the final product. L-allothreonine is a non-proteinogenic amino acid that is significantly more expensive and less readily available than its natural counterpart, L-threonine, creating a bottleneck for cost-effective manufacturing.
The Novel Approach
The innovative strategy outlined in CN1138766C circumvents these issues by employing thionyl chloride as both a dehydrating agent and a cyclization promoter in a solvent system such as toluene or ethyl acetate. Unlike the phosgene method which retains configuration, this novel approach intentionally induces a stereoinversion at the beta-carbon. This mechanistic twist allows manufacturers to start with the abundant and low-cost L-threonine. Through a carefully controlled reaction sequence involving protection, esterification, and thionyl chloride treatment, the process converts the erythro-configuration of threonine into the threo-configuration of the oxooxazoline ring. This shift eliminates the dependency on toxic gases and expensive rare amino acids, representing a paradigm shift in how these valuable intermediates are produced commercially.
Mechanistic Insights into Thionyl Chloride-Mediated Cyclization
The core of this technology is the stereoselective cyclization step where the hydroxyl group of the protected threonine derivative attacks the activated carbamate or amide functionality. When the intermediate is treated with thionyl chloride, the hydroxyl group is converted into a good leaving group (chlorosulfite ester), facilitating an intramolecular nucleophilic attack by the carbonyl oxygen or nitrogen species depending on the specific protection strategy. Crucially, this substitution proceeds via an SN2-like mechanism at the beta-carbon, resulting in the inversion of configuration. This ensures that the methyl group and the oxooxazoline ring end up in the desired trans-relationship relative to the initial threonine backbone, yielding the allo-derivative with high fidelity.
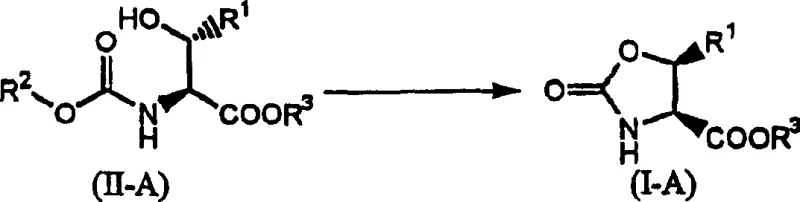
Impurity control is inherently managed by the specificity of the thionyl chloride reaction conditions. The patent data indicates that the ratio of cis-to-trans isomers can be tuned by adjusting the equivalents of thionyl chloride and the solvent volume. Operating with near-stoichiometric amounts of thionyl chloride (close to 1.0 equivalent) in a larger volume of solvent favors the formation of the desired cis-isomer (relative to the oxooxazoline ring substituents), minimizing the generation of unwanted diastereomers. This level of control is vital for pharmaceutical applications where impurity profiles must be strictly defined. The subsequent hydrolysis steps to open the ring and reveal the free alloamino acid are equally robust, utilizing standard acidic or basic conditions that do not racemize the sensitive chiral centers, ensuring the final product meets stringent optical purity requirements.
How to Synthesize Oxooxazoline Derivatives Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the protection of the amino group of L-threonine using a benzyloxycarbonyl (Z) group in an aqueous alkaline environment, followed by esterification of the carboxylic acid to a methyl ester using thionyl chloride in methanol. The critical cyclization is then performed by heating the protected ester in toluene with thionyl chloride. This sequence is designed to maximize yield while maintaining safety. The detailed standardized synthesis steps below outline the precise stoichiometry and thermal conditions required to replicate this high-efficiency pathway.
- Protect the amino group of L-threonine or D-threonine using a benzyloxycarbonyl group (Z-group) in an aqueous alkaline medium.
- Esterify the carboxyl group of the protected intermediate using thionyl chloride in methanol to form the methyl ester.
- Perform the key cyclization step by treating the esterified intermediate with thionyl chloride in toluene at elevated temperatures (60-80°C) to induce stereoinversion and ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this thionyl chloride-based methodology offers profound strategic benefits beyond mere chemical elegance. The primary advantage is the drastic reduction in raw material costs achieved by substituting expensive L-allothreonine with commodity-grade L-threonine. Since L-threonine is produced on a massive fermentation scale globally, its price stability and availability are far superior to specialty amino acids. Additionally, the elimination of phosgene removes the need for complex hazard management infrastructure, lowering the barrier to entry for contract manufacturing organizations (CMOs) and reducing the overall cost of goods sold (COGS) for the final API.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost inputs with low-cost alternatives. By avoiding the use of phosgene, companies save significantly on safety compliance, specialized reactor maintenance, and waste disposal fees associated with highly toxic reagents. The ability to use standard glass-lined or stainless steel reactors without exotic corrosion-resistant linings further reduces capital depreciation costs. Moreover, the high yields reported in the patent examples suggest minimal material loss, directly contributing to a leaner manufacturing budget and improved margin potential for the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Relying on L-threonine as the chiral source insulates the supply chain from the volatility often seen in the market for rare amino acids. L-threonine is a bulk chemical with multiple global suppliers, ensuring continuity of supply even during regional disruptions. The reagents used in the subsequent steps, such as thionyl chloride and toluene, are also standard industrial solvents and reagents with well-established logistics networks. This ubiquity simplifies procurement planning and reduces the risk of production stoppages due to material shortages, allowing for more accurate forecasting and inventory management.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids cryogenic conditions and uses solvents like toluene and ethyl acetate which are easily recovered and recycled via distillation. The waste stream is primarily composed of sulfur dioxide and hydrochloric acid gas, which can be efficiently scrubbed using standard caustic scrubbers, ensuring compliance with modern environmental regulations. This ease of waste treatment contrasts sharply with the heavy metal waste or complex organic byproducts generated by alternative transition-metal catalyzed routes, making the facility easier to permit and operate sustainably over the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxooxazoline synthesis technology. These answers are derived directly from the experimental data and claims within patent CN1138766C, providing a factual basis for evaluating the process feasibility. Understanding these nuances is essential for technical teams assessing the transferability of this method to their own production lines.
Q: Why is the thionyl chloride method superior to conventional phosgene methods?
A: The thionyl chloride method eliminates the need for highly toxic phosgene gas, significantly improving operational safety and reducing environmental compliance costs. Furthermore, it allows for the use of cheaper, naturally abundant L-threonine instead of expensive L-allothreonine by utilizing a stereoinversion mechanism during cyclization.
Q: What is the stereochemical outcome of this cyclization process?
A: The process involves a stereoinversion at the beta-carbon position. Starting from L-threonine (erythro configuration), the reaction with thionyl chloride facilitates an intramolecular nucleophilic substitution that inverts the configuration to yield the allo-threonine derived oxooxazoline (threo configuration) with high stereoselectivity.
Q: Can this process be scaled for industrial API intermediate production?
A: Yes, the patent explicitly describes conditions suitable for mass synthesis, utilizing common solvents like toluene and reagents like thionyl chloride. The avoidance of cryogenic conditions and hazardous gases like phosgene makes the process highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxooxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of oxooxazoline or alloamino acid derivative meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that your downstream synthesis will proceed without unexpected delays caused by impurity-related failures.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall production costs.
