Advanced Synthetic Routes for Neprilysin and ACE Inhibitor Intermediates
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as critical intermediates for cardiovascular therapeutics. Patent CN1170413A discloses a groundbreaking methodology for preparing intermediates of formula I, which are pivotal in the manufacture of enkephalinase and angiotensin-converting enzyme (ACE) inhibitors. This innovation addresses longstanding challenges in constructing the pyrido[2,1-a][2]benzazepine core, offering a versatile platform that accommodates various aromatic substitutions while maintaining rigorous stereochemical integrity. By leveraging novel condensation strategies involving glutaraldehyde or cyano-pyridine derivatives, this technology enables the production of high-purity intermediates essential for next-generation antihypertensive agents. As a leader in fine chemical synthesis, understanding the nuances of this patent provides significant strategic advantages for supply chain optimization and process development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing the intricate pyrido-benzazepine skeleton often suffer from excessive step counts, harsh reaction conditions, and poor control over stereocenters, particularly at the 4-position carboxylic acid moiety. Conventional methodologies frequently rely on late-stage functionalization that introduces impurities difficult to remove, thereby compromising the quality profile required for active pharmaceutical ingredient (API) manufacturing. Furthermore, many legacy processes utilize expensive transition metal catalysts or hazardous reagents that complicate waste management and increase the environmental footprint of production. The lack of modularity in older syntheses also restricts the ability to rapidly generate analog libraries for structure-activity relationship (SAR) studies, slowing down drug discovery timelines. These inefficiencies translate directly into higher costs of goods sold (COGS) and extended lead times for procurement teams seeking reliable sources of complex intermediates.
The Novel Approach
The methodology outlined in CN1170413A revolutionizes this landscape by introducing a streamlined condensation strategy that builds the core heterocycle with remarkable efficiency. As illustrated in the reaction schemes below, the process initiates with the reaction of phthalimido aryl amino acid amides with glutaraldehyde to form 1,4-dihydropyridine derivatives, or alternatively, reacts with 2-cyano-1,2,3,4-tetrahydro-pyridine to yield cyano-substituted precursors. This divergent approach allows manufacturers to select the optimal pathway based on available starting materials and desired throughput. The subsequent cyclization steps utilize strong acid catalysts like trifluoromethanesulfonic acid or sulfuric acid mixtures, which promote rapid ring closure under controlled temperatures ranging from 10°C to 40°C. This precision minimizes side reactions and degradation, ensuring a cleaner crude product profile that simplifies downstream purification efforts significantly.
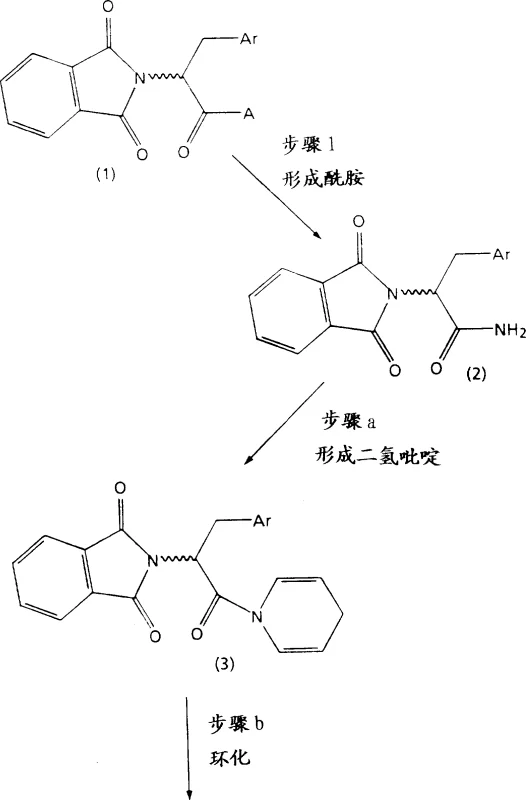
Mechanistically, the success of this novel approach lies in the precise activation of the amino acid derivative and the controlled electrophilic attack on the cyclic amine components. The use of phthalimide as a protecting group is strategic, as it stabilizes the amino functionality during the vigorous cyclization conditions while remaining orthogonal to the subsequent hydrolysis steps. In the alternative Route B, the incorporation of a cyano group serves as a masked carboxylic acid equivalent, allowing for mild hydrolysis conditions that preserve the sensitive stereochemistry at the chiral centers. The acid-catalyzed cyclization likely proceeds through an iminium ion intermediate, which undergoes intramolecular Friedel-Crafts-type alkylation to close the seven-membered azepine ring. This mechanistic pathway is highly favorable entropically compared to intermolecular couplings, driving the reaction to completion with high conversion rates. Furthermore, the ability to perform the final carboxylation via carbon monoxide insertion under pressure provides a direct route to the 4-carboxylic acid without requiring additional carbon chain elongation steps.
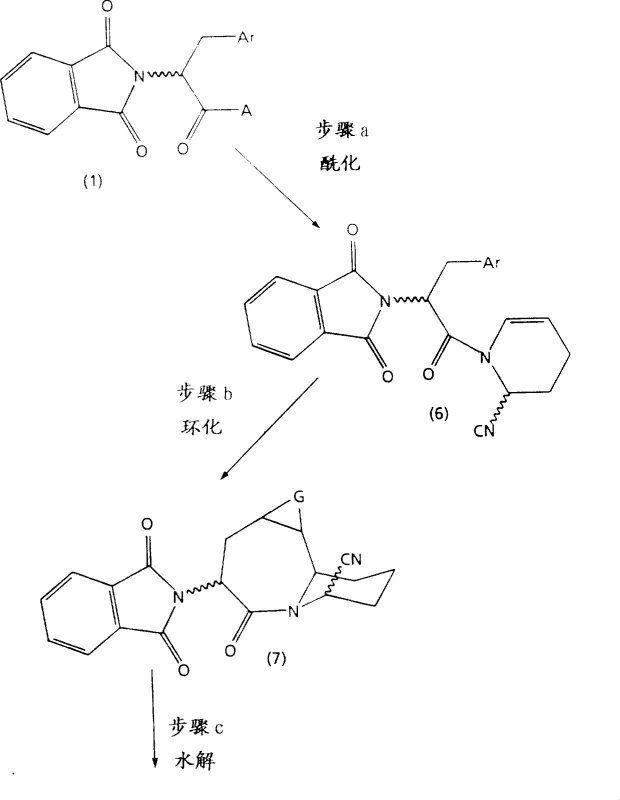
Impurity control is inherently built into this process design through the selection of specific reaction parameters and workup procedures. For instance, the use of molecular sieves during the glutaraldehyde condensation effectively removes water, shifting the equilibrium towards the desired dihydropyridine product and preventing hydrolysis of the sensitive imine intermediates. The patent details specific purification protocols, such as chromatography on silica gel using gradients of ethyl acetate and hexanes, which are scalable and robust for industrial applications. Additionally, the stereochemical outcome at the 4-position can be managed by selecting enantiomerically pure starting amino acids or by employing resolution techniques on the final carboxylic acid derivatives. This level of control ensures that the resulting intermediates meet the stringent enantiomeric excess (ee) requirements necessary for regulatory approval of the final drug substance, reducing the risk of batch rejection due to chiral impurities.
How to Synthesize Neprilysin Inhibitor Intermediate Efficiently
The synthesis of these high-value intermediates requires careful attention to reaction stoichiometry, temperature control, and solvent selection to maximize yield and purity. The process generally involves three critical stages: the initial formation of the dihydropyridine or tetrahydropyridine scaffold, the acid-mediated cyclization to form the benzazepine core, and the final functionalization to introduce the carboxylic acid group. Each step has been optimized in the patent examples to demonstrate feasibility on both laboratory and pilot scales, utilizing reagents that are commercially available and cost-effective. Operators must adhere to strict safety protocols, particularly when handling strong acids like triflic acid or operating pressure vessels for carbon monoxide reactions, to ensure personnel safety and process reliability. Detailed standard operating procedures (SOPs) derived from this patent data provide a clear roadmap for process chemists aiming to implement this technology in a GMP environment.
- React phthalimido aryl amino acid derivatives with glutaraldehyde or 2-cyano-1,2,3,4-tetrahydro-pyridine to form dihydropyridine or tetrahydropyridine precursors.
- Perform acid-catalyzed cyclization using trifluoromethanesulfonic acid or sulfuric acid mixtures to form the hexahydro-oxopyrido-benzazepine core.
- Execute final hydrolysis or carbon monoxide insertion followed by hydration to generate the 4-carboxylic acid functionality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of cost stability and supply security. By simplifying the molecular construction of the core scaffold, the process reduces the total number of unit operations required, which directly correlates to lower manufacturing overhead and reduced consumption of utilities and solvents. The reliance on commodity chemicals such as glutaraldehyde, sulfuric acid, and common organic solvents mitigates the risk of supply disruptions associated with specialized or proprietary reagents. This commoditization of raw materials ensures a more resilient supply chain capable of withstanding market volatility and geopolitical shifts that often impact the availability of exotic catalysts. Furthermore, the robustness of the reaction conditions allows for flexible manufacturing schedules, enabling producers to respond quickly to fluctuations in demand without compromising product quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in step count significantly lower the direct material costs associated with producing these intermediates. By avoiding complex protection-deprotection sequences and utilizing efficient one-pot cyclization strategies, the overall process mass intensity (PMI) is improved, leading to substantial waste reduction and lower disposal costs. The ability to isolate intermediates with high purity reduces the need for extensive recrystallization or chromatographic purification at later stages, further driving down processing expenses. These efficiencies compound to offer a competitive pricing structure for the final API, enhancing the commercial viability of drugs utilizing this scaffold.
- Enhanced Supply Chain Reliability: The use of widely available starting materials like phthalic anhydride and phenylalanine derivatives ensures a stable upstream supply base that is less susceptible to shortages. The process tolerates a range of reaction conditions, providing manufacturing flexibility that allows for production across different facilities without extensive re-validation. This geographical diversification capability is crucial for multinational pharmaceutical companies seeking to de-risk their supply chains against regional disruptions. Additionally, the scalability of the process from gram to kilogram scales has been demonstrated, providing confidence that commercial volumes can be met consistently to support clinical trials and market launch requirements.
- Scalability and Environmental Compliance: The synthetic route is designed with green chemistry principles in mind, minimizing the generation of hazardous byproducts and facilitating easier waste treatment. The aqueous workups and standard solvent recovery methods align with modern environmental regulations, reducing the regulatory burden on manufacturing sites. The process avoids the use of persistent organic pollutants or heavy metals, simplifying the environmental impact assessment and permitting processes for new production lines. This alignment with sustainability goals not only reduces compliance costs but also enhances the corporate social responsibility profile of the supply chain, which is increasingly important for stakeholder engagement and brand reputation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. It clarifies the operational requirements and potential bottlenecks, allowing for more accurate project planning and resource allocation.
Q: How is stereochemical control achieved at the 4-position?
A: Stereochemistry is primarily determined by the starting phthalimido aryl amino acid. Specific isomers can be further resolved via chromatography on chiral stationary phases or fractional recrystallization of the 4-position carboxylic acid derivatives.
Q: What are the advantages of the glutaraldehyde condensation route?
A: This route offers a direct pathway to the 1,4-dihydropyridine scaffold under mild acidic catalysis, avoiding complex multi-step ring constructions and improving overall atom economy compared to traditional methods.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes robust reagents like sulfuric acid and standard solvents such as dichloromethane and ethyl acetate. The reaction conditions, including pressure vessel steps for CO insertion, are well-suited for industrial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Neprilysin Inhibitor Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at translating complex patent methodologies like CN1170413A into robust, GMP-compliant manufacturing processes that deliver consistent quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to ensure every batch meets the highest international standards. Our commitment to excellence extends beyond mere production; we act as a strategic partner, offering deep technical expertise to optimize yields and resolve any process challenges that may arise during scale-up.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized processes can reduce your overall expenditure. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to accelerate your drug development timeline and secure a reliable supply of critical intermediates for your cardiovascular therapeutic programs.
