Advanced Manufacturing of Beta-Alanine Derivatives for Cardiovascular API Intermediates
The pharmaceutical industry continuously seeks robust and scalable pathways for producing complex intermediates, particularly those serving as critical building blocks for cardiovascular therapeutics. Patent CN1329595A introduces a significant advancement in the preparation of beta-alanine derivatives, which function as potent glycoprotein IIb/IIIa antagonists and platelet aggregation inhibitors. This technology addresses the persistent challenges of yield optimization and stereochemical purity that have historically plagued the synthesis of these vital molecules. By leveraging a novel sequence of deprotection and acylation reactions, the disclosed method enables the efficient transformation of protected precursors into high-value active pharmaceutical ingredients. For R&D directors and procurement specialists, understanding the nuances of this patented approach is essential for securing a reliable supply chain and minimizing production bottlenecks in the competitive landscape of anti-thrombotic drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for beta-alanine derivatives often suffer from inefficient multi-step sequences that rely on harsh reaction conditions or expensive, difficult-to-remove reagents. Conventional methodologies frequently struggle with the selective manipulation of amino and carboxyl functionalities without compromising the integrity of the chiral centers, leading to racemization and reduced biological efficacy. Furthermore, older processes may utilize stoichiometric amounts of toxic heavy metals or require extreme temperatures that pose significant safety risks and environmental compliance burdens during commercial scale-up. These inefficiencies result in lower overall yields, increased waste generation, and higher operational costs, making it challenging for manufacturers to maintain consistent quality and supply continuity for downstream API production. The reliance on fragile protecting groups that demand specific, non-standard conditions further complicates the process validation and regulatory approval timelines.
The Novel Approach
The innovative process outlined in the patent data overcomes these historical barriers by implementing a streamlined strategy focused on the sequential elimination of carboxyl protecting groups followed by precise amino group acylation. This method utilizes widely available and cost-effective reagents such as palladium on carbon for catalytic hydrogenation and common acid anhydrides for acylation, ensuring that the reaction environment remains mild and controllable. By optimizing the order of operations, the novel approach minimizes side reactions and preserves the stereochemical configuration of the beta-alanine backbone, resulting in superior product purity. The flexibility to use various standard protecting groups like benzyloxycarbonyl or tert-butoxycarbonyl allows for seamless integration into existing synthetic workflows. This technological shift not only enhances the chemical efficiency but also aligns with modern green chemistry principles by reducing solvent usage and simplifying purification steps, thereby offering a distinct competitive advantage in terms of both cost and sustainability.
Mechanistic Insights into Deprotection and Acylation Chemistry
The core of this synthesis lies in the meticulous control of functional group transformations, beginning with the elimination of the carboxyl protecting group from the precursor compound. This step can be achieved through hydrolysis using aqueous bases or acids, or alternatively via catalytic reduction depending on the specific nature of the protecting moiety employed. For instance, when using benzylic esters, catalytic hydrogenation with palladium catalysts effectively cleaves the bond under ambient pressure, releasing the free carboxylic acid without affecting other sensitive parts of the molecule. The reaction conditions are carefully tuned, often utilizing solvents like methanol or tetrahydrofuran to ensure complete solubility and reaction homogeneity. Following the deprotection, the newly exposed amino group undergoes acylation, a critical step that defines the final pharmacological profile of the derivative. This is typically accomplished using reactive acylating agents such as acetic anhydride or activated esters in the presence of mild bases, facilitating the formation of the amide bond with high specificity.
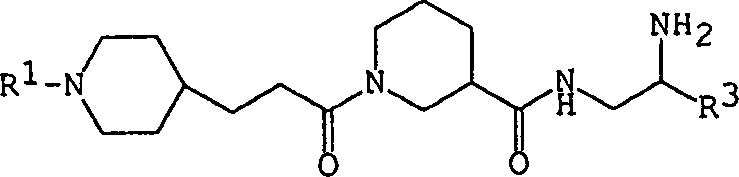
Understanding the impurity profile is paramount for R&D teams aiming to replicate this success at scale. The mechanism inherently suppresses the formation of di-acylated byproducts by controlling the stoichiometry of the acylating agent and maintaining low reaction temperatures during the addition phase. The use of scavengers or specific workup procedures, such as pH adjustment and liquid-liquid extraction, ensures that residual catalysts and unreacted starting materials are effectively removed. This rigorous control over the reaction pathway translates directly into a cleaner crude product, reducing the burden on downstream purification units like crystallization or chromatography. Consequently, the final beta-alanine derivative exhibits the high optical purity required for potent glycoprotein IIb/IIIa antagonism, validating the mechanistic robustness of this patented synthetic route for industrial application.
How to Synthesize Beta-Alanine Derivatives Efficiently
Executing this synthesis requires strict adherence to the optimized parameters regarding temperature, solvent choice, and reagent addition rates to maximize yield and safety. The process begins with the preparation of the protected precursor, followed by the critical deprotection step where monitoring reaction progress via HPLC or TLC is essential to prevent over-reduction or hydrolysis. Once the intermediate is isolated, the acylation must be performed under anhydrous conditions to avoid hydrolysis of the acylating agent, ensuring maximum conversion to the desired amide. Detailed standard operating procedures for scaling this chemistry from gram to kilogram quantities involve specific equipment setups for hydrogenation and exotherm management during acylation.
- Perform elimination of the carboxyl protecting group on the precursor compound using hydrolysis or catalytic reduction under controlled pH and temperature conditions.
- Execute the acylation reaction of the resulting amino group using suitable acylating agents such as acid anhydrides or active esters in organic solvents.
- Isolate and purify the final beta-alanine derivative product through crystallization or column chromatography to meet stringent pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing route offers substantial benefits by leveraging commodity chemicals and standard processing equipment, which significantly mitigates supply chain risks associated with specialized reagents. The elimination of exotic catalysts and the use of common solvents mean that procurement teams can source raw materials from multiple qualified vendors, ensuring business continuity even during market fluctuations. The simplified workflow reduces the number of unit operations required, which directly correlates to lower utility consumption and reduced labor hours per batch. This efficiency drives down the overall cost of goods sold without compromising on the quality standards demanded by regulatory bodies. Furthermore, the robustness of the chemistry allows for flexible production scheduling, enabling manufacturers to respond quickly to changes in market demand for cardiovascular intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive stoichiometric reagents with catalytic systems that can be recovered and reused, such as palladium on carbon. By avoiding complex protection-deprotection sequences that generate excessive waste, the material throughput is improved, leading to better atom economy. The use of standard acid anhydrides instead of custom-synthesized acylating agents further lowers the raw material expenditure. These cumulative efficiencies result in a more economical production model that enhances profit margins while maintaining competitive pricing for downstream API customers.
- Enhanced Supply Chain Reliability: Reliance on globally available chemicals like acetic anhydride, sodium hydroxide, and common organic solvents ensures that production is not held hostage by the availability of niche specialty items. The modular nature of the synthesis allows for easy replication across different manufacturing sites, diversifying the supply base and reducing geopolitical risks. Additionally, the stability of the intermediates formed during the process facilitates safer storage and transportation, minimizing losses due to degradation. This resilience is crucial for maintaining long-term contracts with major pharmaceutical clients who require guaranteed delivery schedules.
- Scalability and Environmental Compliance: The reaction conditions are inherently safe and scalable, operating at near-ambient pressures and temperatures that do not require specialized high-pressure reactors or cryogenic cooling systems. This simplicity facilitates a smoother technology transfer from pilot plant to commercial manufacturing scales. Moreover, the reduced generation of hazardous waste and the potential for solvent recycling align with increasingly stringent environmental regulations, lowering the costs associated with waste disposal and environmental permitting. This sustainable approach future-proofs the manufacturing asset against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-alanine derivative synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for process engineers and project managers. Understanding these details helps in assessing the feasibility of adopting this route for specific project requirements.
Q: What are the key advantages of the new synthesis process for beta-alanine derivatives?
A: The process described in patent CN1329595A offers high yield production and excellent stereochemical control, utilizing standard reagents like palladium catalysts and acid anhydrides which simplifies scale-up.
Q: Can this manufacturing route be scaled for commercial API production?
A: Yes, the methodology employs robust unit operations such as catalytic hydrogenation and solvent extraction, which are well-established in industrial chemical manufacturing for reliable commercial scale-up.
Q: What types of protecting groups are compatible with this synthesis method?
A: The process supports a wide range of conventional protecting groups including benzyloxycarbonyl and tert-butoxycarbonyl, allowing flexibility in upstream synthetic planning.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Alanine Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patented chemistries into commercial reality, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in protective group chemistry and catalytic hydrogenation, ensuring that the transition from laboratory scale to industrial manufacturing is seamless and compliant with cGMP standards. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-alanine derivative meets the exacting requirements of global pharmaceutical partners. Our commitment to quality and consistency makes us the preferred partner for companies seeking to secure their supply of critical cardiovascular intermediates.
We invite you to collaborate with us to optimize your supply chain and achieve significant cost efficiencies in your API manufacturing programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us help you engineer a more resilient and profitable production future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
