Scalable Synthesis of 5-Chloro-2-Pentanone via Novel Composite Catalytic Oxidation for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective pathways for critical intermediates, and the synthesis of 5-chloro-2-pentanone stands as a prime example of where process innovation drives supply chain resilience. Patent CN114591157A introduces a groundbreaking methodology that fundamentally reimagines the production of this vital ketone, shifting away from legacy routes plagued by high costs and hazardous conditions. By leveraging methyl 4-hydroxypentanoate as a foundational building block and employing a sophisticated composite catalytic system, this technology delivers a streamlined four-step sequence that balances high yield with exceptional purity. For R&D leaders and procurement strategists alike, this patent represents a significant leap forward, offering a viable solution to the bottlenecks often encountered in the manufacture of quinolone drugs and antimalarial agents like chloroquine phosphate. The integration of green chemistry principles, specifically the use of molecular oxygen as the terminal oxidant, further underscores the commercial viability of this approach in a regulatory environment that increasingly demands sustainable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 5-chloro-2-pentanone has been hindered by significant technical and economic barriers that limit its scalability and safety profile. Prior art methodologies often rely on 5-chloropentylene as a starting material, necessitating the use of expensive gold catalysts under acidic conditions, which results in suboptimal yields and prohibitive raw material costs that erode profit margins. Alternative routes utilizing 4-chlorobutyryl chloride require the handling of highly reactive and dangerous reagents such as butyl lithium and triphenylphosphine methyl bromide, creating severe safety hazards that are difficult to mitigate in a large-scale production environment. Furthermore, processes starting from acetyl-n-propanol suffer from high input costs and limited availability, making them unsuitable for meeting the surging global demand for this intermediate. These conventional pathways are characterized by harsh reaction conditions, low atom economy, and complex purification requirements, rendering them inefficient for modern, high-volume pharmaceutical manufacturing where consistency and cost control are paramount.
The Novel Approach
In stark contrast to these legacy methods, the novel process detailed in the patent utilizes a strategic protection-reduction-oxidation sequence that begins with the abundant and inexpensive methyl 4-hydroxypentanoate. This innovative route initiates with the protection of the hydroxyl group using 1-(2-chloroethoxy)-2-methoxyethane, establishing a stable intermediate that withstands subsequent rigorous chemical transformations. 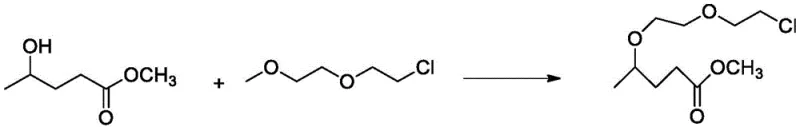 The subsequent steps involve a chemoselective reduction of the ester moiety followed by a tandem chlorination and deprotection event, culminating in a highly efficient catalytic oxidation. This approach not only circumvents the need for precious metal catalysts or pyrophoric reagents but also simplifies the overall operational workflow, allowing for milder reaction temperatures and pressures. By decoupling the synthesis from scarce raw materials and replacing them with commodity chemicals, this method establishes a new benchmark for economic efficiency and operational safety in the production of halogenated ketones.
The subsequent steps involve a chemoselective reduction of the ester moiety followed by a tandem chlorination and deprotection event, culminating in a highly efficient catalytic oxidation. This approach not only circumvents the need for precious metal catalysts or pyrophoric reagents but also simplifies the overall operational workflow, allowing for milder reaction temperatures and pressures. By decoupling the synthesis from scarce raw materials and replacing them with commodity chemicals, this method establishes a new benchmark for economic efficiency and operational safety in the production of halogenated ketones.
Mechanistic Insights into Composite Catalytic Oxidation
The cornerstone of this technological advancement lies in the deployment of a uniquely engineered composite catalyst that drives the final oxidation step with remarkable specificity and efficiency. This catalyst system is constructed by intercalating cobalt tetra(4-sulfophenyl)porphyrin anions into a zinc-aluminum layered double hydroxide (Zn-Al LDH) matrix, creating a heterogeneous structure with expanded interlayer spacing. This structural modification exposes a higher density of alkaline active sites, which accelerates the activation of molecular oxygen and the subsequent generation of phthalimide N-oxyl (PINO) radicals from N-hydroxyphthalimide (NHPI). These radicals act as potent hydrogen abstractors, selectively removing hydrogen atoms from the alcohol intermediate to facilitate its conversion into the target ketone without over-oxidizing the molecule or degrading the sensitive chloro-alkyl chain. 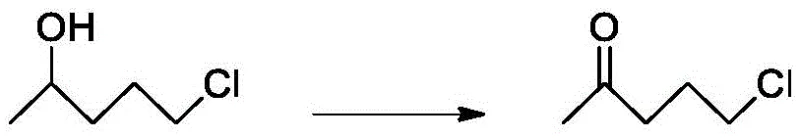 The synergy between the LDH support and the cobalt porphyrin complex ensures that the oxidation proceeds rapidly under mild conditions, minimizing energy consumption and reducing the formation of unwanted byproducts that typically complicate downstream purification.
The synergy between the LDH support and the cobalt porphyrin complex ensures that the oxidation proceeds rapidly under mild conditions, minimizing energy consumption and reducing the formation of unwanted byproducts that typically complicate downstream purification.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over traditional stoichiometric oxidants like chromium or manganese salts, which often leave behind toxic metal residues that are difficult to remove to pharmaceutical standards. The heterogeneous nature of the composite catalyst allows for easy filtration and recovery, ensuring that the final product stream remains free from heavy metal contamination. Furthermore, the selectivity of the PINO radical mechanism prevents side reactions at the chloro-substituted carbon, preserving the integrity of the alkyl halide functionality which is crucial for subsequent nucleophilic substitutions in API synthesis. This level of control over the reaction trajectory translates directly into a cleaner crude product profile, reducing the burden on distillation columns and increasing the overall throughput of the manufacturing facility while maintaining a purity profile that exceeds 99%.
How to Synthesize 5-Chloro-2-Pentanone Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the novel catalytic system and protection strategy. The process is designed to be operationally simple, utilizing standard reactor equipment and common solvents like tetrahydrofuran and toluene, which facilitates easy technology transfer from the laboratory to pilot and commercial scales. Operators must pay close attention to the stoichiometry of the protecting group and the temperature control during the initial protection phase to ensure complete conversion before proceeding to the reduction step. The following guide outlines the critical operational phases required to achieve the high yields and purity reported in the patent data, serving as a foundational reference for process engineers looking to implement this route.
- Protect the hydroxyl group of methyl 4-hydroxypentanoate using 1-(2-chloroethoxy)-2-methoxyethane in THF at 0°C.
- Reduce the ester group to an alcohol using Sodium Borohydride and Zinc Chloride under reflux conditions.
- Perform chlorination and deprotection simultaneously using concentrated hydrochloric acid and anhydrous zinc chloride.
- Oxidize the resulting alcohol to the target ketone using oxygen and a composite catalyst comprising Zn-Al LDH and Cobalt Porphyrin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers transformative potential in terms of cost structure and supply security. By shifting the raw material base from specialized, high-cost precursors like 5-chloropentylene to commodity esters, the bill of materials is significantly optimized, insulating the production cost from the volatility associated with niche chemical markets. The elimination of precious metal catalysts such as gold removes a major capital expense and reduces the dependency on supply chains for critical minerals, thereby enhancing the long-term stability of the manufacturing process. Additionally, the avoidance of hazardous reagents like butyl lithium simplifies regulatory compliance and lowers insurance and safety infrastructure costs, contributing to a leaner and more agile operational model.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive inputs with widely available bulk chemicals and the implementation of a catalytic rather than stoichiometric oxidation system. The use of molecular oxygen as the oxidant is inherently cheaper than purchasing proprietary oxidizing agents, and the recyclability of the composite catalyst further drives down the variable cost per kilogram of product. Moreover, the high selectivity of the reaction minimizes waste generation and solvent usage, leading to substantial savings in waste disposal and solvent recovery operations. These cumulative efficiencies result in a markedly lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing reliability is dramatically improved because the primary starting material, methyl 4-hydroxypentanoate, is produced by multiple suppliers globally, reducing the risk of single-source bottlenecks. The robustness of the chemical process, which tolerates mild conditions and uses stable reagents, ensures consistent batch-to-batch quality and reduces the likelihood of production delays caused by reaction failures or safety incidents. This stability allows for more accurate forecasting and inventory planning, enabling supply chain teams to maintain optimal stock levels without the need for excessive safety buffers. Consequently, lead times for high-purity pharmaceutical intermediates can be reduced, ensuring a steady flow of materials to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, utilizing unit operations such as reflux, filtration, and distillation that are standard in multi-purpose chemical plants. The green chemistry attributes, including the use of oxygen and the absence of heavy metal waste streams, align perfectly with increasingly stringent environmental regulations, minimizing the permitting hurdles for capacity expansion. The ability to run the reaction at atmospheric pressure with oxygen sparging simplifies the engineering requirements for large reactors, making it easier to transition from pilot batches to multi-ton production campaigns. This scalability ensures that the supply can grow in tandem with market demand without requiring prohibitive capital investment in specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative data. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages demonstrated in the experimental examples, highlighting the superiority of the composite catalyst and the overall process design.
Q: What are the advantages of the composite catalyst over traditional oxidants?
A: The novel composite catalyst, featuring a Zn-Al layered double hydroxide intercalated with cobalt tetra(4-sulfophenyl)porphyrin, offers superior activity and selectivity compared to traditional hydrogen peroxide or FeCl3 systems. It facilitates the generation of phthalimide N-oxyl radicals under mild oxygen atmosphere, significantly improving conversion rates and product purity while avoiding heavy metal contamination.
Q: Why is methyl 4-hydroxypentanoate preferred as a starting material?
A: Unlike prior art routes that rely on expensive 5-chloropentylene with gold catalysts or hazardous butyl lithium reagents, methyl 4-hydroxypentanoate is a cost-effective and readily available bulk chemical. This shift in raw material strategy drastically reduces the Bill of Materials (BOM) cost and eliminates the safety risks associated with pyrophoric reagents, making the process inherently safer for large-scale manufacturing.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process achieves high purity through a combination of selective protection-deprotection strategies and a highly specific catalytic oxidation step. The use of the composite catalyst minimizes over-oxidation byproducts, and the final distillation step ensures the removal of residual solvents and impurities, consistently delivering product purity exceeding 99% suitable for sensitive API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-2-Pentanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 5-chloro-2-pentanone meets the exacting standards required by the global pharmaceutical industry. Our facility is equipped to handle the specific nuances of this catalytic oxidation process, delivering a product that supports your drug development timelines with unwavering consistency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a partnership built on transparency, technical excellence, and mutual growth.
