Advanced Photocatalytic Route to Benzoylmethyl Cyclopropanes for Commercial Scale-up
Introduction to Next-Generation Cyclopropane Synthesis
The structural integrity and metabolic stability provided by cyclopropyl rings make them indispensable motifs in modern medicinal chemistry, appearing in blockbuster drugs ranging from respiratory treatments like Montelukast to antiviral agents such as Lumacaftor. As depicted in the diverse array of bioactive molecules below, the ability to efficiently construct these strained three-membered rings is a critical capability for any advanced chemical manufacturer. 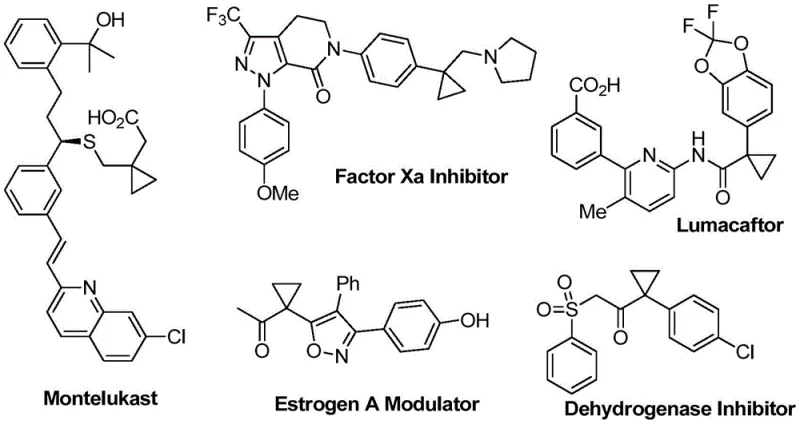 Patent CN110156831B addresses this critical need by disclosing a novel method for the photocatalytic synthesis of benzoylmethyl-substituted cyclopropanes. This technology represents a significant leap forward in synthetic methodology, moving away from thermal activation towards sustainable visible-light-driven processes. By leveraging a unique acyl radical addition and ionic cyclization cascade, this patent offers a pathway to complex scaffolds that are notoriously difficult to access via conventional means, thereby opening new avenues for the development of high-purity pharmaceutical intermediates.
Patent CN110156831B addresses this critical need by disclosing a novel method for the photocatalytic synthesis of benzoylmethyl-substituted cyclopropanes. This technology represents a significant leap forward in synthetic methodology, moving away from thermal activation towards sustainable visible-light-driven processes. By leveraging a unique acyl radical addition and ionic cyclization cascade, this patent offers a pathway to complex scaffolds that are notoriously difficult to access via conventional means, thereby opening new avenues for the development of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of cyclopropane rings has relied heavily on methods that often suffer from significant operational drawbacks, limiting their utility in complex molecule synthesis. Traditional approaches frequently necessitate the use of stoichiometric amounts of strong bases, hazardous carbenoid precursors, or transition metal catalysts that require rigorous removal steps to meet pharmaceutical purity standards. For instance, earlier photocatalytic strategies, such as those utilizing gem-dihalogenated hydrocarbons shown in prior art, often demand stoichiometric amines as reducing agents to drive the catalytic cycle. Furthermore, other established protocols involving diiodomethane require additional additives like sodium sulfite to manage side reactions and facilitate the radical process. These requirements not only increase the material cost but also complicate the downstream purification process, generating substantial waste streams that pose environmental and disposal challenges for large-scale operations.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN110156831B introduces a streamlined, redox-neutral strategy that elegantly bypasses the need for external stoichiometric reductants or harsh additives. 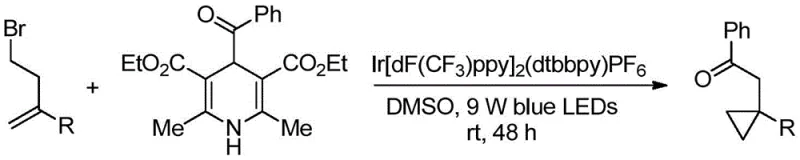 The core innovation lies in the use of 4-benzoyl-1,4-dihydropyridine as a stable and efficient acyl radical precursor, which, under the influence of visible light and an iridium photocatalyst, generates the reactive species necessary for ring closure. This approach operates under exceptionally mild conditions, typically at room temperature in dimethyl sulfoxide (DMSO), thereby preserving sensitive functional groups that might otherwise decompose under thermal stress. The result is a highly versatile synthetic platform capable of tolerating a broad spectrum of substituents, enabling the rapid assembly of diverse benzoylmethyl-substituted cyclopropane libraries essential for drug discovery and process development.
The core innovation lies in the use of 4-benzoyl-1,4-dihydropyridine as a stable and efficient acyl radical precursor, which, under the influence of visible light and an iridium photocatalyst, generates the reactive species necessary for ring closure. This approach operates under exceptionally mild conditions, typically at room temperature in dimethyl sulfoxide (DMSO), thereby preserving sensitive functional groups that might otherwise decompose under thermal stress. The result is a highly versatile synthetic platform capable of tolerating a broad spectrum of substituents, enabling the rapid assembly of diverse benzoylmethyl-substituted cyclopropane libraries essential for drug discovery and process development.
Mechanistic Insights into Photocatalytic Acyl Radical Cyclization
The success of this transformation hinges on the sophisticated interplay between the iridium photocatalyst and the dihydropyridine derivative under blue LED irradiation. Upon absorption of photons, the excited state of the iridium complex engages in a single electron transfer (SET) event with the 4-benzoyl-1,4-dihydropyridine. This oxidation step triggers the fragmentation of the dihydropyridine, releasing a nucleophilic acyl radical along with a pyridine byproduct. The generated acyl radical then undergoes a conjugate addition to the electron-deficient olefin moiety of the homoallyl bromide substrate. This initial addition creates a transient carbon-centered radical intermediate, which is strategically positioned to undergo a rapid intramolecular nucleophilic substitution (SN2) with the pendant bromide leaving group. This cascade sequence—radical addition followed by ionic cyclization—efficiently constructs the cyclopropane ring while installing the ketone functionality in a single operational step, showcasing remarkable atom economy and step efficiency.
From an impurity control perspective, the mild nature of this photocatalytic cycle offers distinct advantages over thermal radical initiators. Because the reaction proceeds at ambient temperature without the need for high-energy thermal activation, the formation of non-specific radical byproducts and polymerization side reactions is significantly suppressed. The specificity of the acyl radical generation ensures that the reaction trajectory remains focused on the desired cyclization pathway, minimizing the generation of regioisomers or over-reduced species. Furthermore, the use of a homogeneous iridium catalyst allows for precise control over the reaction kinetics through light intensity modulation. This level of control is paramount for maintaining high product quality, as it prevents the accumulation of reactive intermediates that could lead to complex impurity profiles, thus simplifying the purification workflow and ensuring the delivery of high-purity intermediates suitable for subsequent coupling reactions in API synthesis.
How to Synthesize Benzoylmethyl Cyclopropane Efficiently
The practical implementation of this photocatalytic protocol is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The process begins by charging a reaction vessel with the homoallyl bromide substrate, the iridium photocatalyst, and the acyl radical precursor under an inert atmosphere to prevent oxygen quenching of the excited states. Following the addition of the polar aprotic solvent, the mixture is subjected to continuous irradiation with blue LEDs while stirring for a period of approximately 45 to 50 hours. This extended reaction time ensures complete conversion of the starting materials, driven by the steady-state concentration of radicals maintained by the photon flux. Upon completion, the workup involves a simple aqueous extraction to remove inorganic salts and polar byproducts, followed by standard chromatographic purification to isolate the target cyclopropane derivative.
- Combine homoallyl bromide, Ir[dF(CF3)ppy]2(dtbbpy)PF6 catalyst, and 4-benzoyl-1,4-dihydropyridine in a reaction tube under nitrogen protection.
- Inject dimethyl sulfoxide (DMSO) as the solvent and stir the mixture under 9W blue LED illumination for 45-50 hours at room temperature.
- Quench the reaction with ethyl acetate and saturated brine, extract the organic phase, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this photocatalytic technology presents a compelling value proposition centered on cost efficiency and operational simplicity. By eliminating the requirement for stoichiometric reducing agents and hazardous additives commonly found in traditional cyclopropanation methods, the overall material cost per kilogram of product is substantially reduced. The reliance on visible light as the primary energy source, rather than high-temperature heating or cryogenic cooling, translates directly into lower utility costs and reduced strain on facility infrastructure. Moreover, the mild reaction conditions minimize the risk of thermal runaway incidents, enhancing workplace safety and potentially lowering insurance premiums associated with chemical manufacturing. These factors collectively contribute to a more robust and economically viable supply chain for complex cyclic intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reagent profile and the reduction of waste disposal costs. Since the method avoids the use of expensive stoichiometric metals or hazardous carbenoid reagents, the raw material expenditure is optimized. Additionally, the high functional group tolerance reduces the need for protecting group strategies, which typically add multiple synthetic steps and associated costs. The streamlined workup procedure, which relies on standard extraction and distillation techniques rather than complex scavenging protocols for heavy metals, further decreases the operational expenditure required to bring the product to specification. This holistic reduction in process complexity allows for significant cost savings in the manufacturing of high-value pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: The robustness of the starting materials used in this protocol ensures a stable and reliable supply chain. Homoallyl bromides and dihydropyridine derivatives are generally commercially available or easily synthesized from commodity chemicals, reducing the risk of supply disruptions caused by niche precursor shortages. The scalability of the photocatalytic setup, which can be adapted from batch reactors to continuous flow systems, provides flexibility in meeting fluctuating demand volumes. This adaptability ensures that production timelines can be maintained even during periods of high market volatility, securing the continuity of supply for downstream API manufacturers who depend on consistent delivery of these critical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this green chemistry approach aligns perfectly with modern sustainability goals. The absence of toxic tin or heavy metal reagents simplifies the environmental impact assessment and facilitates easier regulatory approval for new drug applications. The use of DMSO, a solvent with a favorable safety profile and recyclability potential, supports greener manufacturing practices. Furthermore, the energy efficiency of LED-based photochemistry compared to thermal processes reduces the carbon footprint of the synthesis. These attributes not only enhance the corporate social responsibility profile of the manufacturing partner but also future-proof the supply chain against increasingly stringent environmental regulations regarding chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the feasibility and advantages of adopting this route for industrial applications. Understanding these details is crucial for technical teams evaluating the integration of this technology into existing production workflows.
Q: What are the key advantages of this photocatalytic method over traditional cyclopropanation?
A: Unlike traditional methods that often require stoichiometric reducing agents or harsh conditions, this patent utilizes a visible-light driven acyl radical addition strategy. This allows for milder reaction conditions (room temperature), better functional group compatibility, and eliminates the need for excess amine additives.
Q: What catalyst system is employed in this synthesis?
A: The process employs an Iridium-based photocatalyst, specifically Ir[dF(CF3)ppy]2(dtbbpy)PF6, which facilitates the single electron transfer (SET) necessary to generate the acyl radicals from 4-benzoyl-1,4-dihydropyridine under blue LED irradiation.
Q: Is this method suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the method operates at room temperature and uses standard solvents like DMSO, which simplifies thermal management during scale-up. The robustness of the photocatalytic cycle and the availability of starting materials support potential commercial expansion for complex API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoylmethyl Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in reshaping the landscape of fine chemical synthesis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet the most stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in managing complex photochemical reactions allows us to optimize yield and quality consistently, providing our partners with a competitive edge in the global pharmaceutical market.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how this innovative photocatalytic method can enhance your supply chain resilience and reduce overall manufacturing costs. Let us be your trusted partner in bringing high-quality cyclopropane intermediates to market faster and more efficiently.
