Advanced Synthesis of Gimeracil Intermediates: A Strategic Upgrade for Pharmaceutical Manufacturing
Advanced Synthesis of Gimeracil Intermediates: A Strategic Upgrade for Pharmaceutical Manufacturing
The pharmaceutical landscape is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes for critical active pharmaceutical ingredients (APIs) and their precursors. A pivotal development in this domain is documented in patent CN115504905A, which introduces a groundbreaking methodology for synthesizing gimeracil intermediates. Gimeracil, a potent dihydropyrimidine dehydrogenase inhibitor, is a crucial component of the oral anticancer drug S-1 (Tegafur/Gimeracil/Oteracil). The traditional synthesis pathways have long relied on hazardous starting materials that pose significant safety and environmental challenges. This new technical disclosure outlines a transformative approach that replaces toxic malononitrile with the safer alpha-cyanoacetamide, fundamentally altering the risk profile and economic viability of producing this high-value pharmaceutical intermediate. For R&D directors and procurement strategists, understanding this shift is essential for optimizing supply chains and ensuring regulatory compliance in oncology drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 3-cyano-4-methoxy-2(1H)-pyridone, the key precursor to gimeracil, has been dominated by routes utilizing malononitrile as the primary building block. As illustrated in the conventional reaction pathway below, this legacy method involves the condensation of malononitrile with trimethyl orthoacetate and DMF-DMA to form a butadiene intermediate, followed by cyclization. While chemically feasible, this approach suffers from severe drawbacks that impact both operational safety and bottom-line costs. Malononitrile is classified as a highly toxic substance, requiring stringent containment measures, specialized personal protective equipment, and complex waste treatment protocols to prevent environmental contamination. Furthermore, the volatility and reactivity of malononitrile can lead to inconsistent reaction profiles, often necessitating additional purification steps that erode overall yield and increase production lead times.
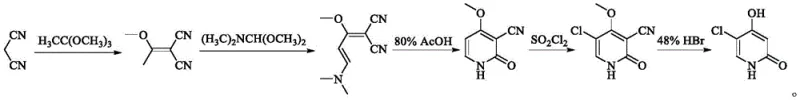
Beyond safety concerns, the economic implications of relying on malononitrile are substantial. The cost of sourcing high-purity malononitrile fluctuates with petrochemical markets, introducing volatility into the supply chain. Moreover, the "one-pot" nature of the traditional synthesis, while seemingly efficient, often traps impurities that are difficult to remove without expensive chromatography or recrystallization processes. For procurement managers overseeing the budget for oncology API production, these hidden costs associated with hazard management and yield loss accumulate rapidly. The reliance on such a hazardous feedstock also complicates regulatory filings in regions with increasingly strict environmental, health, and safety (EHS) standards, potentially delaying market entry for generic versions of S-1.
The Novel Approach
In stark contrast to the legacy methods, the technology disclosed in patent CN115504905A presents a superior synthetic strategy centered on the use of alpha-cyanoacetamide. This novel route begins with the condensation of alpha-cyanoacetamide (SM-1) and triethyl orthoacetate (SM-2), followed by a reaction with N,N-dimethylformamide dimethyl acetal (DMF-DMA, SM-3) to generate a new intermediate compound, designated as I-1. This intermediate is structurally distinct and serves as a stable platform for the subsequent formation of the pyridone ring. The shift to alpha-cyanoacetamide is not merely a substitution of reagents; it represents a fundamental redesign of the process chemistry to prioritize safety and efficiency. By eliminating the most toxic component of the traditional recipe, the new method drastically simplifies the EHS requirements for the manufacturing facility.
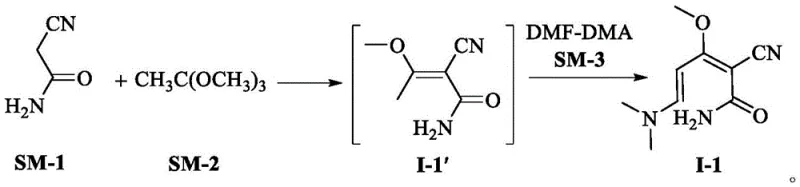
The operational advantages of this new approach are manifold. The reaction conditions are remarkably mild, with the initial condensation occurring at temperatures between 65-70°C and the subsequent amidation step proceeding efficiently at 50-55°C. These moderate thermal requirements reduce energy consumption compared to more vigorous traditional protocols. Furthermore, the isolation of intermediate I-1 is straightforward, typically involving simple concentration under reduced pressure, which minimizes solvent usage and waste generation. The resulting intermediate exhibits exceptional stability and purity, setting the stage for a high-yielding cyclization step. This streamlined workflow translates directly into reduced cycle times and lower operating expenses, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into the Condensation and Cyclization Process
To fully appreciate the technical robustness of this new synthesis, one must delve into the mechanistic details of how intermediate I-1 is constructed and subsequently converted into the target pyridone. The formation of I-1 involves a sequential nucleophilic attack and elimination sequence. Initially, the active methylene group of alpha-cyanoacetamide attacks the electrophilic carbon of triethyl orthoacetate, displacing ethanol and forming an enamine ether species. This transient species then reacts with DMF-DMA, where the dimethylamino group acts as a leaving group or participates in a transamination process, ultimately establishing the conjugated system necessary for ring closure. The presence of the amide functionality in the starting material, as opposed to the nitrile in malononitrile, alters the electronic density of the reaction center, facilitating a cleaner transformation with fewer side products.
Following the isolation of intermediate I-1, the conversion to the final key intermediate, 3-cyano-4-methoxy-2(1H)-pyridone (Compound I), proceeds via an acid-catalyzed cyclization. As shown in the reaction scheme below, treating I-1 with 80% acetic acid at elevated temperatures (110-115°C) triggers an intramolecular cyclization. The methoxy group and the cyano group are positioned perfectly to facilitate ring closure, driven by the thermodynamic stability of the aromatic pyridone system. The use of aqueous acetic acid serves a dual purpose: it provides the acidic protons necessary for catalysis and acts as a solvent that allows for easy crystallization of the product upon cooling. This mechanistic elegance ensures that the final product precipitates as high-quality needle-shaped crystals, inherently excluding many organic impurities that remain in the mother liquor.
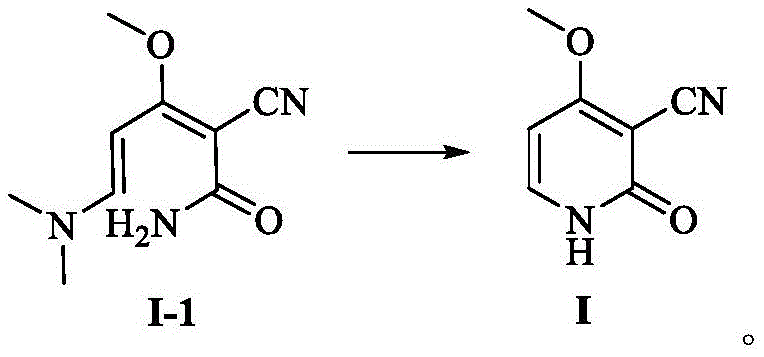
From an impurity control perspective, this mechanism offers significant advantages. The specific reactivity of alpha-cyanoacetamide minimizes the formation of polymeric byproducts that are common in malononitrile chemistry. Additionally, the crystallization step from acetic acid is highly selective, acting as a built-in purification stage. For R&D teams focused on impurity profiling, this means a simpler chromatographic profile and easier validation of the cleaning process. The high purity achieved (often >99.9% by HPLC) reduces the burden on downstream processing, ensuring that the subsequent chlorination and hydrolysis steps to form gimeracil proceed with maximum efficiency. This level of control over the chemical trajectory is critical for maintaining the stringent quality standards required for oncology APIs.
How to Synthesize 3-Cyano-4-Methoxy-2(1H)-Pyridone Efficiently
Implementing this novel synthesis requires precise control over reaction parameters to maximize the benefits of the new chemistry. The process is designed to be robust, yet attention to stoichiometry and temperature is paramount for achieving the reported high yields. The following guide outlines the critical operational phases derived from the patent examples, providing a framework for process engineers to adapt this technology for commercial scale-up. Detailed standardized synthesis steps are provided in the section below to ensure reproducibility and safety during pilot and plant operations.
- Condense alpha-cyanoacetamide (SM-1) with triethyl orthoacetate (SM-2) at 65-70°C, followed by reaction with DMF-DMA (SM-3) at 50-55°C to isolate intermediate I-1.
- Cyclize intermediate I-1 in 80% acetic acid solution at 110-115°C to precipitate the target compound 3-cyano-4-methoxy-2(1H)-pyridone.
- Purify the final product via crystallization and washing to achieve HPLC purity exceeding 99.9%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this new synthetic route offers compelling strategic benefits that extend beyond simple chemistry. The primary value proposition lies in the drastic simplification of the supply chain risk profile. By removing malononitrile from the bill of materials, companies eliminate the need for specialized hazardous material handling, storage, and disposal infrastructure. This reduction in regulatory overhead translates directly into lower fixed costs and reduced insurance premiums. Furthermore, alpha-cyanoacetamide is a widely available commodity chemical with a stable supply base, reducing the risk of raw material shortages that can plague specialty reagent markets. This stability ensures consistent production schedules and reliable delivery commitments to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic impact of switching to alpha-cyanoacetamide is profound. Since the starting material is less expensive and less hazardous than malononitrile, the direct material cost is lowered. More importantly, the simplified workup procedures—often requiring only concentration and crystallization rather than complex extractions or chromatography—significantly reduce labor and utility costs. The high yields reported (exceeding 98% in some examples) mean that less raw material is wasted, maximizing the output per batch. Additionally, the reduced need for extensive waste treatment lowers the overall cost of goods sold (COGS), providing a competitive edge in the pricing of generic oncology medications.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of common, non-restricted reagents. Unlike toxic precursors that may face shipping restrictions or regulatory scrutiny, alpha-cyanoacetamide and triethyl orthoacetate are standard industrial chemicals with multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. For logistics planners, this translates to fewer delays and a more predictable flow of intermediates, ensuring that API production lines remain operational without interruption.
- Scalability and Environmental Compliance: Scaling this process from the laboratory to multi-ton production is facilitated by the mild reaction conditions and the absence of exothermic hazards associated with malononitrile. The use of acetic acid as a cyclization solvent is environmentally favorable compared to chlorinated solvents often used in older methods. The process generates less hazardous waste, aligning with green chemistry principles and simplifying compliance with increasingly strict environmental regulations. This ease of scale-up allows manufacturers to respond quickly to market demand surges for gimeracil, ensuring that the supply of this critical cancer therapy component remains uninterrupted and sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel gimeracil intermediate synthesis. These answers are derived directly from the experimental data and technical specifications outlined in patent CN115504905A, providing clarity for stakeholders evaluating this technology for adoption. Understanding these details is crucial for making informed decisions about process validation and vendor qualification.
Q: Why is alpha-cyanoacetamide preferred over malononitrile for gimeracil synthesis?
A: Alpha-cyanoacetamide significantly reduces toxicity risks associated with handling malononitrile, improving operational safety and lowering environmental compliance costs while maintaining high reaction yields.
Q: What is the expected purity of the intermediate produced via this novel route?
A: The patented process consistently delivers intermediate I-1 and the final pyridone derivative with HPLC purity levels exceeding 99.5%, often reaching up to 99.92% under optimized conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions (50-115°C) and avoids hazardous reagents, making it highly scalable and robust for commercial manufacturing from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gimeracil Intermediate Supplier
The technological advancements detailed in patent CN115504905A represent a significant leap forward in the manufacture of oncology intermediates, yet realizing their full potential requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive CDMO services tailored to the complex needs of pharmaceutical clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-cyano-4-methoxy-2(1H)-pyridone meets the highest global standards.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis can enhance your supply chain efficiency. By leveraging our expertise, you can achieve significant operational improvements and secure a stable source of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for cost-effective and compliant drug manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
