Advanced Fluoroalkane Manufacturing: Safe, Scalable Synthesis from Fluorinated Epoxides
The landscape of fluorinated chemical manufacturing is undergoing a significant transformation driven by the urgent need for safer, more selective, and environmentally sustainable processes. Patent CN114507115A introduces a groundbreaking methodology for preparing fluoroalkane compounds directly from fluorinated epoxides, utilizing amine hydrogen fluoride salts as the fluorinating agent. This innovation addresses critical pain points associated with traditional electrofluorination and elemental fluorine gas methods, which have long plagued the industry with safety hazards and selectivity issues. By leveraging stable amine-HF complexes in either aprotic or organic amine solvents, this technology enables the efficient ring-opening and subsequent fluorination of epoxide structures under moderate thermal conditions ranging from 50 to 200°C. For R&D directors and procurement specialists seeking a reliable fluoroalkane supplier, this patent represents a pivotal shift towards high-purity fluoroalkane compounds that can be produced with exceptional consistency and minimal by-product formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluoroalkane compounds has relied heavily on two primary technologies: electrochemical fluorination and direct fluorination using elemental fluorine gas. Electrochemical fluorination, while established, often suffers from low current efficiency and the generation of complex waste streams that require extensive treatment. More critically, the use of fluorine gas (F2) presents extreme safety challenges due to its high reactivity and toxicity, necessitating specialized, expensive infrastructure for containment and handling. Furthermore, fluorine gas is notoriously non-selective; when reacting with substrates containing other heteroatoms such as hydrogen, chlorine, or bromine, it frequently induces unwanted substitution reactions alongside the desired addition, leading to a proliferation of by-products and a significant reduction in overall yield. These factors collectively drive up the cost of goods sold and complicate the supply chain for high-purity intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in CN114507115A utilizes fluorinated epoxides as the starting material, reacting them with amine hydrogen fluoride salts in a controlled liquid-phase environment. This method fundamentally alters the risk profile of the synthesis by replacing hazardous gaseous fluorine with stable, easy-to-handle solid or liquid amine-HF salts such as triethylamine hydrogen fluoride. The process operates effectively in common organic solvents like diethylene glycol dimethyl ether or even in the amine itself, allowing for homogeneous reaction conditions that enhance heat and mass transfer. 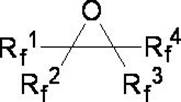 As illustrated by the general structure of the reactants, this method accommodates a wide variety of substituents (Rf groups), providing a versatile platform for generating diverse fluoroalkane derivatives. The result is a streamlined workflow that eliminates the need for specialized electrochemical cells or F2 gas handling systems, thereby facilitating cost reduction in fluorinated material manufacturing through simplified infrastructure requirements.
As illustrated by the general structure of the reactants, this method accommodates a wide variety of substituents (Rf groups), providing a versatile platform for generating diverse fluoroalkane derivatives. The result is a streamlined workflow that eliminates the need for specialized electrochemical cells or F2 gas handling systems, thereby facilitating cost reduction in fluorinated material manufacturing through simplified infrastructure requirements.
Mechanistic Insights into Amine-HF Mediated Ring Opening and Fluorination
The core chemical innovation lies in the dual functionality of the amine hydrogen fluoride salt, which acts as both a source of nucleophilic fluoride ions and a proton donor to facilitate ring opening. The reaction mechanism proceeds through a sequential pathway where the strained three-membered epoxide ring is first attacked by the fluoride species, leading to a ring-opened intermediate containing both hydroxyl and fluorine functionalities. Subsequently, under the thermal conditions provided (50-200°C), the hydroxyl group undergoes a substitution reaction with additional HF species, resulting in the elimination of water and the installation of a second fluorine atom to form the stable gem-difluoro or polyfluoroalkane structure. 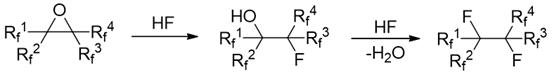 This mechanistic pathway is highly advantageous because the amine component buffers the acidity of the HF, preventing the harsh acidic conditions that typically lead to polymerization or decomposition of sensitive fluorinated skeletons. For technical teams, understanding this buffered mechanism is crucial for optimizing reaction parameters to maximize the yield of the target fluoroalkane while minimizing oligomerization.
This mechanistic pathway is highly advantageous because the amine component buffers the acidity of the HF, preventing the harsh acidic conditions that typically lead to polymerization or decomposition of sensitive fluorinated skeletons. For technical teams, understanding this buffered mechanism is crucial for optimizing reaction parameters to maximize the yield of the target fluoroalkane while minimizing oligomerization.
From an impurity control perspective, this mechanism offers superior selectivity compared to radical-based fluorination methods. Because the reaction proceeds via ionic nucleophilic substitution rather than radical abstraction, there is a markedly lower tendency for C-C bond cleavage or random fluorination at unactivated positions. The use of aprotic solvents further stabilizes the fluoride anion, enhancing its nucleophilicity without promoting side reactions associated with protic media. This precise control over the reaction trajectory ensures that the final crude product contains significantly fewer isomeric impurities, reducing the burden on downstream purification steps such as rectification. Consequently, the process supports the production of high-purity fluoroalkane compounds that meet the stringent specifications required for pharmaceutical and electronic applications, where trace impurities can be detrimental to performance.
How to Synthesize Fluoroalkane Compounds Efficiently
The practical implementation of this synthesis route is designed for straightforward scalability, requiring standard chemical processing equipment rather than exotic reactors. The protocol involves charging the reactor with the chosen solvent system, followed by the sequential addition of the amine hydrogen fluoride salt and the fluorinated epoxide substrate. Once the mixture is homogenized, the system is sealed and heated to the target temperature, typically maintained for a duration of approximately 24 hours to ensure complete conversion. Detailed standardized synthetic steps, including specific molar ratios and workup procedures, are outlined in the technical guide below.
- Charge a dry reaction vessel with aprotic solvent or organic amine, amine hydrogen fluoride salt, and fluorinated epoxide.
- Seal the vessel, stir, and heat the mixture to 50-200°C for 24 hours to facilitate ring-opening and fluorination.
- Cool the reaction, wash with sodium carbonate solution and water, separate the organic phase, and purify via rectification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this fluorinated epoxide route translates into tangible strategic benefits beyond mere technical feasibility. The elimination of elemental fluorine gas from the supply chain removes a major bottleneck associated with hazardous material logistics and regulatory compliance. By utilizing stable amine-HF salts, facilities can operate with reduced insurance premiums and lower capital expenditure on safety infrastructure, directly contributing to substantial cost savings in the overall manufacturing budget. Furthermore, the robustness of the reaction conditions allows for flexible sourcing of raw materials, as the process tolerates a range of commercially available solvents and amine salts, mitigating the risk of supply disruptions.
- Cost Reduction in Manufacturing: The transition from gas-phase fluorination to this liquid-phase amine-HF mediated process drastically simplifies the reactor design and operational requirements. By avoiding the need for nickel-lined reactors or specialized electrochemical cells, manufacturers can utilize standard stainless steel equipment, significantly lowering capital investment. Additionally, the high reaction selectivity minimizes the loss of valuable fluorinated starting materials to by-products, ensuring that a greater proportion of the input mass is converted into saleable product, which optimizes the cost per kilogram of the final fluoroalkane.
- Enhanced Supply Chain Reliability: Dependence on elemental fluorine gas often ties production to a limited number of specialized suppliers with complex distribution networks. In contrast, amine hydrogen fluoride salts and common organic solvents are widely available commodity chemicals with stable global supply chains. This shift enhances supply continuity and reduces lead time for high-purity fluoroalkanes, as manufacturers are no longer vulnerable to the logistical constraints and safety embargoes often associated with toxic gas transport.
- Scalability and Environmental Compliance: The process generates fewer hazardous by-products and avoids the creation of heavy metal waste streams common in electrochemical methods. The workup procedure involves simple aqueous washing and distillation, which are easily scalable from pilot plant to commercial tonnage without exponential increases in waste treatment costs. This environmental friendliness aligns with increasingly strict global regulations on fluorinated emissions, future-proofing the manufacturing asset against potential regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners.
Q: Why is this method safer than traditional fluorine gas fluorination?
A: Traditional fluorine gas fluorination involves highly dangerous F2 gas which poses severe safety risks and often leads to poor selectivity due to side reactions with other heteroatoms. This patented method utilizes stable amine hydrogen fluoride salts, eliminating the need for handling elemental fluorine gas and significantly improving reaction selectivity and operational safety.
Q: What yields can be expected from this fluorinated epoxide route?
A: According to the experimental data in patent CN114507115A, this process consistently achieves reaction yields of over 90 percent. For instance, specific examples demonstrated yields ranging from 88.4% to 91.6%, indicating a highly efficient conversion suitable for industrial scale-up.
Q: What solvents are compatible with this fluorination process?
A: The process is versatile regarding solvent choice, supporting both aprotic solvents such as glycol dimethyl ether, acetonitrile, and sulfolane, as well as organic amine solvents like triethylamine and tributylamine. This flexibility allows manufacturers to optimize cost and solubility parameters for specific substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this epoxide-based fluorination technology for the next generation of high-performance materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of fluoroalkane intermediate meets the exacting standards required for pharmaceutical and electronic grade applications.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this cost-effective route into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your target molecules. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your project requirements.
