Advanced Synthesis Strategy for Flucloxacillin Sodium: Enhancing Purity and Commercial Scalability
Advanced Synthesis Strategy for Flucloxacillin Sodium: Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for semi-synthetic penicillins to ensure consistent supply and high quality standards. Patent CN102964356A introduces a refined methodology for the production of Flucloxacillin Sodium, a critical beta-lactam antibiotic known for its resistance to penicillinase. This technical disclosure addresses longstanding challenges in the acylation and purification stages that have historically plagued manufacturers. By optimizing the activation of the side chain acid and implementing a novel crystallization protocol for the intermediate acid, the process achieves superior impurity profiles and operational simplicity. This report analyzes the technical merits of this approach and its implications for global supply chains seeking reliable antibiotic intermediate suppliers. The structural integrity of the final product is paramount for therapeutic efficacy, and the chemical architecture is defined by the specific isoxazole side chain attached to the penicillin nucleus.
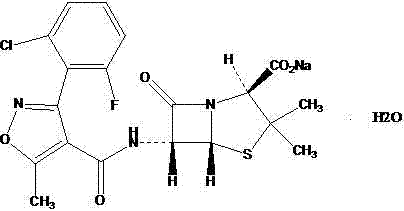
Flucloxacillin Sodium serves as a vital component in treating infections caused by penicillinase-producing Staphylococcus aureus. The chemical stability and bioavailability of this molecule are directly correlated with the precision of its synthesis. Traditional methods often struggle with the purification of the free acid intermediate, leading to variable quality in the final salt form. The disclosed technology leverages a controlled acylation environment followed by a dedicated crystallization step that significantly enhances the physical properties of the intermediate. For procurement managers and technical directors, understanding these mechanistic improvements is essential for evaluating potential manufacturing partners. The ability to consistently produce high-purity material reduces downstream processing burdens and ensures regulatory compliance in stringent markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Flucloxacillin Sodium has been hindered by complex purification requirements and suboptimal yield profiles. Prior art, such as the method described in CN101475578A, relies on the formation of a dibenzylethylenediamine salt followed by cationic resin exchange. This multi-step ion exchange process is not only operationally cumbersome but also introduces significant risks of contamination from the resin matrix itself. Furthermore, the yield in these traditional routes is often compromised by the inefficiency of the salt conversion steps. Another existing approach, detailed in CN102702227A, attempts to simplify the process via a one-pot reaction but fails to adequately purify the Flucloxacillin Acid intermediate before salt formation. This omission results in high levels of residual solvents and impurities carrying over into the final product, complicating crystallization and reducing overall stability. These legacy methods impose unnecessary costs and quality control burdens on the supply chain.
The Novel Approach
The methodology outlined in CN102964356A represents a strategic departure from these inefficient legacy processes by decoupling the acylation and salt formation steps with a dedicated purification phase. Instead of proceeding directly to salt formation from a crude reaction mixture, this novel approach isolates the Flucloxacillin Acid through a precise crystallization technique using an alcohol-water solvent system. This step is critical as it allows for the rejection of unreacted starting materials and side products before the final salting step. By avoiding the use of cationic resins and complex salt exchange mechanisms, the process flow is drastically simplified. The result is a more linear and controllable manufacturing sequence that minimizes unit operations while maximizing the recovery of high-quality material. This streamlined workflow translates directly into enhanced process reliability and reduced operational complexity for industrial scale-up.
Mechanistic Insights into POCl3-Mediated Acylation and Crystallization
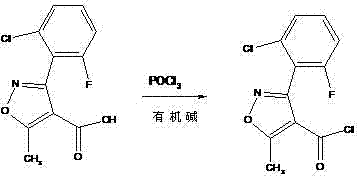
The core of this synthetic advancement lies in the efficient activation of the carboxylic acid side chain and the subsequent coupling with 6-aminopenicillanic acid (6-APA). The process utilizes phosphorus oxychloride (POCl3) in the presence of an organic amine catalyst, such as DMF or DMAc, to generate the reactive acid chloride intermediate in situ. This activation method is superior to alternatives using thionyl chloride or triphosgene due to its high conversion rates and manageable reaction exotherms. The reaction conditions are maintained at mild temperatures, typically between 0 to 50 degrees Celsius, which preserves the integrity of the sensitive isoxazole ring. Following activation, the acid chloride is coupled with 6-APA in an aqueous alkaline medium. The precise control of pH and temperature during this acylation step is crucial to prevent hydrolysis of the beta-lactam ring while ensuring complete conversion of the amine. The mechanistic pathway ensures that the amide bond is formed with high regioselectivity.
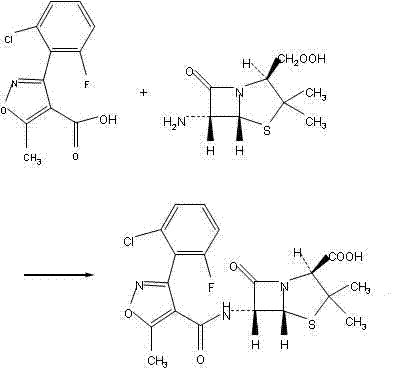
Following the acylation, the isolation of Flucloxacillin Acid is achieved through a sophisticated crystallization protocol that addresses the historical difficulty of crystallizing this intermediate. The patent specifies the use of an alcohol solvent, such as methanol or ethanol, mixed with water in specific ratios to induce crystallization. This solvent system is engineered to maximize the solubility difference between the desired product and impurities. By slowly adding water to the alcoholic solution of the crude acid at low temperatures, the product precipitates in a highly pure crystalline form. This step effectively removes residual organic solvents and inorganic salts that would otherwise contaminate the final API. The subsequent conversion to the sodium salt using sodium isooctanoate is then performed on this purified acid, ensuring that the final Flucloxacillin Sodium monohydrate meets stringent purity specifications with total impurities well below 0.5 percent.
How to Synthesize Flucloxacillin Sodium Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and temperature control to replicate the high yields reported in the patent data. The process begins with the activation of the side chain acid, followed by the coupling reaction with the penicillin nucleus, and concludes with the purification and salting steps. Each stage is designed to minimize waste and maximize throughput, making it an ideal candidate for commercial manufacturing environments. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature profiles, are critical for achieving the reported purity levels of over 99 percent. Operators must adhere strictly to the crystallization parameters to ensure the correct crystal form is obtained, as this impacts the downstream processing and stability of the final drug product.
- Activate 3-(2-chloro-6-fluorophenyl)-5-methylisoxazole-4-carboxylic acid using phosphorus oxychloride and organic amine to form the acid chloride intermediate.
- React the acid chloride with 6-APA in aqueous alkaline conditions, followed by acidification and extraction to isolate Flucloxacillin Acid.
- Crystallize Flucloxacillin Acid using an alcohol-water system, then react with sodium isooctanoate to form the final Flucloxacillin Sodium monohydrate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this optimized synthesis route offers substantial benefits for procurement managers and supply chain directors focused on cost efficiency and reliability. The elimination of the cationic resin exchange step, which is prevalent in older methods, removes a significant cost center associated with resin purchase, regeneration, and disposal. This simplification of the process flow reduces the overall cycle time and minimizes the consumption of auxiliary materials. Furthermore, the improved yield and purity reduce the need for reprocessing or rework, leading to more predictable production schedules. For organizations seeking cost reduction in antibiotic manufacturing, this technology provides a clear pathway to lower unit costs without compromising on quality standards. The robustness of the crystallization step also ensures a more consistent supply of the intermediate, mitigating risks associated with batch-to-batch variability.
- Cost Reduction in Manufacturing: The removal of complex ion-exchange resin steps significantly lowers the operational expenditure associated with consumables and waste management. By streamlining the synthesis to fewer unit operations, the process reduces labor hours and energy consumption per kilogram of product. The high conversion efficiency of the acid chloride formation minimizes the loss of expensive starting materials like 6-APA. These factors combine to create a more economically viable production model that enhances margin potential for manufacturers. The qualitative reduction in process complexity directly correlates with lower overhead costs and improved asset utilization rates in the production facility.
- Enhanced Supply Chain Reliability: The use of readily available reagents such as phosphorus oxychloride and common organic solvents ensures that the supply chain is not dependent on specialized or scarce materials. The robustness of the crystallization process reduces the likelihood of batch failures, ensuring a more consistent output of qualified material. This reliability is crucial for maintaining continuous supply to downstream formulation partners who depend on timely deliveries. By mitigating the risks associated with difficult purification steps, manufacturers can offer more stable lead times and commit to larger volume contracts with greater confidence. The process stability supports a resilient supply chain capable of withstanding market fluctuations.
- Scalability and Environmental Compliance: The simplified workflow facilitates easier scale-up from pilot to commercial production volumes without requiring specialized equipment. The reduction in solvent usage and the elimination of resin waste contribute to a lower environmental footprint, aligning with modern green chemistry principles. Efficient solvent recovery systems can be integrated more easily into this streamlined process, further reducing waste discharge. The high purity of the intermediate reduces the burden on wastewater treatment systems by lowering the load of organic impurities. These environmental advantages support regulatory compliance and enhance the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Flucloxacillin Sodium. These insights are derived from the specific technical advantages disclosed in the patent literature and are intended to clarify the value proposition for potential partners. Understanding the nuances of the crystallization and acylation steps is key to appreciating the quality improvements offered by this method. Stakeholders are encouraged to review these details when evaluating manufacturing capabilities and quality assurance protocols.
Q: How does the new crystallization method improve Flucloxacillin Acid quality?
A: The optimized process utilizes a specific alcohol-water solvent system for crystallization, which effectively removes impurities and residual solvents that are common in one-pot methods, resulting in significantly higher purity and better crystal form.
Q: What are the advantages of avoiding cationic resin exchange in this synthesis?
A: Eliminating the cationic resin exchange step simplifies the operational workflow, reduces the risk of introducing new impurities from the resin, and lowers overall production costs by removing the need for resin regeneration and replacement.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method uses readily available reagents and mild reaction conditions, avoiding complex equipment requirements. The high conversion rates and simplified purification steps make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flucloxacillin Sodium Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, leveraging advanced synthetic technologies like the one described in CN102964356A to deliver superior products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Flucloxacillin Sodium meets the highest global standards. Our commitment to process optimization allows us to offer high-purity Flucloxacillin Sodium that supports the production of safe and effective antibiotic formulations. We understand the critical nature of supply continuity in the pharmaceutical sector and have built our operations to prioritize reliability and quality.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain requirements. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product portfolio. By partnering with our technical procurement team, you gain access to a wealth of expertise in semi-synthetic penicillin production. We are dedicated to supporting your growth with reliable supply solutions and continuous process improvements. Contact us today to discuss how we can collaborate to optimize your antibiotic supply chain and achieve your commercial objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
