Advanced Green Synthesis of Daidzein for Scalable Pharmaceutical Manufacturing
Advanced Green Synthesis of Daidzein for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for producing high-value bioactive compounds like daidzein. Patent CN113896703A introduces a groundbreaking green preparation process that fundamentally redefines the synthetic landscape for this critical isoflavone. By shifting away from traditional, energy-intensive methodologies, this innovation leverages a sophisticated catalytic system involving boron trifluoride diethyl etherate and phosphorus pentoxide to achieve unprecedented efficiency. The technical breakthrough lies not merely in incremental improvements but in a holistic redesign of the reaction thermodynamics and kinetics, resulting in a total yield surge from historical lows of approximately 25% to an industrial-grade benchmark exceeding 88%. For R&D directors and procurement strategists, this patent represents a pivotal opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity daidzein with drastically reduced environmental footprints and operational costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of daidzein has been plagued by severe inefficiencies and hazardous operating conditions that hinder large-scale commercial viability. One prevalent prior art method relies on zinc chloride as a catalyst for the condensation of resorcinol and p-hydroxyphenylacetic acid, requiring extreme temperatures ranging from 135°C to 160°C. As illustrated in the reaction scheme below, this thermal stress not only consumes excessive energy but also leads to significant side reactions and product degradation, capping the first-step yield at a mere 50%.
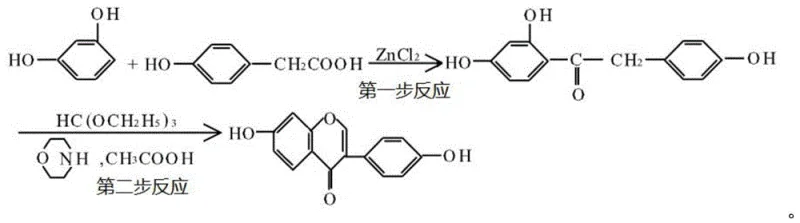
Furthermore, the subsequent cyclization step in these legacy processes often employs triethyl orthoformate and morpholine under boiling conditions, which further erodes the overall yield to a cumulative total of only roughly 25%. Another problematic conventional approach utilizes aluminum trichloride promoters alongside boron trifluoride, which suffers from significant catalyst loss due to hydrolysis. Additionally, some existing routes depend on highly toxic reagents like methylsulfonyl chloride for cyclization, creating substantial regulatory hurdles and waste disposal challenges that inflate the cost reduction in API manufacturing efforts. These combined factors render traditional methods economically unviable for modern, compliance-driven supply chains.
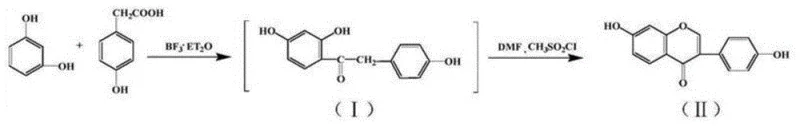
The Novel Approach
In stark contrast, the novel green preparation process detailed in the patent data introduces a mild, controllable, and highly efficient two-step synthesis strategy. The core innovation involves the strategic addition of phosphorus pentoxide (P2O5) as a potent dehydrating agent during the initial condensation phase. This modification effectively scavenges water generated during the reaction, thereby preventing the hydrolysis and deactivation of the expensive boron trifluoride diethyl etherate catalyst. Consequently, the reaction can proceed at a significantly lower temperature range of 75°C to 80°C, achieving a first-step yield of over 95%. The second step replaces toxic sulfonyl chlorides with phosphorus oxychloride in a diethylformamide (DEF) solvent system, facilitating cyclization at ambient temperatures between 20°C and 30°C. This paradigm shift ensures that the commercial scale-up of complex pharmaceutical intermediates becomes not only feasible but highly profitable.
Mechanistic Insights into BF3/P2O5 Catalyzed Condensation and Cyclization
The mechanistic superiority of this new route stems from the synergistic interaction between the Lewis acid catalyst and the dehydrating agent. In the absence of phosphorus pentoxide, the water byproduct from the condensation of resorcinol and p-hydroxyphenylacetic acid would rapidly hydrolyze the boron trifluoride etherate, rendering it inactive and necessitating large excesses of the catalyst. By integrating P2O5, the reaction equilibrium is driven forward according to Le Chatelier's principle, ensuring near-quantitative conversion of raw materials into the key intermediate, 4,6-dihydroxybenzene-4'-hydroxyacetophenone. This precise control over the reaction environment minimizes the formation of polymeric byproducts and tars, which are common impurities in high-temperature Friedel-Crafts acylations. The result is a crude product of such high quality that downstream purification burdens are significantly alleviated, directly impacting the purity profile of the final high-purity OLED material or pharmaceutical grade daidzein.
Regarding the cyclization mechanism, the substitution of methylsulfonyl chloride with phosphorus oxychloride (POCl3) offers distinct kinetic and safety advantages. POCl3 acts as both a chlorinating and dehydrating agent, activating the carbonyl group of the intermediate for intramolecular nucleophilic attack by the phenolic hydroxyl group. This reaction proceeds smoothly at 20-30°C, avoiding the thermal degradation pathways associated with higher temperature protocols. The use of DEF as a solvent further stabilizes the transition state and facilitates the dissolution of polar intermediates. Crucially, the mild conditions prevent the racemization or decomposition of sensitive functional groups, ensuring that the final daidzein product maintains a purity specification exceeding 98%, which is critical for meeting stringent pharmacopoeial standards.
How to Synthesize Daidzein Efficiently
Implementing this green synthesis route requires precise adherence to the molar ratios and thermal profiles established in the patent examples to maximize yield and minimize waste. The process begins with the careful charging of resorcinol, p-hydroxyphenylacetic acid, and the catalytic system into a reactor equipped for reduced pressure operations. Operators must monitor the temperature closely to maintain the 75-80°C window, ensuring complete melting and reaction without thermal runaway. Following the condensation, the recovery of the catalyst via vacuum concentration is a critical step for economic viability. The subsequent cyclization demands strict moisture control to prevent POCl3 decomposition, followed by a straightforward ethanol recrystallization to achieve final purity. For detailed standard operating procedures and specific equipment requirements, please refer to the technical guide below.
- Condense resorcinol and p-hydroxyphenylacetic acid using boron trifluoride diethyl etherate and phosphorus pentoxide at 75-80°C.
- Recover the catalyst via reduced pressure concentration to obtain the intermediate 4,6-dihydroxybenzene-4'-hydroxyacetophenone.
- Perform cyclization with phosphorus oxychloride in DEF solvent at 20-30°C, followed by ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented green process offers transformative benefits that extend far beyond simple yield improvements. The elimination of high-temperature steps and toxic reagents fundamentally alters the cost structure of daidzein manufacturing, making it a highly attractive candidate for long-term sourcing contracts. By reducing the complexity of waste treatment and lowering energy consumption, manufacturers can pass significant savings down the supply chain, enhancing the competitiveness of the final therapeutic or agricultural products. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the risk of supply disruptions caused by failed production runs or off-spec material.
- Cost Reduction in Manufacturing: The integration of phosphorus pentoxide allows for the recycling of the boron trifluoride catalyst, which is typically a major cost driver in Lewis acid-catalyzed reactions. By preventing catalyst loss through hydrolysis, the process eliminates the need for continuous fresh catalyst charging, leading to substantial raw material cost savings. Additionally, the ability to recover and recycle the DEF solvent through reduced pressure concentration further drives down operational expenditures. The removal of expensive and hazardous reagents like methylsulfonyl chloride also reduces the financial burden associated with specialized storage, handling, and neutralization of toxic waste streams.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade raw materials such as resorcinol and p-hydroxyphenylacetic acid ensures a stable supply base that is not subject to the volatility of niche reagent markets. The mild reaction conditions (20-80°C) reduce the dependency on specialized high-temperature reactors or cryogenic cooling systems, allowing for production across a wider range of manufacturing facilities. This flexibility enhances supply chain resilience, enabling rapid scale-up to meet surging market demand for reducing lead time for high-purity pharmaceutical intermediates without compromising on safety or quality standards.
- Scalability and Environmental Compliance: The drastic reduction in "three wastes" (waste gas, waste water, and waste solids) aligns perfectly with increasingly stringent global environmental regulations. The process generates minimal hazardous byproducts, simplifying the permitting process for new production lines and reducing the long-term liability associated with environmental remediation. The low-temperature cyclization step inherently lowers the risk of thermal runaway incidents, making the process safer to operate at the multi-ton scale. This combination of safety and sustainability makes the technology ideal for commercial scale-up of complex flavonoid derivatives in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green daidzein synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the operational realities. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: How does the new process improve yield compared to traditional zinc chloride methods?
A: Traditional methods using zinc chloride often suffer from yields as low as 25% due to harsh conditions (135-160°C). The new process utilizes phosphorus pentoxide as a dehydrating agent, pushing total yields to over 88% by preventing catalyst hydrolysis.
Q: What safety advantages does phosphorus oxychloride offer over methylsulfonyl chloride?
A: Phosphorus oxychloride replaces highly toxic methylsulfonyl chloride in the cyclization step, significantly reducing occupational health risks and simplifying waste treatment protocols while maintaining reaction efficiency at lower temperatures.
Q: Is the boron trifluoride catalyst recoverable in this green process?
A: Yes, the addition of phosphorus pentoxide prevents the loss of boron trifluoride diethyl etherate by removing generated water, allowing the catalyst to be recovered via reduced pressure concentration for reuse.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Daidzein Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this green synthesis route to revolutionize the availability of high-quality daidzein for the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing environments. Our state-of-the-art facilities are equipped to handle the specific requirements of Lewis acid catalysis and solvent recovery, guaranteeing stringent purity specifications and rigorous QC labs oversight for every batch produced. We are committed to delivering a reliable daidzein supplier experience that combines technical excellence with unwavering supply security.
We invite forward-thinking partners to collaborate with us on optimizing their supply chains through this advanced technology. By leveraging our expertise, you can achieve significant Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific rigor and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
