Advanced CDI-Mediated Synthesis of Iguratimod Intermediates for Scalable Pharmaceutical Manufacturing
Advanced CDI-Mediated Synthesis of Iguratimod Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking robust, safe, and efficient synthetic routes for critical active pharmaceutical ingredient (API) intermediates. A significant breakthrough in this domain is detailed in patent CN112209859B, which discloses a novel preparation method for an important Iguratimod intermediate. This technology shifts the paradigm from hazardous, heterogeneous reactions to a streamlined active ester methodology utilizing N,N-Carbonyldiimidazole (CDI). For global procurement and R&D teams, this represents a pivotal opportunity to enhance supply chain reliability while adhering to stricter environmental and safety standards. The core innovation lies in the activation of formic acid to form a highly reactive formyl imidazole species, which subsequently couples with the amine precursor under mild conditions. This approach not only mitigates the risks associated with traditional acylation agents but also delivers exceptional chemical purity, addressing the rigorous quality demands of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iguratimod intermediates has relied on chemically aggressive and operationally complex pathways that pose significant challenges for industrial scalability. One prevalent conventional route involves the use of pivaloyl chloride and sodium formate to generate a mixed anhydride in situ. As illustrated in the reaction scheme below, this method suffers from inherent heterogeneity because sodium formate is insoluble in common organic solvents like acetone. This solid-liquid interface severely limits the reaction rate and efficiency. Furthermore, the sodium chloride byproduct generated during the reaction tends to coat the surface of the unreacted sodium formate, creating a passivation layer that halts further conversion. This often leads to incomplete reactions and the formation of difficult-to-remove impurities, such as pivaloyl amide, which compromises the final product quality and necessitates costly purification steps.
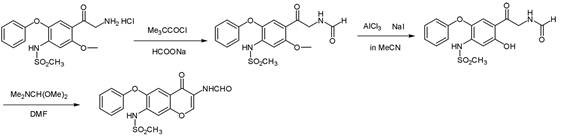
Alternative historical routes have attempted to bypass these issues but introduced even greater safety liabilities. For instance, certain synthetic strategies employ bromination followed by azidation using sodium azide. While chemically feasible, sodium azide is notoriously unstable and poses a severe explosion hazard, making it unsuitable for large-scale manufacturing environments. Additionally, the use of elemental bromine requires specialized corrosion-resistant equipment and rigorous containment protocols due to its high toxicity and volatility. These factors collectively increase the capital expenditure and operational risk profile for manufacturers, rendering such methods economically unviable for reliable long-term supply chains.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a homogeneous active ester strategy that fundamentally resolves the kinetic and safety bottlenecks. By reacting formic acid with CDI, the process generates formyl imidazole, a potent acylating agent that remains soluble in the reaction medium. This ensures uniform mixing and rapid reaction kinetics without the mass transfer limitations seen in solid-liquid systems. The subsequent coupling with the amine hydrochloride salt proceeds smoothly at ambient temperatures, typically between 15°C and 25°C. This mild thermal profile drastically reduces energy consumption compared to processes requiring reflux or cryogenic cooling. Moreover, the byproducts of the CDI activation are imidazole and carbon dioxide, which are easily removed during workup, leading to a cleaner crude product profile and simplifying downstream processing.
Mechanistic Insights into CDI-Mediated Active Ester Formation
The success of this synthetic route hinges on the unique reactivity of N,N-Carbonyldiimidazole (CDI) as a dehydrating coupling agent. Mechanistically, the nucleophilic nitrogen of the imidazole ring attacks the carbonyl carbon of formic acid, displacing a hydroxyl group and forming an O-acylimidazole intermediate which rapidly rearranges or reacts to form the stable N-formylimidazole species. This activated ester possesses a highly electrophilic carbonyl carbon, making it exceptionally susceptible to nucleophilic attack by the amino group of the substrate. Unlike acid chlorides which release corrosive HCl gas immediately upon hydrolysis or reaction, the CDI-derived intermediate releases imidazole, a weak base that can be easily neutralized or washed away. This mechanistic pathway avoids the generation of strong acids that could degrade sensitive functional groups on the aromatic ring, such as the methanesulfonamide moiety, thereby preserving the structural integrity of the molecule throughout the synthesis.
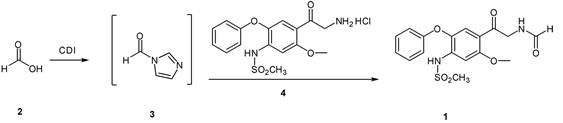
From an impurity control perspective, this mechanism offers distinct advantages over mixed anhydride routes. In traditional methods, the competition between the desired amine and trace moisture or other nucleophiles for the mixed anhydride often leads to hydrolysis products or symmetrical anhydrides. The CDI-activated formyl species is sufficiently reactive to preferentially acylate the primary amine even in the presence of minor impurities, yet stable enough to be handled without immediate decomposition. The patent data indicates that optimizing the molar ratio of CDI to formic acid and the amine substrate is critical; a ratio of approximately 1.5:1:1 ensures complete conversion of the starting materials while minimizing excess reagent waste. This precise stoichiometric control, combined with the clean reaction profile, results in a final product purity exceeding 99.95%, significantly reducing the burden on analytical quality control laboratories.
How to Synthesize Formamidomethyl-2-methoxy-4-methanesulfonamide-5-phenoxyphenyl ketone Efficiently
Implementing this advanced synthesis protocol requires careful attention to solvent selection and temperature control to maximize the benefits of the CDI activation. The process is designed to be operationally simple, utilizing common laboratory glassware and standard workup procedures that translate seamlessly to pilot and production scales. Dichloromethane is identified as the preferred solvent due to its ability to dissolve both the organic substrates and the CDI reagent effectively, ensuring a truly homogeneous reaction environment. The following guide outlines the standardized procedure derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate these high-yield results in their own facilities.
- Dissolve formic acid in dichloromethane and react with N,N-Carbonyldiimidazole (CDI) at 15-25°C to generate the active formyl imidazole intermediate.
- Add 2-amino-1-(2-methoxy-4-methanesulfonamido-5-phenoxyphenyl) ethanone hydrochloride to the reaction mixture and stir for 1-2 hours.
- Quench the reaction with water, separate the organic phase, concentrate, and recrystallize from isopropanol to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this CDI-based methodology offers substantial strategic benefits beyond mere chemical yield. The elimination of hazardous reagents like sodium azide and pivaloyl chloride directly translates to reduced regulatory compliance costs and lower insurance premiums associated with handling dangerous goods. By adopting a process that relies on stable, commercially available reagents, companies can mitigate the risk of supply disruptions caused by the strict transportation regulations governing explosive or highly corrosive chemicals. This stability ensures a more predictable lead time for raw material acquisition, allowing for smoother production planning and inventory management.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the reaction workflow and the reduction of waste treatment expenses. Since the reaction proceeds homogeneously without the need for phase transfer catalysts or vigorous stirring to suspend solids, the energy demand for agitation and heating is significantly lowered. Furthermore, the high selectivity of the CDI coupling minimizes the formation of side products like pivaloyl amide, which reduces the volume of solvent and adsorbents required for purification. This efficiency gain leads to substantial cost savings in raw material consumption and waste disposal, enhancing the overall margin profile for the intermediate.
- Enhanced Supply Chain Reliability: Sourcing chemicals that do not require special hazardous material permits simplifies the logistics network. The reagents used in this novel route, such as formic acid and CDI, are widely produced commodity chemicals with robust global supply chains. This abundance reduces the vulnerability to single-source supplier failures or geopolitical trade restrictions that often affect specialty reagents like sodium azide. Consequently, manufacturers can maintain higher safety stock levels with lower holding costs, ensuring continuous operation even during market fluctuations.
- Scalability and Environmental Compliance: The mild reaction conditions (15-25°C) eliminate the need for complex cryogenic cooling systems or high-pressure reactors, making the process inherently safer and easier to scale from kilogram to tonne quantities. From an environmental standpoint, the absence of heavy metals and the generation of benign byproducts align with green chemistry principles, facilitating easier permitting and reducing the carbon footprint of the manufacturing site. This alignment with sustainability goals is increasingly critical for maintaining partnerships with major pharmaceutical clients who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the viability of technology transfer.
Q: Why is the CDI active ester method superior to the traditional mixed anhydride route?
A: The CDI method avoids the use of pivaloyl chloride, which creates heterogeneous reaction conditions and corrosive byproducts. It ensures a homogeneous reaction system with higher yields (up to 91.7%) and eliminates the risk of pivaloyl amide byproduct formation.
Q: What are the safety advantages of this new synthesis route?
A: Unlike alternative routes that utilize explosive sodium azide or highly active bromine, this method operates under mild conditions (15-25°C) using stable reagents, significantly reducing industrial safety risks and equipment corrosion.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process features simple operation, low energy consumption due to ambient temperature reactions, and excellent product purity (99.95%), making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iguratimod Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative CDI-based route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available.
