Advanced Synthesis of Hydroxypyrone Intermediates for Commercial Scale Antiviral Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical antiviral intermediates, particularly those serving high-demand medications like Xofluza (Baloxavir Marvoxil). Patent CN111320598A, published on June 23, 2020, introduces a groundbreaking preparation method for 3-hydroxy-2-(hydroxymethyl)-4H-pyran-4-one, a pivotal building block in the synthesis of this cap-dependent endonuclease inhibitor. This innovation addresses long-standing supply chain vulnerabilities by replacing expensive and difficult-to-purify precursors with accessible carbohydrate derivatives, thereby establishing a new benchmark for reliability in the production of complex heterocyclic scaffolds. The disclosed methodology not only enhances product purity but also streamlines the operational workflow, making it an attractive option for contract development and manufacturing organizations aiming to secure the global supply of next-generation influenza treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key pyranone intermediates has relied heavily on the use of pyroconic acid as a starting material, a pathway fraught with significant technical and economic inefficiencies. As illustrated in the prior art, this traditional approach often encounters severe hurdles regarding the purification of the raw pyroconic acid, which directly translates to poor reaction reproducibility and inconsistent batch quality. The inherent instability and high cost of these specialized starting materials create a bottleneck for manufacturers, rendering the process unsuitable for the rigorous demands of industrial-scale production. Furthermore, the harsh conditions often required to manipulate these unstable precursors can lead to the formation of complex impurity profiles, necessitating costly and time-consuming downstream purification processes that erode overall profit margins.
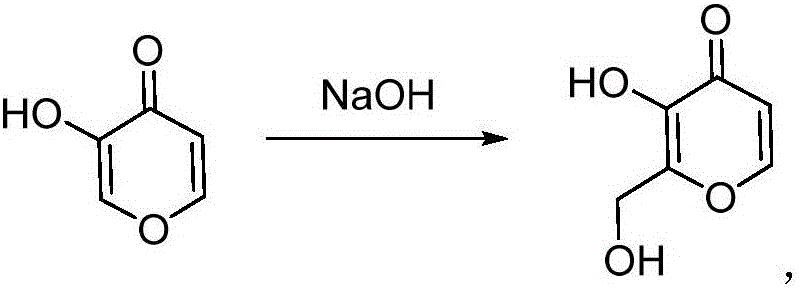
The Novel Approach
In stark contrast, the novel methodology presented in CN111320598A leverages a carbohydrate-based strategy that fundamentally reimagines the construction of the pyranone core. By initiating the synthesis from stable, protected sugar derivatives, the process bypasses the volatility associated with pyroconic acid, offering a much more controlled and predictable reaction environment. This strategic shift allows for the implementation of mild reaction conditions and standard work-up procedures, such as simple filtration and distillation, which are highly compatible with large-scale manufacturing infrastructure. The result is a synthetic route that not only delivers the target molecule with exceptional purity but also significantly reduces the complexity of the supply chain by utilizing commoditized starting materials that are readily available from global chemical suppliers.
Mechanistic Insights into Carbohydrate-Derived Pyranone Synthesis
The core of this innovative synthesis lies in a meticulously orchestrated four-step sequence that transforms a protected hexose derivative into the desired hydroxypyrone scaffold through protection, acylation, oxidation, and hydrolysis. The process begins with the formation of an acetal protecting group using dimethoxypropane and a catalytic amount of p-toluenesulfonic acid, which selectively masks specific hydroxyl functionalities to direct subsequent reactivity. Following this, a selective acylation step introduces a benzoyl group to the primary alcohol, setting the stage for the critical oxidation event. The use of 2-iodoxybenzoic acid (IBX) as the oxidant is particularly noteworthy, as it facilitates the conversion of the secondary alcohol to a ketone under relatively mild thermal conditions, typically between 40°C and 90°C, minimizing the risk of over-oxidation or degradation of the sensitive sugar backbone.
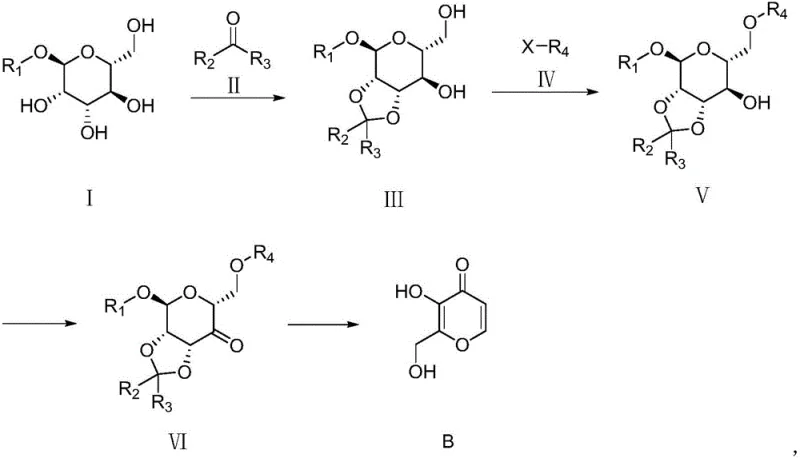
Impurity control is inherently built into this mechanistic pathway through the strategic use of crystallization and precipitation steps at various stages of the synthesis. For instance, the intermediate compounds formed after acylation and oxidation can be isolated via filtration or solvent exchange, effectively removing soluble byproducts and unreacted reagents before they can interfere with downstream transformations. The final acidic hydrolysis step not only removes the protecting groups but also induces the necessary rearrangement to form the aromatic pyranone ring, a transformation that is driven thermodynamically to favor the desired product. This cascade of reactions ensures that the final API intermediate meets stringent purity specifications, often exceeding 97%, without the need for elaborate chromatographic separations that are impractical at a commercial scale.
How to Synthesize 3-Hydroxy-2-(hydroxymethyl)-4H-pyran-4-one Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to maximize yield and minimize side reactions, particularly during the oxidation and hydrolysis phases. Operators must ensure that the acetal protection is complete before proceeding to acylation to prevent regioisomer formation, and the oxidation step benefits from careful monitoring of the IBX consumption to avoid residual oxidant in the final product. The detailed standardized synthetic steps, including specific solvent volumes, reaction times, and isolation techniques described in the patent examples, provide a robust framework for technology transfer and process validation.
- Protect the diol system of the starting sugar derivative using dimethoxypropane and p-toluenesulfonic acid to form the acetal intermediate.
- Perform selective acylation of the primary hydroxyl group using benzoyl chloride and a base such as triethylamine.
- Oxidize the secondary alcohol to a ketone using 2-iodoxybenzoic acid (IBX) in a suitable organic solvent.
- Execute final deprotection and rearrangement via acidic hydrolysis to yield the target hydroxypyrone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this carbohydrate-based synthesis represents a strategic opportunity to mitigate risk and optimize cost structures within the antiviral manufacturing sector. By eliminating the dependency on scarce and expensive pyroconic acid, manufacturers can leverage the abundant global supply of sugar derivatives, which are produced at massive scales for the food and fermentation industries, ensuring a stable and continuous flow of raw materials. This shift not only insulates the supply chain from price volatility associated with niche fine chemicals but also simplifies vendor qualification processes, as the starting materials are widely sourced from established chemical producers with proven quality systems.
- Cost Reduction in Manufacturing: The economic benefits of this new route are driven primarily by the substitution of high-cost specialty reagents with low-cost commodity chemicals, drastically lowering the bill of materials for each production batch. Additionally, the simplified work-up procedures, which rely on filtration and distillation rather than complex chromatography, reduce solvent consumption and labor hours, leading to substantial operational expenditure savings. The high yields reported in the patent examples further contribute to cost efficiency by maximizing the output per unit of input, effectively reducing the waste disposal costs associated with low-yielding processes.
- Enhanced Supply Chain Reliability: Utilizing widely available carbohydrate starting materials significantly de-risks the supply chain against disruptions that often plague the sourcing of exotic heterocyclic precursors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, allows for greater flexibility in manufacturing scheduling and reduces the likelihood of batch failures due to minor process deviations. This reliability ensures that downstream API manufacturers can maintain consistent production schedules, meeting market demand for critical influenza medications without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, as it avoids the use of hazardous cryogenic reagents or high-pressure equipment, making it suitable for deployment in existing multi-purpose reactor facilities. The use of common organic solvents like ethyl acetate and dichloromethane, which have well-established recovery and recycling protocols, aligns with modern environmental, health, and safety (EHS) standards, facilitating easier regulatory approval and permitting for new production lines. This alignment with green chemistry principles enhances the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its adoption for commercial production. These insights are derived directly from the experimental data and process descriptions contained within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the new synthesis route superior to the pyroconic acid method?
A: The conventional method relies on pyroconic acid, which suffers from significant purification challenges and high raw material costs, leading to poor batch-to-batch reproducibility. The novel carbohydrate-based route utilizes readily available starting materials and achieves high purity (over 97%) through straightforward crystallization and filtration steps, ensuring consistent quality for GMP manufacturing.
Q: What are the critical reaction conditions for the oxidation step?
A: The oxidation step utilizing 2-iodoxybenzoic acid (IBX) operates effectively within a temperature range of 40°C to 90°C. The patent specifies that solvents such as ethyl acetate or acetonitrile can be used, with molar ratios of oxidant to substrate ranging from 1:1 to 1:10, allowing for flexible optimization based on scale and cost considerations.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It avoids extreme cryogenic conditions, with most steps proceeding between room temperature and 90°C. Furthermore, the work-up procedures involve standard operations like distillation, filtration, and liquid-liquid extraction, which are easily adaptable to multi-kilogram and metric ton reactors without requiring specialized exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2-(hydroxymethyl)-4H-pyran-4-one Supplier
As the global demand for effective antiviral therapies continues to rise, securing a dependable source of high-quality intermediates is paramount for pharmaceutical companies. NINGBO INNO PHARMCHEM stands ready to support your development and commercialization goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 3-hydroxy-2-(hydroxymethyl)-4H-pyran-4-one we deliver adheres to the highest industry standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this carbohydrate-based method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and accelerate your time to market.
