Scalable Production of Optically Active Heptadecene Compounds for Advanced Pest Control Solutions
Scalable Production of Optically Active Heptadecene Compounds for Advanced Pest Control Solutions
The global agricultural sector is increasingly shifting towards sustainable pest management strategies, moving away from broad-spectrum chemical pesticides that pose environmental risks. A pivotal technology in this transition is the use of species-specific sex pheromones for mating disruption and mass trapping. Patent CN105420285B discloses a breakthrough industrial preparation method for (2S,12Z)-2-acetoxy-12-heptadecene, a critical sex pheromone component for controlling the pistachio tree branch borer (Karmania pisticaciella). This document provides a comprehensive technical analysis of the patented enzymatic kinetic resolution process, highlighting its superiority over traditional synthetic routes. For R&D directors and procurement specialists in the agrochemical industry, understanding this methodology is essential for securing a reliable supply of high-purity pheromone intermediates while optimizing manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active heptadecene derivatives has been plagued by significant scalability and cost challenges. Previous methodologies, such as those disclosed by Gries et al., often relied on the use of expensive chiral starting materials like optically active (S)-propylene oxide. The high cost and limited availability of such chiral pool reagents create a bottleneck for large-scale production, making the final pheromone product economically unviable for widespread agricultural application. Furthermore, alternative enzymatic routes utilizing vinyl acetate as an acyl donor resulted in product mixtures where the ester and the unreacted alcohol had very similar physical properties. This similarity necessitated the use of silica gel column chromatography for separation, a technique that is notoriously difficult to scale industrially due to high solvent consumption, low throughput, and significant waste generation.
The Novel Approach
The patented process introduces a strategic modification to the acyl donor, utilizing vinyl benzoate instead of vinyl acetate in the presence of a lipase catalyst. This seemingly minor chemical change has profound implications for downstream processing. By introducing a benzoyl group rather than an acetyl group, the process generates (2R,12Z)-2-benzoyloxy-12-heptadecene, which possesses a significantly higher boiling point compared to the unreacted (2S,12Z)-2-hydroxy-12-heptadecene. This substantial difference in volatility allows for the separation of the enantiomers using simple distillation apparatus rather than complex chromatography. The reaction scheme below illustrates this elegant kinetic resolution strategy where the lipase selectively benzoylates the (2R)-enantiomer, leaving the desired (2S)-alcohol intact for subsequent conversion.
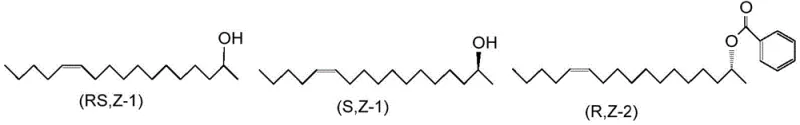
This approach not only simplifies the purification workflow but also enhances the overall yield and optical purity of the final product. The ability to separate components via distillation means the process can be easily adapted to continuous flow systems or large batch reactors, addressing the critical need for cost reduction in agrochemical intermediate manufacturing. By eliminating the chromatography step, the process drastically reduces solvent usage and waste disposal costs, aligning with modern green chemistry principles while ensuring a robust supply chain for pheromone-based pest control agents.
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution
The core of this technology lies in the stereoselectivity of the lipase enzyme, specifically those derived from microorganisms such as Candida antarctica (e.g., Novozym435). In the reaction mixture containing racemic (2RS,12Z)-2-hydroxy-12-heptadecene, the lipase exhibits a strong preference for acylating the (2R)-enantiomer when vinyl benzoate is used as the acyl donor. This enantioselectivity is driven by the specific spatial arrangement of the enzyme's active site, which accommodates the (2R)-alcohol more favorably for nucleophilic attack on the vinyl benzoate. The reaction proceeds under mild conditions, typically between 40°C to 60°C in organic solvents like n-hexane or toluene, preserving the integrity of the Z-alkene moiety which is crucial for biological activity. The use of vinyl esters serves a dual purpose: they act as irreversible acyl donors because the byproduct, vinyl alcohol, tautomerizes to acetaldehyde, driving the equilibrium towards product formation.
Impurity control is inherently managed through the physical separation step. Since the enzymatic reaction yields a mixture of the benzoylated (2R)-ester and the free (2S)-alcohol, any unreacted (2R)-alcohol or over-reacted (2S)-ester (if selectivity were poor) would have boiling points distinct from the main products. However, the high enantioselectivity of the lipase ensures that the (2S)-alcohol remains largely unreacted. The subsequent distillation step acts as a powerful purification tool, removing not only the (2R)-ester but also any residual solvent or low-boiling impurities. The residue, enriched in the (2R)-benzoate, can be recycled or converted if necessary, while the distillate containing the (2S)-alcohol is collected with high optical purity (often >95% ee), ready for the final acetylation step to form the target pheromone.
How to Synthesize (2S,12Z)-2-Acetoxy-12-heptadecene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for industrial implementation, focusing on operational simplicity and high recovery rates. The process begins with the preparation of the racemic alcohol, followed by the critical enzymatic resolution step. Operators must carefully control the molar ratio of vinyl benzoate to the racemic substrate, typically maintaining a slight deficit (0.6 to 0.8 equivalents) to maximize the optical purity of the unreacted alcohol. Following the reaction, the immobilized enzyme is removed by filtration, allowing for potential catalyst recycling. The crude mixture is then subjected to reduced pressure distillation, a unit operation familiar to most chemical manufacturing facilities, ensuring easy technology transfer and scale-up.
- React racemic (2RS,12Z)-2-hydroxy-12-heptadecene with vinyl benzoate in the presence of immobilized lipase (e.g., Novozym435) in an organic solvent like n-hexane at 40-60°C to obtain a mixture of (2R,12Z)-2-benzoyloxy-12-heptadecene and (2S,12Z)-2-hydroxy-12-heptadecene.
- Filter the reaction mixture to remove the enzyme catalyst, wash with aqueous sodium hydrogencarbonate, and remove the solvent under reduced pressure to isolate the concentrated reaction mixture.
- Subject the mixture to distillation under reduced pressure (0.013Pa to 1.333 Pa) to separate the volatile (2S,12Z)-2-hydroxy-12-heptadecene from the residue containing (2R,12Z)-2-benzoyloxy-12-heptadecene, followed by acetylation of the distillate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic distillation-based process offers tangible benefits beyond mere technical feasibility. The elimination of silica gel chromatography removes a major bottleneck in production capacity, allowing for significantly increased throughput without proportional increases in capital expenditure. The reliance on immobilized enzymes, which are stable and reusable, reduces the recurring cost of biocatalysts compared to stoichiometric chiral reagents. Furthermore, the use of common organic solvents like hexane and standard distillation equipment ensures that the process can be implemented in existing multipurpose chemical plants without requiring specialized infrastructure.
- Cost Reduction in Manufacturing: The replacement of expensive chiral starting materials with inexpensive racemic substrates and vinyl benzoate leads to substantial raw material cost savings. Additionally, the removal of the chromatography step drastically cuts down on solvent consumption and waste treatment costs. The process avoids the use of precious metal catalysts or complex chiral ligands, further simplifying the bill of materials and reducing the overall cost of goods sold (COGS) for the final pheromone product.
- Enhanced Supply Chain Reliability: By utilizing robust, immobilized enzymes and standard chemical reagents, the supply chain becomes less vulnerable to disruptions associated with specialty chiral chemicals. The process parameters are forgiving, with wide temperature and pH ranges for enzyme activity, ensuring consistent batch-to-batch quality. This reliability is crucial for meeting the seasonal demands of the agricultural sector, where pheromone dispensers must be available precisely before the pest mating season begins.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory to multi-ton production due to the use of continuous or batch distillation rather than batch chromatography. From an environmental perspective, the enzymatic nature of the reaction operates under mild conditions, reducing energy consumption. The reduction in solvent waste and the absence of heavy metal catalysts simplify regulatory compliance and waste disposal, aligning with increasingly stringent environmental regulations in chemical manufacturing jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process optimization and product quality assurance. Understanding these details is vital for technical teams evaluating the feasibility of adopting this synthesis route for commercial production.
Q: Why is vinyl benzoate preferred over vinyl acetate for this enzymatic resolution?
A: According to the patent data, using vinyl benzoate creates a benzoyloxy group which significantly increases the boiling point difference between the ester product and the unreacted alcohol. This physical property allows for efficient separation via simple distillation, whereas vinyl acetate produces an acetoxy group with a boiling point too close to the alcohol for effective thermal separation without complex chromatography.
Q: What represents the primary cost driver in conventional pheromone synthesis that this method eliminates?
A: Conventional methods often rely on expensive chiral starting materials like optically active (S)-propylene oxide or require silica gel column chromatography for purification. This novel process eliminates the need for costly chiral pool reagents and replaces energy-intensive, solvent-heavy chromatography with a simple distillation step, drastically reducing both raw material and operational expenditures.
Q: Can the enzyme catalyst be reused in this industrial process?
A: Yes, the process utilizes immobilized lipase, such as Novozym435 (lipase from Candida antarctica immobilized on acrylic resin). Because the enzyme is heterogeneous (solid) and the reaction occurs in an organic solvent, the catalyst can be easily recovered via simple filtration after the reaction is complete, allowing for potential reuse and further enhancing the economic viability of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,12Z)-2-Acetoxy-12-heptadecene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity pheromones play in sustainable agriculture. Our technical team has extensively analyzed the pathway described in CN105420285B and possesses the expertise to optimize this enzymatic resolution process for commercial scale-up. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of (2S,12Z)-2-acetoxy-12-heptadecene meets the highest standards for biological efficacy.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your pest control formulations. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the agrochemical market through superior supply chain solutions.
