Scalable Modular Synthesis of Complex Benzophenanthridine and Protoberberine Alkaloids for Commercial Production
Scalable Modular Synthesis of Complex Benzophenanthridine and Protoberberine Alkaloids for Commercial Production
The pharmaceutical industry continuously seeks robust methodologies for constructing complex nitrogen-containing heterocycles, particularly those found in bioactive natural products. A recent technological breakthrough documented in patent CN112851661B introduces a highly modular and diverse synthetic strategy for benzophenanthridine and protoberberine alkaloids. This innovation addresses critical bottlenecks in traditional organic synthesis by leveraging hypervalent iodine chemistry and transition metal catalysis to achieve unprecedented efficiency. The methodology facilitates the rapid assembly of multi-substituted isoquinoline scaffolds, which serve as pivotal advanced intermediates for drug discovery programs. By integrating mild pyridyne generation with novel iridium-catalyzed cross-coupling techniques, this approach offers a reliable pathway for producing high-purity pharmaceutical intermediates. For global procurement teams and R&D directors, understanding the mechanistic depth and commercial viability of this patent is essential for securing a competitive supply chain. The following analysis dissects the technical merits and supply chain implications of this transformative synthetic route.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of isoquinoline-based alkaloids has been plagued by severe operational constraints and limited substrate scope. Traditional routes often rely on harsh reaction conditions, such as strong acidic or basic environments at elevated temperatures, which can degrade sensitive functional groups and compromise overall yield. Furthermore, many established methods require electron-donating groups on the substrate to proceed effectively, drastically restricting the chemical diversity accessible to medicinal chemists. This limitation hinders the exploration of structure-activity relationships, as researchers cannot easily introduce varied substituents to optimize biological potency. Additionally, conventional processes frequently involve multiple protection and deprotection steps, increasing the step count and generating substantial chemical waste. These inefficiencies translate directly into higher manufacturing costs and longer lead times for critical pharmaceutical intermediates. The inability to efficiently connect isoquinoline modules with other templates has also stalled the development of novel derivatives containing this privileged scaffold.
The Novel Approach
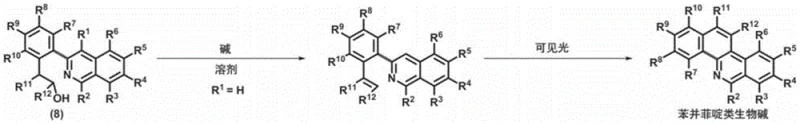
In stark contrast, the novel methodology outlined in the patent data employs a sophisticated cascade of reactions that operate under significantly milder conditions. The process initiates with the generation of pyridyne from improved hypervalent iodine salts using potassium tert-butoxide, a reagent that allows for controlled reactivity at ambient or near-ambient temperatures. This pyridyne intermediate undergoes a [4+2] cycloaddition with dienes to construct multi-substituted isoquinoline ring precursors with high regioselectivity. Subsequently, a newly developed iridium-catalyzed cross-coupling method facilitates the ring-opening and aromatization of these precursors. This step is crucial as it installs halogen handles that enable further diversification through palladium-catalyzed coupling with cyclic boronic acids. The result is a versatile library of 3-arylisoquinoline advanced intermediates that can be tuned via two distinct chemical principles. This modularity empowers researchers to access a wide array of benzophenanthridine and protoberberine structures without redesigning the entire synthetic sequence for each new target.
Mechanistic Insights into Iridium-Catalyzed Cross-Coupling and Aromatization
The core innovation driving the efficiency of this synthetic route lies in the intricate mechanism of the iridium-catalyzed transformation. The reaction utilizes specific iridium complexes, such as [Ir(COD)Cl]2, in conjunction with bulky phosphine ligands like bis(dicyclohexylphosphinophenyl)ether. These ligands create a steric environment that promotes oxidative addition into the carbon-heteroatom bond of the isoquinoline precursor. Following this, the presence of Lewis acids, specifically scandium trifluoromethanesulfonate, activates the substrate for nucleophilic attack, facilitating the ring-opening event. The subsequent aromatization step restores aromatic stability to the isoquinoline core, yielding stable 3-haloisoquinoline products. This mechanistic pathway is highly advantageous because it tolerates a broad range of functional groups, including halogens and alkoxy substituents, which are often incompatible with other metal-catalyzed systems. The precise control over the catalytic cycle ensures that side reactions, such as homocoupling or premature decomposition, are minimized. Understanding this mechanism is vital for R&D directors aiming to replicate or scale this chemistry, as slight deviations in ligand choice or Lewis acid loading can impact the reaction kinetics and product distribution significantly.

Impurity control is another critical aspect where this mechanistic design excels, directly impacting the quality of the final pharmaceutical intermediates. The use of defined stoichiometric ratios, such as a 1:10 molar ratio of isoquinoline precursor to heteroatom source, ensures complete conversion while limiting the formation of over-reacted byproducts. The reaction is typically conducted under an inert nitrogen atmosphere at controlled temperatures ranging from 40°C to 80°C, which prevents thermal degradation of sensitive intermediates. Furthermore, the selection of solvents like tetrahydrofuran provides an optimal polarity balance that stabilizes the catalytic species throughout the transformation. By rigorously optimizing these parameters, the process achieves a clean reaction profile that simplifies downstream purification. For quality assurance teams, this means that the resulting 3-haloisoquinolines possess a consistent impurity profile, reducing the burden on analytical laboratories during batch release testing. The ability to predict and manage impurity formation at the mechanistic level is a hallmark of a mature, scalable chemical process suitable for GMP manufacturing environments.
How to Synthesize 3-Arylisoquinoline Efficiently
The practical execution of this synthesis involves a sequential workflow designed to maximize yield and operational simplicity. Initially, the hypervalent iodine salt is reacted with a diene in the presence of a base to form the isoquinoline precursor, a step that benefits from the mild conditions described earlier. Once the precursor is isolated, it is subjected to the iridium-catalyzed ring-opening protocol to generate the 3-haloisoquinoline intermediate. This intermediate then serves as the electrophile in a Suzuki-Miyaura type coupling with multi-substituted cyclic boronic acids. The coupling reaction utilizes palladium catalysts like Pd(dba)2 and specialized ligands to ensure high turnover numbers. Finally, the resulting 3-arylisoquinoline undergoes cyclization under either basic conditions with visible light irradiation or via intramolecular nucleophilic substitution to yield the target alkaloids. Detailed standardized synthesis steps see the guide below.
- Generate pyridyne from hypervalent iodine salts under mild potassium tert-butoxide conditions and perform [4+2] cycloaddition with dienes.
- Execute iridium-catalyzed cross-coupling to achieve ring-opening and aromatization of isoquinoline precursors into 3-haloisoquinolines.
- Couple 3-haloisoquinolines with cyclic boronic acids using palladium catalysis, followed by cyclization to form target alkaloids.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic technology offers profound benefits for procurement managers and supply chain heads focused on cost efficiency and reliability. The shift towards milder reaction conditions eliminates the need for expensive high-pressure reactors or extreme temperature control systems, leading to substantial capital expenditure savings. Moreover, the use of readily available starting materials, such as substituted phenethyl alcohols for boronic acid synthesis, mitigates the risk of raw material shortages. This accessibility ensures a stable supply chain, reducing the vulnerability to market fluctuations that often plague specialty chemical sourcing. The modular nature of the synthesis also allows for flexible production scheduling, as different alkaloid variants can be produced using the same core infrastructure with minimal changeover time. These factors collectively contribute to a more resilient and cost-effective manufacturing operation that can adapt quickly to changing market demands.
- Cost Reduction in Manufacturing: The elimination of harsh reagents and the reduction in synthetic step count directly lower the cost of goods sold. By avoiding expensive transition metal removal steps often required with other catalysts, the process simplifies purification and reduces solvent consumption. The high atom economy of the cycloaddition and coupling steps ensures that raw materials are converted into product with minimal waste. This efficiency translates into significant operational cost savings without compromising the quality of the high-purity pharmaceutical intermediates. Additionally, the ability to conduct reactions at lower temperatures reduces energy consumption, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commercially available reagents and robust catalytic systems minimizes supply chain disruptions. Unlike proprietary reagents that may have single-source suppliers, the chemicals used in this protocol are widely sourced from multiple vendors globally. This diversification reduces lead time for high-purity pharmaceutical intermediates and ensures continuity of supply even during geopolitical or logistical challenges. The scalability of the process from gram to kilogram levels means that procurement teams can secure sufficient quantities for clinical trials and commercial launch without needing to revalidate entirely new synthetic routes. This reliability is crucial for maintaining production schedules and meeting delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex alkaloids, with reaction conditions that are safe and manageable in large vessels. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, lowering disposal costs and regulatory risks. The use of visible light for cyclization in certain pathways represents a green chemistry advancement that reduces the carbon footprint of the manufacturing process. These environmental benefits not only improve corporate sustainability metrics but also facilitate smoother regulatory approvals in markets with strict ecological standards. The combination of scalability and compliance makes this technology a strategic asset for long-term manufacturing planning.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These answers are derived from the specific technical details and beneficial effects described in the patent documentation. They provide clarity on the operational feasibility and strategic value of adopting this route for alkaloid production. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their existing portfolios. The responses cover mechanistic robustness, purity considerations, and scalability potential.
Q: What are the primary advantages of this iridium-catalyzed method over traditional alkaloid synthesis?
A: This method utilizes mild reaction conditions, such as room temperature cycloaddition, avoiding harsh reagents. It allows for diverse functional group tolerance and modular construction of complex skeletons like benzophenanthridines.
Q: How does this protocol impact the purity profile of pharmaceutical intermediates?
A: The specific ligand and catalyst systems minimize side reactions and byproduct formation. The stepwise purification of 3-haloisoquinoline intermediates ensures high-purity final alkaloids suitable for rigorous medicinal chemistry applications.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the use of readily available starting materials and robust catalytic cycles supports scalability. The elimination of extreme conditions reduces equipment stress and enhances operational safety for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzophenanthridine Supplier
The technical potential of this modular synthesis route is immense, offering a pathway to produce complex alkaloids with high efficiency and purity. NINGBO INNO PHARMCHEM stands ready to leverage this expertise as a dedicated CDMO partner, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to ensure that every batch meets the highest industry standards. We understand the critical nature of pharmaceutical intermediates in the drug development timeline and are committed to delivering consistent quality. By partnering with us, clients gain access to advanced catalytic technologies and process optimization capabilities that accelerate time to market.
We invite you to initiate a dialogue regarding your specific supply chain optimization needs. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your project requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Together, we can explore how this innovative synthetic strategy can enhance your product portfolio and drive commercial success. Let us collaborate to transform complex chemical challenges into scalable manufacturing realities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
