Advanced Catalytic Route for High-Purity Beta-Bromoformate Intermediates and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN107628945B, which details a novel method for synthesizing β-bromoformate compounds. This technology represents a paradigm shift from traditional halogenation methods that often rely on toxic, unstable reagents and harsh conditions. By leveraging a unique combination of bromate-intercalated zinc-aluminum hydrotalcite (ZnAl-BrO3--LDHs) and alkali metal bromides in a formic acid medium, this process achieves high efficiency under remarkably mild conditions. For R&D directors and procurement specialists, understanding this innovation is crucial, as it directly addresses long-standing challenges regarding reagent stability, product purity, and operational safety in the production of high-value organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of β-bromoformate derivatives has been plagued by significant technical and safety hurdles. Conventional methodologies frequently employ reagents such as N-bromosuccinimide (NBS) or N,N-dibromo-p-toluenesulfonamide in the presence of polar aprotic solvents like N,N-dimethylformamide (DMF). These traditional approaches suffer from inherent drawbacks that impact both cost and safety profiles. Firstly, bromine sources like NBS are notoriously hygroscopic and unstable, requiring stringent storage conditions and careful handling to prevent degradation before use. Secondly, the toxicity associated with these electrophilic brominating agents poses serious occupational health risks and complicates waste disposal protocols. Furthermore, the reliance on DMF as a solvent introduces severe downstream processing challenges; removing high-boiling point DMF from the final product is energy-intensive and often requires complex extraction or distillation steps, which can lead to product loss and increased impurity levels. These factors collectively result in lower overall yields and higher manufacturing costs, making conventional routes less attractive for modern, sustainability-focused supply chains.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN107628945B offers a streamlined and greener alternative that effectively circumvents the limitations of prior art. This innovative route utilizes a heterogeneous catalyst system comprising bromate-intercalated zinc-aluminum hydrotalcite alongside a simple alkali metal bromide, such as potassium bromide. The reaction proceeds in formic acid, which serves a dual function as both the reaction medium and the nucleophilic reagent, thereby simplifying the stoichiometry and reducing the number of raw materials required. The process operates under mild thermal conditions, typically between 30°C and 60°C, which significantly reduces energy consumption compared to high-temperature alternatives. The use of a solid, layered double hydroxide catalyst ensures excellent stability and ease of separation; post-reaction, the catalyst can be removed simply by centrifugation, leaving a clear solution for straightforward extraction. This not only enhances the purity of the crude product but also drastically simplifies the workup procedure, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
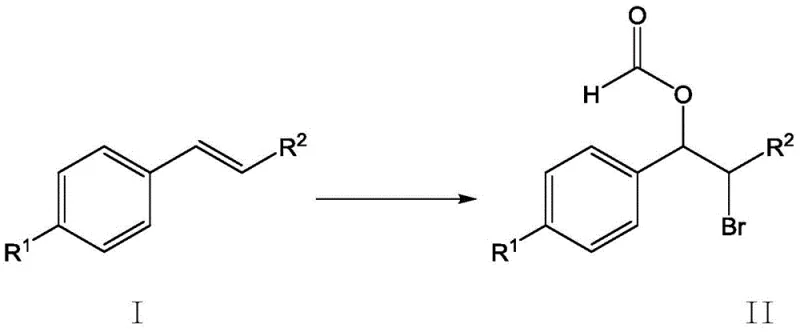
Mechanistic Insights into ZnAl-BrO3-LDHs Catalyzed Bromoformylation
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic role of the zinc-aluminum hydrotalcite catalyst. The ZnAl-BrO3--LDHs structure features intercalated bromate anions within its layered lattice, which act as a controlled reservoir for active bromine species. Upon interaction with the alkali metal bromide in the acidic formic acid environment, the catalyst facilitates the generation of electrophilic bromine in situ. This controlled release mechanism prevents the localized high concentrations of free bromine that often lead to poly-bromination or other undesirable side reactions in homogeneous systems. The formic acid molecule attacks the transient bromonium ion intermediate formed on the styrene double bond, leading to the anti-addition product with high regioselectivity. This precise control over the reaction pathway is critical for R&D teams aiming to minimize impurity profiles, as it ensures that the resulting β-bromoformate possesses the specific structural integrity required for downstream transformations, such as cyclization into epoxides or further functionalization.
Furthermore, the heterogeneous nature of the catalyst plays a pivotal role in impurity control and product isolation. In homogeneous catalysis, trace metal residues often contaminate the final API intermediate, necessitating expensive purification steps like scavenging or recrystallization. With the ZnAl-BrO3--LDHs system, the catalyst remains a distinct solid phase throughout the reaction. Post-reaction centrifugation effectively removes the bulk of the catalytic material, significantly reducing the metal load in the organic phase before any chromatographic purification is even attempted. This physical separation capability translates directly into higher purity specifications and reduced solvent usage during the refining stage. For quality assurance professionals, this means a more consistent impurity spectrum and a lower risk of batch-to-batch variability, which are essential metrics for maintaining regulatory compliance in pharmaceutical production.
How to Synthesize Beta-Bromoformate Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and reproducibility. The protocol involves charging a reactor with the styrene substrate and an alkali metal bromide, followed by the addition of formic acid as the solvent. The key catalytic component, ZnAl-BrO3--LDHs, is then introduced to the mixture. The reaction is maintained at a moderate temperature, optimally around 40°C, for a duration of approximately 2 hours, although the patent indicates a viable range of 1 to 4 hours depending on the specific substrate substituents. Upon completion, the solid catalyst is separated via centrifugation, and the filtrate undergoes a standard liquid-liquid extraction using dichloromethane and water. The organic layer is dried and concentrated, with final purification achieved through column chromatography using a petroleum ether and ethyl acetate gradient. This straightforward workflow minimizes unit operations and equipment requirements, facilitating easier technology transfer from the laboratory to pilot and commercial scales.
- Mix styrene derivative, potassium bromide, and formic acid solvent in a reactor.
- Add bromate-intercalated zinc-aluminum hydrotalcite catalyst and stir at 30-60°C for 1-4 hours.
- Centrifuge to remove solid catalyst, extract organic phase with dichloromethane, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrotalcite-catalyzed route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial optimization of the cost structure associated with raw materials and waste management. By replacing expensive and hazardous brominating agents like NBS with stable, commodity-grade potassium bromide and a reusable solid catalyst, the direct material costs are significantly lowered. Moreover, the elimination of DMF removes the need for specialized solvent recovery systems and reduces the environmental fees associated with disposing of toxic amide wastes. The simplified workup procedure, which relies on centrifugation rather than complex distillation or multiple wash cycles, reduces labor hours and utility consumption, contributing to a leaner manufacturing process that enhances overall margin potential without compromising product quality.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the substitution of high-cost reagents with inexpensive alternatives and the reduction of processing time. The use of formic acid as a dual-purpose solvent and reactant eliminates the need for additional nucleophiles, streamlining the bill of materials. Additionally, the mild reaction conditions reduce energy expenditure for heating and cooling, while the efficient catalyst separation minimizes product loss during filtration. These cumulative efficiencies result in a more competitive cost base for producing high-purity pharmaceutical intermediates, allowing manufacturers to offer better pricing to downstream clients while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the volatility of specialized reagent markets. This method mitigates such risks by relying on widely available and stable raw materials. Potassium bromide and formic acid are commodity chemicals with robust global supply chains, ensuring that production schedules are not disrupted by shortages of niche brominating agents. Furthermore, the stability of the ZnAl-BrO3--LDHs catalyst allows for long-term storage without degradation, enabling manufacturers to maintain strategic inventory buffers. This resilience is critical for meeting the just-in-time delivery demands of multinational pharmaceutical companies and securing long-term supply agreements.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this route is inherently designed for expansion. The heterogeneous catalyst system is easily adaptable to large-scale reactors, and the absence of highly exothermic steps or toxic gas evolution simplifies engineering controls. From an environmental perspective, the process aligns with green chemistry principles by reducing hazardous waste generation and avoiding persistent organic pollutants. This compliance facilitates smoother regulatory approvals and reduces the carbon footprint of the manufacturing site, appealing to increasingly eco-conscious stakeholders and investors in the global chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement officers assessing supplier qualifications.
Q: What are the advantages of using ZnAl-BrO3-LDHs over traditional brominating agents?
A: Unlike traditional agents like N-bromosuccinimide (NBS) which are toxic and moisture-sensitive, the ZnAl-BrO3-LDHs catalyst offers superior stability, lower toxicity, and easier separation via centrifugation, significantly simplifying post-processing.
Q: How does this method improve environmental compliance in pharmaceutical manufacturing?
A: The process utilizes formic acid as both solvent and nucleophile, eliminating the need for hazardous solvents like DMF. Additionally, the heterogeneous catalyst can be recovered, reducing heavy metal waste and aligning with green chemistry principles.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the mild reaction conditions (30-60°C) and simple workup procedure involving centrifugation and standard extraction make this method highly scalable for industrial applications, ensuring consistent quality and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Bromoformate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical industry. Our technical team has extensively analyzed the potential of hydrotalcite-catalyzed pathways, confirming their viability for producing high-quality intermediates with exceptional purity profiles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-bromoformate delivered meets the exacting standards required for API synthesis and fine chemical applications.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and operational excellence can drive value and reliability for your organization, ensuring a steady supply of critical intermediates for your most important projects.
