Advanced Palladium-Catalyzed Synthesis of Polysubstituted 2-Naphthol Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Polysubstituted 2-Naphthol Intermediates for Commercial Scale-Up
According to the technical disclosures in patent CN102503752B, the chemical industry is witnessing a significant shift towards more efficient and environmentally benign methodologies for constructing polycyclic aromatic scaffolds. Specifically, the preparation of polysubstituted 2-naphthol derivatives, which serve as critical building blocks for dyes, agrochemicals, and pharmaceutical active ingredients, has traditionally been plagued by multi-step sequences and harsh reaction conditions. This new proprietary technology introduces a streamlined, two-step catalytic sequence that leverages carbon monoxide as a C1 building block, fundamentally altering the economic and operational landscape for manufacturers of high-purity pharmaceutical intermediates. By integrating a palladium-catalyzed carbonylative coupling with a subsequent intramolecular Heck cyclization, this process achieves what previous methods could not: a direct, high-yield pathway from simple benzyl halides to complex naphthol cores without the formation of difficult-to-separate isomers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted beta-naphthols has relied heavily on classical condensation reactions such as the Dieckmann cyclization or acid-catalyzed ring closures of cyclohexadione derivatives. These legacy processes often necessitate the use of stoichiometric amounts of strong mineral acids like sulfuric acid or perchloric acid, creating severe corrosion issues for reactor equipment and generating substantial quantities of hazardous acidic waste that requires costly neutralization and disposal. Furthermore, these thermal cyclizations frequently suffer from poor regioselectivity, leading to mixtures of structural isomers that drastically reduce the overall yield of the desired target molecule and complicate the purification process. In many cases, the starting materials for these traditional routes, such as ortho-alkynyl phenyl epoxides or specific aryl ketenes, are themselves expensive and require multi-step preparation, thereby inflating the total cost of goods and extending the manufacturing lead time significantly.
The Novel Approach
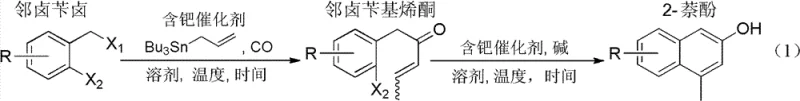
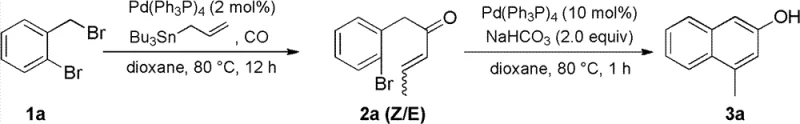
In stark contrast, the methodology described in CN102503752B utilizes a modern organometallic strategy that begins with the carbonylative coupling of readily available o-halobenzyl halides with tributylallyltin. This initial step constructs the necessary carbon skeleton and installs the ketone functionality in a single operation under mild carbon monoxide pressure, typically ranging from 0.1 to 1.0 MPa. The subsequent transformation involves an elegant intramolecular Heck reaction that closes the second aromatic ring with high precision, driven by a palladium catalyst system that tolerates a wide array of functional groups including ethers, esters, and halogens. This approach not only shortens the synthetic route by eliminating intermediate isolation steps in some configurations but also ensures that the final product is obtained as a single isomer, thereby enhancing the overall process mass intensity and reducing solvent consumption during purification.
Mechanistic Insights into Palladium-Catalyzed Carbonylation and Cyclization
The core of this technological breakthrough lies in the dual catalytic cycles orchestrated by zero-valent or divalent palladium species. In the first stage, the palladium catalyst undergoes oxidative addition into the carbon-halogen bond of the o-halobenzyl halide substrate, forming an organopalladium intermediate. This species then coordinates and inserts carbon monoxide to generate an acyl-palladium complex, which subsequently reacts with the allyl-tin reagent via transmetallation to release the o-halobenzyl enone product and regenerate the active catalyst. The beauty of this mechanism is its robustness; the use of tin reagents facilitates the transmetallation step efficiently even at moderate temperatures of around 80°C, preventing the decomposition of sensitive functional groups that might occur under more vigorous thermal conditions required by non-catalytic methods.
Following the formation of the enone intermediate, the second catalytic cycle initiates the ring-closing event through an intramolecular Heck mechanism. The palladium catalyst activates the remaining aryl halide bond on the same molecule, positioning the metal center in proximity to the alkene moiety of the enone side chain. Migratory insertion of the alkene into the palladium-carbon bond forms a new carbon-carbon bond, creating the bicyclic naphthalene framework. Subsequent beta-hydride elimination restores the aromaticity of the newly formed ring and releases the final polysubstituted 2-naphthol product. This cascade-like efficiency ensures that the reaction proceeds with high atom economy, minimizing the generation of organic waste and simplifying the downstream workup to basic extraction and chromatography, which is crucial for maintaining high purity standards in pharmaceutical manufacturing.
How to Synthesize Polysubstituted 2-Naphthol Efficiently
The practical implementation of this synthesis involves carefully controlling the stoichiometry of the palladium catalyst and the base to maximize turnover numbers while minimizing metal residue in the final product. Operators typically dissolve the o-halobenzyl halide and tributylallyltin in a polar aprotic solvent such as 1,4-dioxane or acetonitrile, pressurize the reactor with carbon monoxide, and heat the mixture to approximately 80°C for 12 to 24 hours. Once the intermediate enone is formed, it can be isolated or treated directly with a base like sodium bicarbonate or potassium phosphate in the presence of additional palladium catalyst to effect the cyclization. For detailed operational parameters, safety guidelines regarding carbon monoxide handling, and specific purification protocols, please refer to the standardized synthesis steps provided below.
- Perform palladium-catalyzed carbonylative coupling of o-halobenzyl halide with tributylallyltin under CO pressure (0.1-1.0 MPa) to form o-halobenzyl enone.
- Isolate the intermediate enone via silica gel chromatography or proceed directly depending on purity requirements.
- Execute intramolecular Heck cyclization using a palladium catalyst and base (e.g., NaHCO3, K3PO4) in polar aprotic solvents at 80-130°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented process offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility often associated with complex aromatic intermediates. By utilizing carbon monoxide, a commodity chemical available at a fraction of the cost of specialized organometallic reagents, the raw material cost basis is significantly reduced compared to routes relying on precious metal alkynes or pre-functionalized cyclic ketones. Moreover, the operational simplicity of running the reaction at atmospheric or slightly elevated pressures in standard glass-lined or stainless steel reactors means that existing manufacturing infrastructure can be utilized without the need for capital-intensive equipment upgrades, thereby accelerating the time-to-market for new drug candidates requiring these scaffolds.
- Cost Reduction in Manufacturing: The elimination of stoichiometric strong acids and the reduction in synthetic steps from multiple stages to a concise two-step sequence drastically lowers the consumption of utilities and consumables. Since the process avoids the use of expensive ruthenium catalysts often cited in alternative literature, the direct material cost is optimized, and the simplified purification workflow reduces the volume of organic solvents required for chromatography, leading to substantial savings in waste treatment and solvent recovery costs.
- Enhanced Supply Chain Reliability: The starting materials, specifically o-halobenzyl halides and tributylallyltin, are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the palladium catalytic system against moisture and oxygen variations ensures consistent batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply lines to downstream API manufacturers who demand rigorous quality consistency and on-time delivery performance.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, having been demonstrated effectively from gram scale to multi-kilogram batches without loss of yield or selectivity. The environmental profile is markedly improved due to the absence of corrosive acid waste streams and the potential for recycling the palladium catalyst from the reaction residue, aligning with modern green chemistry principles and helping procurement teams meet increasingly stringent corporate sustainability and environmental compliance targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide clarity on feasibility and performance. Understanding these details is essential for project managers evaluating the integration of this route into their existing production portfolios.
Q: What are the key advantages of this Pd-catalyzed route over traditional acid-cyclization methods?
A: This method avoids harsh strong acid or base conditions required in traditional Dieckmann condensations, offering better functional group tolerance and eliminating isomer byproducts common in older synthetic routes.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes commercially available raw materials like CO and simple o-halobenzyl halides, operates at moderate temperatures (80°C), and uses standard palladium catalysts suitable for large-scale reactor operations.
Q: What is the typical purity profile of the resulting 2-naphthol derivatives?
A: The method produces single products without isomers due to the specificity of the intramolecular Heck reaction, simplifying downstream purification and ensuring high purity specifications for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 2-Naphthol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into industrial reality. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify the absence of isomers and metal residues, guaranteeing that every batch of polysubstituted 2-naphthol meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall cost of goods.
