Scalable Synthesis of Chiral 4-Chloro-3-Hydroxybutyrate via Advanced Cobalt-Catalyzed Hydroesterification Technology
Scalable Synthesis of Chiral 4-Chloro-3-Hydroxybutyrate via Advanced Cobalt-Catalyzed Hydroesterification Technology
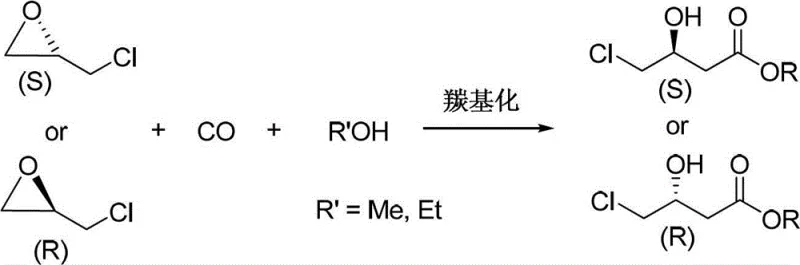
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access critical chiral building blocks, and the technology disclosed in Chinese Patent CN103420837A represents a significant leap forward in this domain. This patent details a robust preparation method for chiral 4-chloro-3-hydroxybutyrate (CHBE), a versatile intermediate essential for synthesizing statins, L-carnitine, and various alkaloids. By leveraging advanced carbonylation chemistry, the invention enables the direct conversion of chiral epichlorohydrin into the target ester using carbon monoxide and alcohols. This one-step hydroesterification process stands out for its exceptional atom economy and operational simplicity, addressing long-standing challenges related to toxicity and cost in traditional synthetic routes. For R&D directors and procurement specialists, understanding this technology is crucial for optimizing supply chains and reducing the environmental footprint of API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 4-chloro-3-hydroxybutyrate has relied on methods that present significant safety and economic hurdles for large-scale operations. Traditional chemical routes often involve the use of highly toxic sodium cyanide for ring-opening reactions, followed by hydrolysis and esterification, which generates substantial hazardous waste and requires complex purification protocols to remove cyanide residues. Alternatively, asymmetric catalytic hydrogenation of 4-chloroacetoacetate has been explored, but this approach suffers from the high cost of chiral catalysts and the need for specialized equipment capable of withstanding strong acidic conditions and high pressures. Furthermore, enzymatic methods, while environmentally friendly, are frequently limited by the high purity requirements of the substrate and the necessity for expensive coenzymes, making them less viable for cost-sensitive bulk production. These legacy processes often result in lower overall yields and increased lead times, creating bottlenecks for reliable pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast to these conventional limitations, the novel approach described in the patent utilizes a cobalt-catalyzed hydroesterification of chiral epichlorohydrin, offering a streamlined and economically superior alternative. This method achieves 100% atom economy by directly incorporating carbon monoxide into the epoxide ring in the presence of an alcohol, effectively constructing the carbon skeleton in a single operational step. The reaction conditions are remarkably mild, typically operating at temperatures between 60°C and 100°C and carbon monoxide pressures ranging from 4 MPa to 8 MPa, which are well within the capabilities of standard industrial high-pressure reactors. By eliminating the need for toxic cyanides or expensive enzymatic systems, this route significantly simplifies the downstream processing and waste treatment requirements. The ability to use readily available bulk chemicals like methanol or ethanol as reactants further enhances the commercial viability of this process for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cobalt-Catalyzed Hydroesterification
The core of this innovative synthesis lies in the sophisticated catalytic system comprising a metal compound containing cobalt carbonyl and a nitrogen-containing ligand. The cobalt source, such as dicobalt octacarbonyl (Co2(CO)8) or in-situ generated cobalt carbonyl species, acts as the active center for carbon monoxide activation and insertion. The mechanism likely involves the coordination of the epoxide oxygen to the cobalt center, facilitating the nucleophilic attack or insertion of the carbonyl group. Crucially, the presence of nitrogen-containing compounds like pyridine derivatives or zinc-pyridine complexes serves to modulate the electronic properties of the cobalt catalyst, enhancing its stability and selectivity. This ligand acceleration effect ensures that the ring-opening occurs regioselectively and stereospecifically, preventing the formation of unwanted by-products. The synergy between the cobalt carbonyl and the nitrogen ligand creates a highly efficient catalytic cycle that drives the reaction to completion with high turnover numbers, even at relatively low catalyst loadings of 0.5% to 10% molar ratio.
From a stereochemical perspective, the preservation of optical purity is paramount for the utility of CHBE in drug synthesis, and this mechanism excels in maintaining configuration integrity. The patent data explicitly demonstrates that both (R)- and (S)-epichlorohydrin undergo the reaction without racemization, retaining their initial enantiomeric excess values which can be as high as 99.9%. This retention of configuration suggests that the reaction proceeds through a pathway that does not involve the formation of a planar carbocation intermediate, which would otherwise lead to loss of chirality. Instead, the concerted nature of the carbonylation likely ensures that the stereocenter remains undisturbed throughout the transformation. For quality control teams, this means that the optical purity of the final product is directly dependent on the quality of the starting epichlorohydrin, allowing for predictable and consistent output of high-purity chiral building blocks without the need for costly chiral resolution steps post-reaction.
How to Synthesize Chiral 4-Chloro-3-Hydroxybutyrate Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalytic system and the control of reaction parameters to maximize yield and selectivity. The process begins with the charging of a high-pressure reactor with the chiral epichlorohydrin substrate, the chosen alcohol solvent (methanol or ethanol), and the pre-mixed or in-situ formed catalyst solution. It is critical to ensure that the reactor is thoroughly purged with inert gas and subsequently with carbon monoxide to eliminate oxygen, which could deactivate the sensitive cobalt catalyst. Once sealed, the vessel is pressurized with CO to the desired level, typically around 6.0 MPa, and heated to the target temperature, often optimized around 60°C to 80°C for balanced reaction kinetics and safety. Following the reaction period, which may last from 1 to 16 hours depending on the specific catalyst loading and temperature, the mixture is cooled and depressurized. The crude product is then subjected to simple distillation under reduced pressure to isolate the high-purity ester, achieving yields often exceeding 95% with purity levels greater than 99%.
- Load chiral epichlorohydrin, alcohol (methanol or ethanol), and a catalyst system comprising a cobalt carbonyl compound and a nitrogen-containing ligand into a high-pressure reactor.
- Purge the reactor with carbon monoxide gas multiple times to remove air, then pressurize the system to between 1 MPa and 12 MPa.
- Heat the reaction mixture to a temperature between 40°C and 140°C for 1 to 16 hours, then cool and purify the product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydroesterification technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift from multi-step, hazardous chemistries to a direct, one-step carbonylation process fundamentally alters the cost structure and risk profile of producing chiral 4-chloro-3-hydroxybutyrate. By utilizing bulk commodity chemicals such as carbon monoxide and simple alcohols, the reliance on specialized, high-cost reagents is drastically minimized, leading to substantial cost savings in raw material procurement. Furthermore, the elimination of toxic cyanide reagents removes the need for expensive hazardous waste disposal protocols and specialized containment infrastructure, thereby reducing the overall operational expenditure associated with environmental compliance. This streamlined approach not only lowers the barrier to entry for production but also enhances the resilience of the supply chain against regulatory changes regarding chemical safety.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by its high atom efficiency and the simplicity of the catalyst system. Unlike enzymatic routes that require costly coenzymes and complex protein engineering, or hydrogenation routes needing precious metal catalysts, the cobalt-based system utilizes inexpensive and abundant metals. The high selectivity of the reaction minimizes the formation of by-products, which reduces the load on purification units and increases the overall mass balance efficiency. Consequently, the cost per kilogram of the final API intermediate is significantly lowered, allowing for more competitive pricing in the global market without compromising on quality margins. This efficiency makes the process highly attractive for scaling up to meet the demands of blockbuster drug production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of sourcing specialized reagents, but this method mitigates such risks by relying on widely available feedstocks. Chiral epichlorohydrin is a commercially mature chemical with a stable global supply, and carbon monoxide is a standard industrial gas produced in vast quantities. This accessibility ensures that production schedules are less likely to be disrupted by raw material shortages compared to processes dependent on niche biological enzymes or custom-synthesized chiral ligands. Additionally, the robustness of the catalyst system allows for flexible manufacturing campaigns, enabling suppliers to respond quickly to fluctuations in market demand. This reliability is critical for pharmaceutical companies that require just-in-time delivery of key intermediates to maintain their own production timelines.
- Scalability and Environmental Compliance: The transition from laboratory scale to industrial production is seamless with this technology due to the use of standard high-pressure reactor equipment found in most fine chemical plants. The mild reaction conditions reduce the energy consumption required for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. Moreover, the 'three wastes' (waste water, waste gas, waste residue) are significantly reduced compared to traditional cyanide-based routes, aligning with increasingly stringent global environmental regulations. This green chemistry profile not only facilitates easier permitting for new production facilities but also enhances the corporate sustainability credentials of the manufacturer, a factor that is becoming increasingly important in vendor selection criteria for major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process performance and scalability. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial implementation. Understanding these nuances is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: Does the hydroesterification process cause racemization of the chiral center?
A: No, the patent data confirms that R- or S-type epichlorohydrin does not undergo racemization under the specified reaction conditions, maintaining an enantiomeric excess (ee) value of over 99.9% in optimized examples.
Q: What are the primary advantages of this method over enzymatic routes?
A: Unlike enzymatic methods which require expensive coenzymes and highly pure substrates, this chemical carbonylation route uses bulk chemicals, offers 100% atom economy, and avoids the complex derivatization steps associated with biocatalysts.
Q: Is the catalyst system suitable for large-scale industrial production?
A: Yes, the process utilizes simple cobalt carbonyl catalysts and nitrogen ligands that are easy to prepare. The reaction conditions (4-8 MPa, 60-100°C) are mild enough for standard high-pressure reactors used in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 4-Chloro-3-Hydroxybutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify enantiomeric excess and chemical purity. By leveraging advanced technologies like the cobalt-catalyzed hydroesterification described in CN103420837A, we are able to offer a reliable pharmaceutical intermediate supplier partnership that balances technical excellence with commercial viability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to review specific COA data and route feasibility assessments, we are ready to provide the support necessary to accelerate your development timeline. Contact us today to explore how our manufacturing capabilities can drive efficiency and reliability in your production of high-purity chiral building blocks.
