Advanced Fluorene Spirophenyl Tricarbazole Derivatives for High-Efficiency OLED Manufacturing
Advanced Fluorene Spirophenyl Tricarbazole Derivatives for High-Efficiency OLED Manufacturing
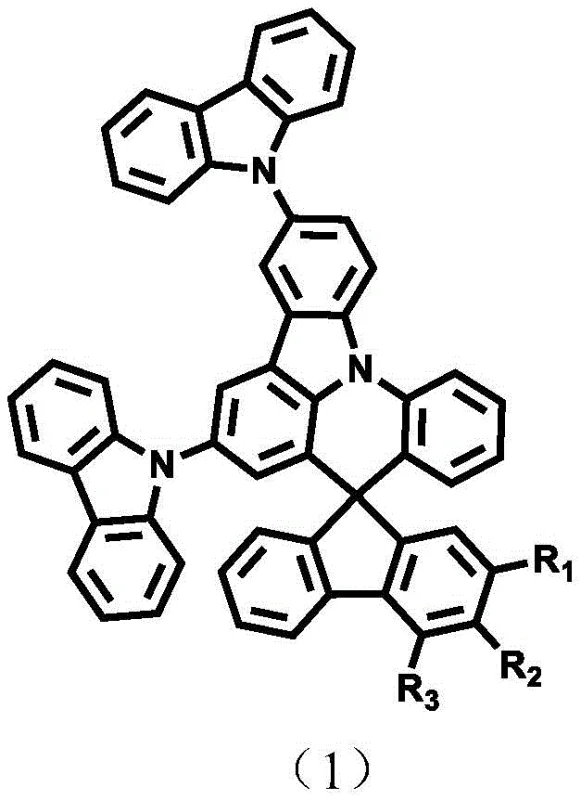
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that can simultaneously deliver high efficiency, long operational lifetime, and minimal efficiency roll-off at high brightness. Patent CN110483574B introduces a novel class of fluorene spirophenyl tricarbazole derivatives designed to address these critical challenges in electronic chemical manufacturing. These compounds feature a unique spiro-structural motif that decouples the electronic conjugation between the fluorene and tricarbazole units, resulting in an exceptionally high triplet energy level of up to 2.85 eV. This technical breakthrough positions them as superior candidates for use as host materials, exciton blocking layers, or electron transport materials in next-generation display and lighting applications. For R&D directors and procurement specialists, understanding the synthesis and commercial viability of these materials is essential for securing a competitive edge in the supply chain of high-performance optoelectronic components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional host materials currently dominating the commercial OLED market, such as 1,3-bis(carbazol-9-yl)benzene (mCP), suffer from inherent limitations regarding efficiency stability under high current densities. While mCP offers decent triplet energy, devices utilizing it often exhibit severe efficiency roll-off, meaning their external quantum efficiency drops significantly as brightness increases to practical levels like 5000 candela per square meter. This phenomenon is primarily due to triplet-triplet annihilation and other non-radiative decay pathways that become prevalent when the host material cannot effectively confine excitons or manage charge balance. Furthermore, the planar structure of many conventional hosts can lead to crystallization issues over time, compromising the morphological stability of the thin film and shortening the overall device lifespan. These drawbacks necessitate the development of new molecular architectures that can maintain high performance without sacrificing stability.
The Novel Approach
The innovative approach detailed in the patent leverages a rigid spiro-linkage to construct a three-dimensional molecular framework. By connecting a fluorene unit orthogonally to a phenyl tricarbazole core, the synthesis creates a steric bulk that prevents intermolecular aggregation and pi-stacking, which are common causes of efficiency quenching. This structural design not only preserves the high triplet energy required for blue and green phosphorescent dopants but also enhances the glass transition temperature, ensuring the amorphous state of the film is maintained during device operation. The versatility of this scaffold allows for further functionalization at the 2, 3, or 4 positions of the fluorene moiety, enabling fine-tuning of charge transport properties and solubility. This modularity provides a robust platform for developing a library of materials tailored to specific device architectures, offering a significant upgrade over static, single-purpose host materials.
Mechanistic Insights into Spiro-Linkage Formation and Functionalization
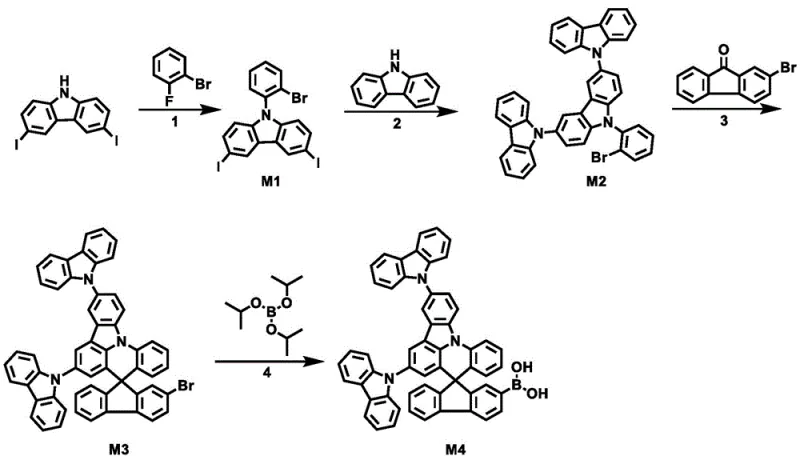
The synthesis of these advanced derivatives relies on a sophisticated sequence of organometallic transformations that ensure high regioselectivity and purity. The core construction begins with the formation of the indoloacridine-tricarbazole precursor, followed by a critical lithiation step to generate the spiro-center. Specifically, the reaction involves treating a brominated tricarbazole intermediate with a strong base like n-butyllithium at cryogenic temperatures (-78°C) to generate a nucleophilic carbanion. This species then attacks a fluorenone derivative, forming a tertiary alcohol intermediate which is subsequently dehydrated under acidic conditions to close the spiro-ring. This mechanism is crucial because it establishes the orthogonal geometry that defines the material's electronic properties. Any deviation in temperature or stoichiometry during this step could lead to open-chain byproducts or polymerization, underscoring the need for precise process control.
Following the core assembly, the final functionalization is typically achieved through palladium-catalyzed cross-coupling reactions, such as Suzuki-Miyaura or Buchwald-Hartwig amination. These reactions allow for the introduction of diverse aryl or heteroaryl groups (R1, R2, R3) to modulate the HOMO/LUMO levels. For instance, introducing electron-withdrawing groups like cyano or triazine units can enhance electron transport capabilities, while electron-donating carbazole derivatives can improve hole injection. The patent highlights that controlling the reaction conditions, such as catalyst loading and solvent choice (e.g., toluene/ethanol/water mixtures), is vital for achieving yields in the range of 70-92% for intermediates and 65-85% for final products. This high efficiency in synthesis translates directly to reduced waste and lower raw material consumption, a key factor for sustainable manufacturing.
How to Synthesize Fluorene Spirophenyl Tricarbazole Derivatives Efficiently
The preparation of these high-performance OLED materials follows a modular synthetic strategy that balances complexity with scalability. The process initiates with the N-arylation of carbazole derivatives to build the tricarbazole backbone, followed by cyclization to form the rigid indoloacridine core. Subsequent lithiation and reaction with fluorenone establish the spiro-junction, creating the key brominated intermediate M3. From this versatile pivot point, various functional groups can be attached using standard cross-coupling protocols. The detailed standardized synthesis steps for producing the key boronic acid intermediate M4 and final derivatives are outlined in the structured guide below, ensuring reproducibility and quality control for industrial batches.
- Preparation of Intermediate M1 via nucleophilic substitution of 3,6-diiodocarbazole with 1-bromo-2-fluorobenzene using cesium carbonate in DMF at 150°C.
- Synthesis of Intermediate M2 through copper-catalyzed coupling of M1 with carbazole, followed by cyclization to form the indoloacridine core.
- Formation of the spiro-core M3 by reacting M2 with 3-bromofluorenone using n-butyllithium at -78°C, followed by acid-catalyzed dehydration.
- Final functionalization via Suzuki or Buchwald-Hartwig coupling of the brominated spiro-intermediate M3 with various aryl boronic acids or amines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this new class of materials offers distinct strategic advantages beyond mere performance metrics. The synthetic route utilizes widely available commodity chemicals such as carbazole, fluorenone, and simple halobenzenes, which mitigates the risk of raw material shortages often associated with exotic heterocycles. Furthermore, the reliance on established catalytic processes like Suzuki coupling means that existing manufacturing infrastructure can be adapted with minimal capital expenditure. The high yields reported in the patent examples suggest a streamlined production process that minimizes the need for extensive recycling of unreacted starting materials, thereby reducing the overall cost of goods sold (COGS). By integrating these derivatives into the supply chain, companies can secure a reliable OLED material supplier partnership that ensures continuity of supply for high-volume display production.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for rare or prohibitively expensive metal catalysts in the final steps, relying instead on standard palladium systems that can be recovered and recycled. The high conversion rates observed in the lithiation and coupling steps mean that less solvent and energy are required per kilogram of product, leading to substantial cost savings in utility and waste disposal. Additionally, the structural rigidity of the spiro-compounds often simplifies the purification process, as they tend to have distinct solubility profiles compared to linear byproducts, allowing for efficient recrystallization rather than resource-intensive chromatography.
- Enhanced Supply Chain Reliability: Since the precursors are derived from bulk petrochemical feedstocks, the supply chain is less vulnerable to the bottlenecks that plague specialty fine chemical markets. The robustness of the synthetic route, which tolerates a range of reaction conditions without significant degradation of yield, ensures consistent batch-to-batch quality. This reliability is critical for maintaining the tight production schedules of panel manufacturers, reducing the lead time for high-purity electronic chemicals and preventing costly downtime in fabrication lines.
- Scalability and Environmental Compliance: The processes described avoid the use of highly toxic reagents where possible, favoring safer alternatives like potassium carbonate over stronger, more hazardous bases in certain steps. The ability to scale these reactions from gram to kilogram scale without losing efficiency demonstrates the commercial scale-up of complex OLED materials is feasible. Moreover, the high atom economy of the coupling reactions aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process and facilitating compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of fluorene spirophenyl tricarbazole derivatives in OLED stacks. These insights are derived directly from the experimental data and performance metrics provided in the patent documentation, offering clarity on how these materials compare to existing standards and how they can be integrated into current device architectures.
Q: What is the key advantage of fluorene spirophenyl tricarbazole derivatives over traditional hosts like mCP?
A: These derivatives exhibit a significantly higher triplet energy level of up to 2.85 eV, which effectively suppresses energy loss and reduces efficiency roll-off at high brightness levels compared to conventional mCP hosts.
Q: How does the spiro-structure contribute to device performance?
A: The spiro-linkage interrupts the conjugation of the entire molecule, which helps maintain a high triplet energy level while providing good thermal stability and morphological stability essential for long device lifetime.
Q: Are the synthetic routes scalable for industrial production?
A: Yes, the synthesis relies on standard industrial reactions such as Suzuki coupling and nucleophilic substitution, utilizing commercially available starting materials like carbazole and fluorenone, ensuring feasible commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene Spirophenyl Tricarbazole Derivative Supplier
The technical potential of the fluorene spirophenyl tricarbazole platform represents a significant leap forward in OLED host technology, offering a solution to the persistent issue of efficiency roll-off in high-brightness applications. As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that these complex molecular architectures can be delivered with the consistency required by the electronics industry. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards necessary for high-performance display manufacturing, minimizing defects and maximizing yield in your final devices.
We invite you to engage with our technical procurement team to discuss how these advanced materials can optimize your current product lines. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to this novel host system can impact your overall manufacturing economics. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a seamless transition to next-generation materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
