Advanced Synthesis of (R)-4-Propyl Dihydrofuran-2-one for High-Purity Brivaracetam Manufacturing
Introduction to Next-Generation Chiral Synthesis
The pharmaceutical industry's relentless pursuit of safer and more effective antiepileptic treatments has placed significant demand on the supply chain for high-purity intermediates, specifically for the synthesis of Brivaracetam. A pivotal breakthrough in this domain is detailed in patent CN112062740A, which discloses a robust and novel method for synthesizing (R)-4-propyl dihydrofuran-2-one. This compound serves as a critical chiral building block, and its efficient production is paramount for the downstream manufacturing of active pharmaceutical ingredients (APIs). The disclosed technology addresses long-standing challenges regarding cost, scalability, and optical purity that have plagued previous synthetic methodologies. By leveraging a unique combination of Meldrum's acid and (R)-epichlorohydrin, the inventors have established a pathway that not only simplifies the reaction sequence but also ensures exceptional stereochemical control.
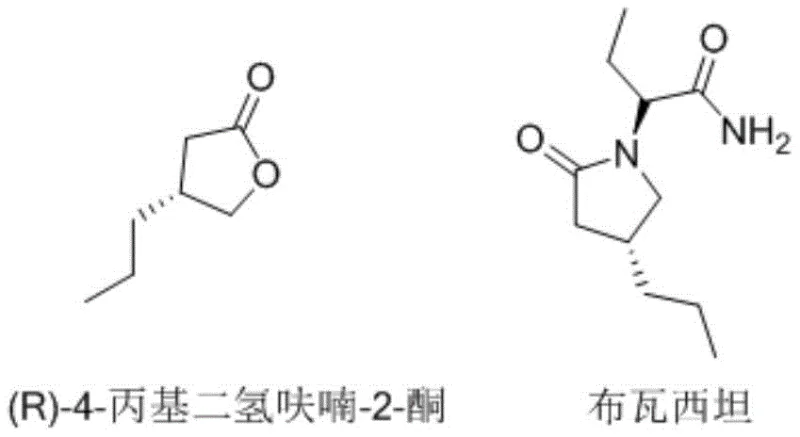
For R&D directors and procurement specialists, understanding the structural relationship between this intermediate and the final drug is crucial. As illustrated in the chemical structures, the integrity of the chiral center in the lactone ring is preserved throughout the synthesis, directly influencing the efficacy of the final therapeutic agent. The patent highlights a total molar yield exceeding 50%, a figure that represents a substantial improvement over many legacy processes which often suffer from cumulative yield losses across multiple steps. This technical advancement positions the material as a viable candidate for reliable pharmaceutical intermediate supplier networks aiming to secure stable, high-quality feedstock for global drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-4-propyl dihydrofuran-2-one has been fraught with significant technical and economic hurdles. One of the earliest approaches, often referred to as Route 1, relied heavily on the use of potassium cyanide (KCN) to extend the carbon chain of chiral epichlorohydrin. While this method utilized a cheap chiral source, the reliance on cyanide introduces severe safety hazards, requiring specialized containment facilities and complex waste treatment protocols that drastically inflate operational costs. Furthermore, experimental data indicated that maintaining high chiral purity through repeated recrystallizations in this route was difficult, often resulting in suboptimal enantiomeric excess values that are unacceptable for modern GMP manufacturing.
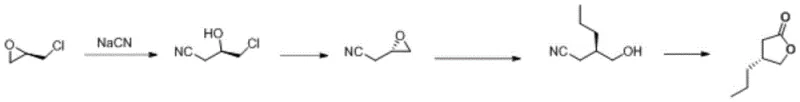
Other historical routes presented their own distinct bottlenecks. For instance, enzymatic kinetic resolution strategies (Route 2) offered better selectivity but were hampered by low catalytic capacity and the necessity of harsh reduction conditions in subsequent steps, limiting their throughput. Similarly, routes employing chiral auxiliary groups (Route 3) achieved good optical purity but suffered from excessive step counts and the economic burden of recovering and recycling expensive auxiliary reagents. Even the more advanced Route 4, which utilized dimethyl sulfoxide and lithium chloride for hydrolysis, struggled with high reaction temperatures and yields that barely met the threshold for commercial viability, often failing to reach the >99.9% purity standards demanded by top-tier pharmaceutical clients.
The Novel Approach
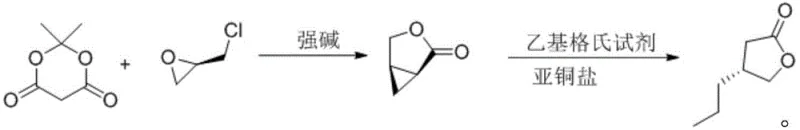
In stark contrast to these legacy methods, the novel approach described in CN112062740A revolutionizes the synthesis by adopting a concise two-step strategy centered around a bicyclic intermediate. The process initiates with the reaction of Meldrum's acid and (R)-epichlorohydrin under basic conditions to form (1S,5R)-3-oxabicyclo[3.1.0]hexan-2-one. This cyclization step is highly efficient and sets the stereochemistry early in the sequence. The subsequent step involves a copper-catalyzed ring-opening reaction with a Grignard reagent, which installs the propyl group with high fidelity. This methodology eliminates the need for toxic cyanides, expensive enzymes, or cumbersome chiral auxiliaries, thereby streamlining the entire production workflow.
The strategic advantage of this new route lies in its simplicity and robustness. By reducing the number of isolation steps and utilizing common industrial reagents, the process inherently lowers the barrier to entry for scale-up. The avoidance of extreme conditions, such as the high temperatures required in Route 4, further enhances the safety profile and energy efficiency of the manufacturing process. For supply chain heads, this translates to a more resilient production capability that is less susceptible to disruptions caused by the scarcity of specialized reagents or the regulatory complexities associated with hazardous chemical handling.
Mechanistic Insights into Copper-Catalyzed Grignard Ring Opening
The core of this synthetic innovation lies in the precise mechanistic control exerted during the second step of the reaction sequence. The transformation of the bicyclic lactone intermediate into the final (R)-4-propyl dihydrofuran-2-one is mediated by a copper catalyst, typically cuprous iodide (CuI), in the presence of an ethyl Grignard reagent. This reaction is a classic example of a conjugate addition or 1,4-addition, but in this specific bicyclic system, it functions as a regioselective and stereoselective ring-opening event. The copper species coordinates with the carbonyl oxygen and the alkene-like character of the strained ring system, activating it for nucleophilic attack by the ethyl group from the Grignard reagent.
The stereochemical outcome is dictated by the rigid geometry of the (1S,5R)-3-oxabicyclo[3.1.0]hexan-2-one intermediate. The approach of the nucleophile is sterically guided by the bridgehead protons and the oxygen atom, ensuring that the ethyl group adds to the specific face of the molecule required to generate the (R)-configuration at the C4 position. The use of a copper salt is critical here; without it, Grignard reagents might attack the carbonyl carbon directly (1,2-addition) or cause non-selective ring opening, leading to a mixture of byproducts. The patent specifies that maintaining the reaction temperature between -40°C and -30°C is essential to suppress side reactions and maximize the formation of the desired product. This low-temperature protocol, combined with the specific choice of catalyst, ensures that the optical purity remains exceptionally high, with ee values consistently reported above 99%.
Furthermore, the mechanism allows for excellent impurity control. Since the starting bicyclic intermediate is formed with high diastereoselectivity in the first step, and the second step proceeds with high stereospecificity, the generation of diastereomeric impurities is minimized. This reduces the burden on downstream purification, often allowing the final product to be isolated simply by distillation rather than requiring costly and wasteful chromatographic separation. For quality assurance teams, this mechanistic clarity provides confidence in the consistency of the batch-to-batch quality, a key requirement for regulatory filings.
How to Synthesize (R)-4-Propyl Dihydrofuran-2-one Efficiently
Implementing this synthesis in a pilot or commercial plant requires strict adherence to the optimized parameters outlined in the patent examples. The process is divided into two distinct stages: the formation of the chiral bicyclic precursor and the subsequent Grignard coupling. The first stage involves the deprotonation of Meldrum's acid with a strong base like sodium ethoxide in ethanol, followed by the addition of (R)-epichlorohydrin. This step requires careful temperature management, starting at 0-5°C and ramping up to reflux to ensure complete conversion while minimizing racemization. The second stage demands anhydrous conditions and inert atmosphere protection (nitrogen) to prevent the decomposition of the Grignard reagent. The detailed standardized operating procedures for these steps, including specific stoichiometric ratios and workup protocols, are critical for achieving the reported yields and purity levels.
- React Meldrum's acid with (R)-epichlorohydrin in the presence of a strong base like sodium ethoxide to form the chiral bicyclic intermediate (1S,5R)-3-oxabicyclo[3.1.0]hexan-2-one.
- Perform a stereoselective ring-opening reaction by adding ethyl magnesium bromide to the intermediate solution containing a copper catalyst (e.g., cuprous iodide) at low temperatures (-40 to -30°C).
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, and purify the crude product via reduced pressure distillation to obtain the final ketone with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers transformative benefits for procurement managers and supply chain strategists. The primary driver of value is the drastic simplification of the supply chain for raw materials. By replacing hazardous cyanides and specialized enzymes with commodity chemicals like Meldrum's acid, epichlorohydrin, and ethyl magnesium bromide, manufacturers can source inputs from a broader, more competitive vendor base. This diversification mitigates the risk of supply shortages and price volatility often associated with niche reagents. Additionally, the elimination of toxic cyanide handling removes the need for expensive dedicated infrastructure and complex environmental compliance measures, leading to significant capital expenditure (CAPEX) and operational expenditure (OPEX) savings.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted. Firstly, the overall yield of over 50% across just two steps is highly competitive compared to multi-step routes where yields compound negatively. Secondly, the avoidance of chiral auxiliaries means there is no cost associated with purchasing, attaching, and recovering these expensive molecules. Thirdly, the purification strategy relies on distillation rather than column chromatography, which significantly reduces solvent consumption and waste disposal costs. These factors collectively contribute to a lower cost of goods sold (COGS), enabling more aggressive pricing strategies in the competitive API intermediate market without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical manufacturers. This route enhances reliability by utilizing robust chemistry that is less sensitive to minor fluctuations in reaction conditions compared to enzymatic processes. The reagents used are stable and widely available globally, reducing lead times for raw material procurement. Furthermore, the process generates fewer hazardous byproducts, simplifying logistics and storage requirements. The ability to produce high-purity material consistently ensures that downstream API synthesis is not delayed by quality failures, thereby stabilizing the entire production schedule and improving on-time delivery performance to customers.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden bottlenecks, but this methodology is inherently scalable. The exothermic nature of the Grignard addition is manageable with standard cooling jackets, and the workup involves simple liquid-liquid extractions that translate easily from laboratory to plant scale. From an environmental standpoint, the process aligns with green chemistry principles by avoiding heavy metals (other than catalytic copper which is removed in workup) and persistent organic pollutants. The reduced waste stream and lower energy consumption (no cryogenic temperatures below -40°C or extreme heating) facilitate easier permitting and compliance with increasingly stringent environmental regulations, future-proofing the manufacturing asset.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology, we have compiled answers to common questions regarding the practical implementation and performance of this synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these nuances is vital for making informed decisions about process adoption and vendor qualification.
Q: What is the optical purity achieved with this new synthesis method?
A: The patented process consistently achieves an enantiomeric excess (ee) value of greater than 99%, with specific examples demonstrating up to 99.6% ee, meeting the stringent requirements for pharmaceutical grade intermediates.
Q: How does this route improve safety compared to traditional cyanide-based methods?
A: Unlike conventional Route 1 which relies on highly toxic potassium cyanide (KCN) for chain extension, this novel method utilizes Meldrum's acid and Grignard reagents, eliminating the need for hazardous cyanide handling and significantly improving workplace safety and waste disposal protocols.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It uses readily available starting materials like (R)-epichlorohydrin and employs standard unit operations such as reflux, extraction, and distillation, avoiding complex chromatographic separations or expensive enzymatic resolutions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-4-Propyl Dihydrofuran-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving medications like Brivaracetam. Our technical team has thoroughly analyzed the synthesis method described in CN112062740A and possesses the expertise to execute this route with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercial launch. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, including the high optical purity (>99% ee) demanded by this specific application.
We invite you to collaborate with us to optimize your supply chain for this key intermediate. Our engineering team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our implementation of this novel route can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities. Let us be your partner in delivering excellence in pharmaceutical manufacturing.
