Scalable Synthesis of Novel Oxadiazole Hydrazide Intermediates for Next-Generation Fungicides
The agricultural sector faces a persistent challenge in managing fungal pathogens that threaten crop yields and food security, necessitating the continuous development of novel active ingredients with distinct modes of action. Patent CN114409611A discloses a significant advancement in this field by introducing a class of substituted benzyl and phenoxy(thio)methylene 1,3,4-oxadiazole hydrazide compounds. These molecules represent a strategic evolution in agrochemical design, merging the broad-spectrum biological activity of the 1,3,4-oxadiazole scaffold with the potent pesticidal properties of the hydrazide motif. 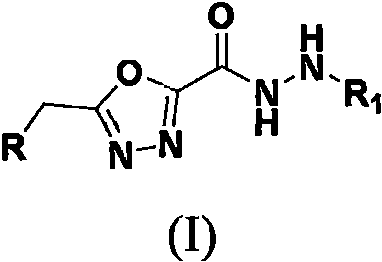 For procurement and R&D teams, this technology offers a robust platform for generating high-value intermediates. The structural versatility allows for extensive SAR (Structure-Activity Relationship) exploration, targeting resistant strains of pathogens such as Gibberella zeae and Sclerotinia sclerotiorum, thereby addressing the critical industry need for resistance management solutions.
For procurement and R&D teams, this technology offers a robust platform for generating high-value intermediates. The structural versatility allows for extensive SAR (Structure-Activity Relationship) exploration, targeting resistant strains of pathogens such as Gibberella zeae and Sclerotinia sclerotiorum, thereby addressing the critical industry need for resistance management solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing complex heterocyclic pesticides often suffer from significant inefficiencies that impact both cost and environmental compliance. Conventional methods for synthesizing oxadiazole derivatives frequently rely on harsh dehydrating agents or multi-step sequences that require rigorous anhydrous conditions and extended reaction times. These processes often result in lower overall yields due to side reactions and the formation of difficult-to-remove impurities, which complicates downstream purification. Furthermore, the reliance on volatile organic solvents and stoichiometric amounts of corrosive reagents increases the operational expenditure related to waste treatment and safety protocols. For a reliable agrochemical intermediate supplier, these factors translate into higher production costs and longer lead times, creating bottlenecks in the supply chain for active pharmaceutical and agricultural ingredients.
The Novel Approach
The methodology outlined in the patent data presents a streamlined and efficient alternative that overcomes these historical hurdles through innovative catalytic strategies. By employing phosphorus oxychloride (POCl3) for the initial cyclization and utilizing an ionic liquid, specifically 1-butyl-3-methylimidazolium chloride, for the subsequent amidation step, the process achieves remarkable improvements in reaction kinetics and selectivity. This approach not only reduces the reaction temperature requirements but also simplifies the workup procedure, as the ionic liquid can potentially be recovered and reused. The result is a cleaner reaction profile with fewer byproducts, directly enhancing the purity of the final crude product. This technological leap enables cost reduction in agrochemical intermediate manufacturing by minimizing raw material consumption and reducing the burden on waste management infrastructure.
Mechanistic Insights into POCl3-Mediated Cyclization and Ionic Liquid Catalysis
The core of this synthetic innovation lies in the precise control of the heterocyclic ring formation and the subsequent nucleophilic substitution. The first stage involves the cyclodehydration of substituted phenylacethydrazides with methyl chlorooxalate (or equivalent activated species) mediated by POCl3. This reagent acts as both a solvent and a dehydrating agent, driving the equilibrium towards the formation of the 1,3,4-oxadiazole ring by sequestering water molecules generated during the condensation. The second stage is particularly noteworthy for its use of green chemistry principles; the ionic liquid serves as a dual solvent-catalyst system. It stabilizes the transition state of the nucleophilic attack by the hydrazine on the ester carbonyl, lowering the activation energy required for the amide bond formation. 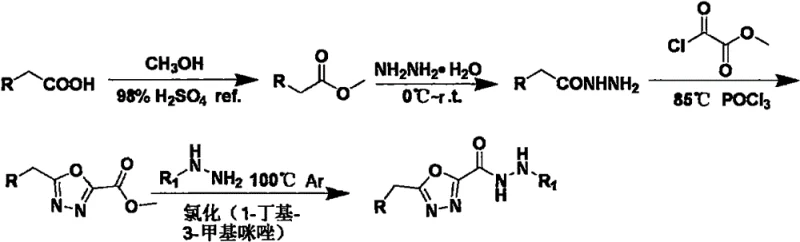 This mechanistic pathway ensures high regioselectivity and prevents the hydrolysis of the sensitive oxadiazole ring, which is a common failure mode in aqueous or protic solvent systems.
This mechanistic pathway ensures high regioselectivity and prevents the hydrolysis of the sensitive oxadiazole ring, which is a common failure mode in aqueous or protic solvent systems.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity oxadiazole hydrazide production. The use of POCl3 ensures complete conversion of the hydrazide starting material, minimizing the risk of unreacted amine impurities that could interfere with biological testing or formulation stability. Additionally, the ionic liquid medium suppresses competing hydrolysis reactions that typically generate carboxylic acid byproducts. The resulting product profile is characterized by a narrow impurity spectrum, primarily consisting of unreacted hydrazine which is easily removed during the aqueous wash steps. This level of chemical fidelity is crucial for meeting the stringent purity specifications required by regulatory bodies for new pesticide registrations, reducing the need for resource-intensive chromatographic purification.
How to Synthesize Oxadiazole Hydrazide Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and reproducibility. The process begins with the preparation of the oxadiazole ester intermediate, followed by a direct coupling with various substituted hydrazines. This modular approach allows for the rapid generation of diverse libraries of compounds for biological screening. The detailed standardized synthesis steps below outline the specific conditions, reagent ratios, and workup procedures necessary to achieve the reported yields and purity levels, serving as a foundational guide for process chemists looking to replicate or optimize this route for commercial purposes.
- Cyclization of substituted phenylacethydrazide with methyl chlorooxalate using POCl3 at 75°C to form the oxadiazole ester intermediate.
- Reaction of the oxadiazole ester intermediate with substituted hydrazines in the presence of 1-butyl-3-methylimidazolium chloride at 100°C under argon.
- Purification via aqueous workup, drying, and recrystallization from ethanol to obtain the final hydrazide product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemical efficacy. The process is designed with scalability in mind, utilizing reagents and equipment that are standard in fine chemical manufacturing facilities. This compatibility means that existing production lines can be adapted with minimal capital expenditure, ensuring a faster time-to-market for new products derived from this scaffold. The robustness of the reaction conditions reduces the risk of batch failures, thereby enhancing supply chain reliability and ensuring consistent delivery schedules for downstream formulators.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of recyclable ionic liquids significantly lower the raw material costs per kilogram of product. Furthermore, the simplified purification process reduces solvent consumption and energy usage associated with distillation and drying, leading to substantial operational savings. By minimizing the generation of hazardous waste, the facility also avoids high disposal fees, contributing to a more favorable cost structure for high-volume production.
- Enhanced Supply Chain Reliability: The starting materials, such as substituted phenylacetic acids and hydrazines, are commodity chemicals available from multiple global sources. This diversity in the supply base mitigates the risk of shortages or price volatility associated with single-source specialty reagents. The robust nature of the synthesis ensures that production can be maintained even if minor variations in raw material quality occur, providing a buffer against supply chain disruptions.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic or regulated substances that would trigger complex environmental permitting processes. The ability to run reactions at moderate temperatures (75°C to 100°C) reduces energy consumption and thermal hazards, making it safer to scale from 100 kgs to 100 MT/annual commercial production. This alignment with green chemistry principles supports corporate sustainability goals and simplifies regulatory compliance in jurisdictions with strict environmental laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these oxadiazole hydrazide derivatives. The answers are derived from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and process capabilities. Understanding these details is essential for partners evaluating the feasibility of integrating these intermediates into their own product pipelines or formulation strategies.
Q: What are the key advantages of using ionic liquids in this synthesis?
A: The use of 1-butyl-3-methylimidazolium chloride as a catalyst significantly enhances reaction rates and yields under milder conditions compared to traditional solvents, while also facilitating easier product separation and potential catalyst recycling.
Q: How does the biological activity compare to commercial standards?
A: Specific derivatives, such as Compound 18, demonstrated superior fungicidal activity against Gibberella zeae with an EC50 of 0.45 μg/mL, outperforming the commercial standard carbendazim (0.95 μg/mL).
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the process utilizes standard unit operations like reflux, distillation, and crystallization, avoiding exotic reagents or extreme pressures, which makes it highly amenable to commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxadiazole Hydrazide Supplier
As the global demand for high-efficacy agrochemicals continues to rise, having a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of oxadiazole hydrazide intermediate meets the highest industry standards for potency and safety.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can optimize your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing decisions, helping you secure a competitive advantage in the dynamic agrochemical market.
