Scalable Synthesis of Double-Chiral Carbazole Derivatives for Next-Generation Agrochemicals
Scalable Synthesis of Double-Chiral Carbazole Derivatives for Next-Generation Agrochemicals
The agricultural sector faces escalating challenges from resistant bacterial pathogens, necessitating the development of novel, high-efficacy active ingredients. Patent CN112624962A introduces a groundbreaking class of carbazolyl isopropanolamine derivatives featuring double chiral centers, designed specifically to combat devastating crop diseases such as rice bacterial leaf blight and citrus canker. This technology represents a significant leap forward in agrochemical intermediate design, leveraging precise stereochemical control to maximize biological potency while maintaining a synthetic route that is robust and commercially viable. By integrating chiral epichlorohydrin and chiral amines into the carbazole scaffold, the invention achieves a level of molecular precision that traditional racemic syntheses cannot match, offering a compelling value proposition for manufacturers seeking next-generation bactericidal solutions.
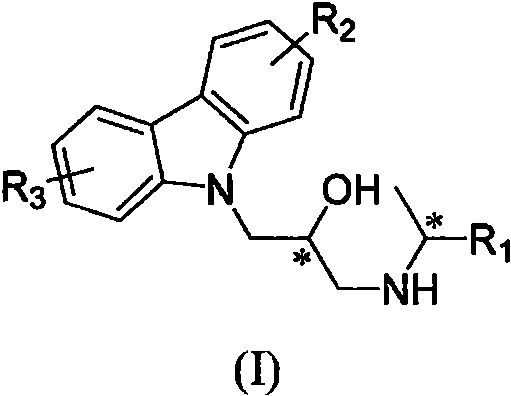
For procurement specialists and supply chain directors, the strategic importance of this patent lies in its balance of high performance and manufacturability. The core structure allows for extensive derivatization at the R1, R2, and R3 positions, enabling a broad portfolio of potential products from a single platform technology. This flexibility is crucial for a reliable agrochemical intermediate supplier aiming to diversify their catalog without overhauling their entire production infrastructure. Furthermore, the documented efficacy against multiple high-value targets, including kiwifruit canker and wheat scab, suggests a wide market applicability that can mitigate supply chain risks associated with single-crop dependency. The ability to produce these complex molecules with high yields and defined stereochemistry positions this technology as a cornerstone for future crop protection strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing carbazole-based pesticides often rely on racemic mixtures or single-step functionalizations that lack stereochemical definition. These conventional approaches frequently suffer from lower biological activity because only one enantiomer typically possesses the desired interaction with the pathogen's biological target, while the other may be inactive or even detrimental. Additionally, older synthetic routes often require harsh reaction conditions, such as high temperatures or the use of toxic heavy metal catalysts, which complicate waste treatment and increase the overall environmental footprint of the manufacturing process. The purification of racemic mixtures to isolate the active enantiomer is also notoriously difficult and costly, involving complex resolution steps that drastically reduce overall yield and inflate production costs, making the final active ingredient less competitive in the global market.
The Novel Approach
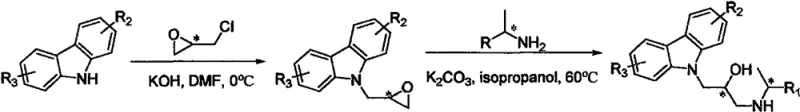
The novel approach detailed in the patent overcomes these hurdles by employing a convergent synthesis strategy that installs chirality early in the process using commercially available chiral building blocks. By reacting carbazole with chiral epichlorohydrin under mild basic conditions, the method efficiently constructs the key epoxy intermediate with high optical purity. This is followed by a regioselective ring-opening with a chiral amine, which simultaneously establishes the second chiral center and the critical isopropanolamine linkage. This two-step sequence avoids the need for post-synthetic resolution, thereby streamlining the workflow and significantly improving atom economy. The use of inexpensive inorganic bases like potassium carbonate and common solvents like isopropanol further underscores the practical advantages of this method, facilitating a smoother transition from laboratory discovery to industrial application.
Mechanistic Insights into Stereoselective Epoxide Ring-Opening
The chemical elegance of this synthesis lies in the mechanistic control exerted during the nucleophilic ring-opening of the epoxide intermediate. In the first stage, the deprotonated carbazole nitrogen acts as a strong nucleophile, attacking the less hindered carbon of the chiral epichlorohydrin in an SN2 fashion. This step is critical as it transfers the chirality from the starting epichlorohydrin to the intermediate without racemization, provided the reaction temperature is carefully controlled, typically around 0°C in DMF. The resulting intermediate retains the epoxide functionality, which serves as a versatile electrophile for the subsequent transformation. The preservation of stereochemical integrity at this stage is paramount, as any loss of optical purity would compromise the efficacy of the final double-chiral product, highlighting the importance of precise process control in the manufacturing environment.
In the second stage, the chiral amine attacks the epoxide ring of the intermediate, a process catalyzed by weak bases like potassium carbonate in isopropanol at moderate temperatures (60°C). This ring-opening reaction proceeds with inversion of configuration at the attack site, allowing for the predictable construction of the second chiral center relative to the first. The hydroxyl group generated during this step not only completes the isopropanolamine motif but also provides a hydrogen-bonding donor that is often essential for the compound's biological activity against bacterial enzymes. The synergy between the rigid carbazole core and the flexible, chiral side chain creates a unique three-dimensional architecture that fits precisely into the active sites of target pathogens, explaining the observed high potency and selectivity reported in the biological assays.
How to Synthesize Carbazolyl Isopropanolamine Derivatives Efficiently
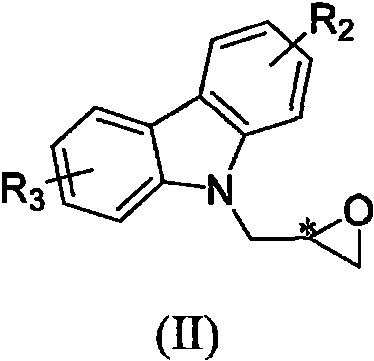
The synthesis of these high-value agrochemical intermediates follows a streamlined two-step protocol that balances reaction efficiency with ease of operation. The process begins with the N-alkylation of the carbazole nucleus, followed by the stereoselective coupling with a chiral amine. This methodology is designed to minimize side reactions and maximize the recovery of the desired stereoisomer, making it ideal for both pilot-scale validation and full commercial production. Operators should pay close attention to the stoichiometry of the base and the temperature profiles during the alkylation step to ensure complete conversion without degrading the sensitive epoxide moiety. The detailed standardized synthesis steps for implementing this route in a GMP-compliant facility are outlined below.
- Perform N-alkylation of carbazole with chiral epichlorohydrin using KOH in DMF at 0°C to form the epoxy intermediate.
- Conduct nucleophilic ring-opening of the epoxy intermediate with a chiral amine using K2CO3 in isopropanol at 60°C.
- Purify the final double-chiral product via column chromatography to achieve high optical purity and yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages in terms of cost structure and supply chain resilience. The reliance on commodity chemicals such as carbazole, epichlorohydrin, and simple chiral amines ensures that raw material availability is not a bottleneck, even during periods of global supply chain disruption. The elimination of expensive transition metal catalysts removes the need for complex and costly metal scavenging steps, which are often required to meet strict regulatory limits on residual metals in agricultural products. This simplification of the downstream processing directly translates to reduced operational expenditures and a faster time-to-market for new formulations. Furthermore, the high yields reported in the patent examples suggest that the process is materially efficient, reducing the volume of waste generated per kilogram of product and aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The process utilizes inexpensive inorganic bases like KOH and K2CO3 instead of costly organometallic reagents, significantly lowering the direct material costs associated with the synthesis. By avoiding the need for chiral resolution of racemic mixtures, the method effectively doubles the theoretical yield of the active isomer compared to traditional resolution techniques, providing a massive boost in overall process efficiency. The use of common solvents like DMF and isopropanol, which are easily recovered and recycled in standard distillation units, further contributes to long-term cost savings and reduces the dependency on specialized solvent supply chains.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are bulk commodities produced by multiple global suppliers, mitigating the risk of single-source dependency that often plagues specialty chemical manufacturing. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without significant loss of yield or purity, ensures consistent production output even in facilities with varying levels of equipment sophistication. This operational flexibility allows for decentralized manufacturing strategies, enabling regional production hubs to serve local markets more effectively and reducing the logistical burden and lead times associated with long-distance transportation of hazardous intermediates.
- Scalability and Environmental Compliance: The reaction pathway generates minimal hazardous byproducts, primarily inorganic salts that can be easily separated and disposed of according to standard environmental protocols. The absence of heavy metals simplifies the wastewater treatment process, reducing the capital investment required for effluent treatment plants and ensuring compliance with increasingly stringent environmental regulations. The exothermic nature of the alkylation step is manageable with standard cooling systems, and the subsequent ring-opening reaction proceeds at moderate temperatures, making the process inherently safer and easier to scale from kilogram to multi-ton quantities without requiring exotic high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this technology into existing product pipelines or developing new formulations that leverage the unique properties of double-chiral centers.
Q: What is the biological significance of the double chiral centers in these carbazole derivatives?
A: The introduction of double chiral centers significantly enhances the stereo-specificity of the molecule, allowing for better binding affinity to bacterial targets. Patent data indicates EC50 values as low as 1.00 µg/mL against rice bacterial leaf blight, demonstrating superior potency compared to racemic mixtures.
Q: Are the reaction conditions suitable for large-scale industrial production?
A: Yes, the process utilizes mild conditions (0°C to 60°C) and common solvents like DMF and isopropanol. The absence of expensive transition metal catalysts and the use of robust inorganic bases like KOH and K2CO3 make the process highly amenable to commercial scale-up with minimal safety risks.
Q: Which plant pathogens show the highest sensitivity to these compounds?
A: The compounds exhibit exceptional activity against Xanthomonas oryzae pv. oryzae (rice bacterial leaf blight), Xanthomonas axonopodis pv. citri (citrus canker), and Pseudomonas syringae pv. actinidiae (kiwifruit canker), addressing critical gaps in current agricultural disease management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazolyl Isopropanolamine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing the technical expertise to translate complex patent methodologies like CN112624962A into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the optical purity and chemical identity of every batch, meeting the exacting standards required by global agrochemical leaders.
We invite you to collaborate with us to unlock the full potential of these high-performance bactericidal intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timeline and secure a competitive advantage in the rapidly evolving crop protection market.
