Revolutionizing Herbicide Intermediate Production via Optimized Uracil-First Synthesis
Revolutionizing Herbicide Intermediate Production via Optimized Uracil-First Synthesis
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for complex heterocyclic structures. Patent CN113880774A introduces a groundbreaking preparation method for phenylisoxazoline compounds, a class of molecules exhibiting potent herbicidal activity against a broad spectrum of weeds including cockspur grass and green bristlegrass. This technical disclosure represents a significant leap forward in process chemistry, shifting the synthetic paradigm to prioritize the formation of the uracil ring before constructing the isoxazoline moiety. By fundamentally reordering the synthetic sequence, this methodology addresses critical stability issues inherent in previous generations of synthesis, offering a pathway that is not only chemically robust but also economically superior for large-scale production.
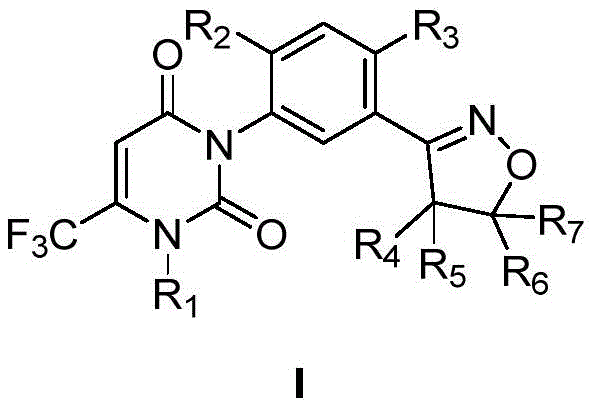
The strategic value of this patent lies in its ability to transform a previously cumbersome multi-step synthesis into a streamlined, high-yielding process suitable for modern industrial application. For R&D directors and procurement specialists alike, understanding the nuances of this uracil-first approach is essential for evaluating potential supply chain partners capable of delivering high-purity agrochemical intermediates. The following analysis dissects the technical merits of this invention, contrasting it with legacy methods to highlight the tangible benefits in cost reduction and operational reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in patent WO2016095768, typically employ a synthetic strategy where the isoxazoline ring is constructed prior to the formation of the uracil ring. While theoretically viable, this conventional sequence suffers from severe practical drawbacks that hinder efficient commercial manufacturing. The primary failure point is the inherent instability of the isoxazoline intermediate when subjected to the rigorous conditions required for subsequent uracil ring closure. These conditions often involve high temperatures and strong alkaline environments, which frequently lead to the decomposition of the sensitive isoxazoline structure, resulting in a complex mixture of byproducts that are difficult to separate.
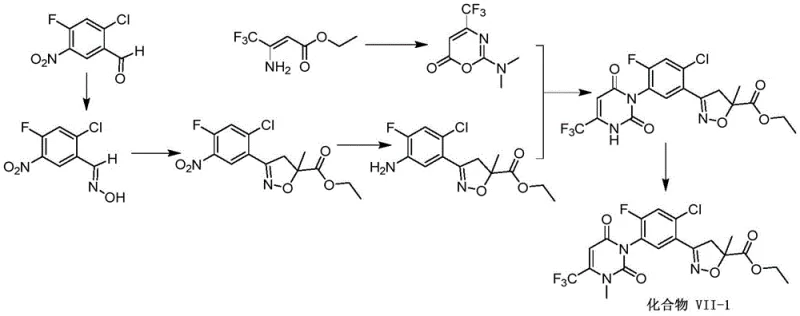
Furthermore, the conventional route often necessitates the use of less accessible or more expensive starting materials, such as nitro-substituted benzaldehydes, which require additional reduction steps to achieve the necessary amine functionality for uracil coupling. This elongation of the synthetic timeline not only increases the consumption of solvents and reagents but also amplifies the cumulative loss of yield at each stage. The combination of low overall yield, often reported around 20% in comparative examples, and the generation of significant chemical waste makes these legacy processes economically unviable for competitive agrochemical intermediate suppliers seeking to optimize cost structures.
The Novel Approach
In stark contrast, the novel approach detailed in CN113880774A ingeniously reverses the synthetic order, establishing the robust uracil scaffold first before introducing the isoxazoline ring. This strategic pivot leverages the chemical stability of the uracil intermediate, allowing it to withstand the subsequent reaction conditions required for isoxazoline formation without degradation. The process begins with the readily available 2-fluoro-4-chloro-5-methylaniline, which is converted into a carbamate and then cyclized with trifluoro-aminocrotonic acid ester to form the uracil core. This foundational step sets the stage for a highly efficient downstream process.
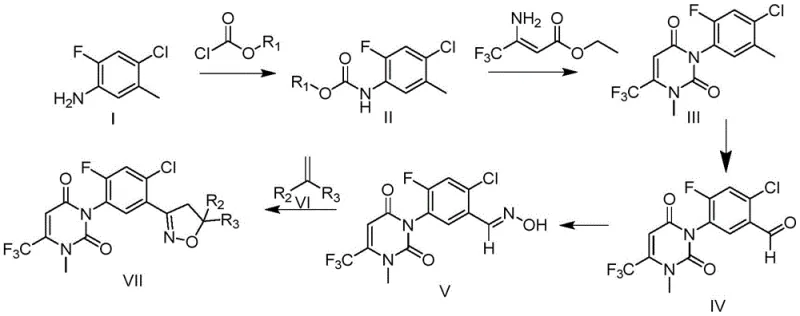
By securing the uracil structure early, the subsequent steps involving oxidation to the aldehyde, oxime formation, and final cyclization with an alkene proceed with remarkable cleanliness and efficiency. The patent data indicates that this reordered pathway can achieve total yields exceeding 60%, representing a threefold improvement over conventional methods. This dramatic increase in efficiency translates directly into substantial cost reduction in herbicide manufacturing, as less raw material is wasted and purification burdens are significantly minimized. For supply chain heads, this reliability ensures a consistent output of high-purity intermediates, mitigating the risks associated with batch-to-batch variability.
Mechanistic Insights into Uracil-First Heterocyclic Construction
The mechanistic elegance of this synthesis lies in the careful management of functional group compatibility and reaction thermodynamics. The initial formation of the uracil ring involves a condensation reaction between the carbamate intermediate and ethyl 3-amino-4,4,4-trifluorocrotonate, facilitated by phase transfer catalysts such as tetrabutylammonium bromide. This catalytic system enhances the nucleophilicity of the reactants in organic solvents, driving the cyclization to completion under reflux conditions. Following this, a methylation step using agents like methyl iodide installs the necessary N-methyl group, stabilizing the nitrogen center for subsequent transformations.
The conversion of the uracil derivative to the corresponding benzaldehyde is a critical junction in the pathway. This is achieved through a controlled halogenation followed by hydrolysis, a sequence that effectively functionalizes the aromatic ring without compromising the integrity of the uracil moiety. The resulting aldehyde is then condensed with hydroxylamine to form the oxime, a pivotal precursor for the isoxazoline ring. The final cyclization step utilizes N-chlorosuccinimide (NCS) to generate a nitrile oxide intermediate in situ, which undergoes a 1,3-dipolar cycloaddition with the selected alkene. This mechanism is highly selective and proceeds under mild conditions, preserving the stereochemical and structural integrity of the final phenylisoxazoline product.
How to Synthesize Phenylisoxazoline Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize the benefits of the uracil-first strategy. The process is designed to be operationally simple, utilizing standard reactor configurations and workup procedures that are familiar to any experienced chemical manufacturing team. From the initial carbamate formation to the final vacuum distillation, each step has been optimized to minimize solvent usage and energy consumption while maximizing throughput.
- React 2-fluoro-4-chloro-5-methylaniline with chloroformate to form a carbamate intermediate under alkaline conditions.
- Cyclize the carbamate with trifluoro-aminocrotonic acid ester and methylate to form the stable uracil core structure.
- Convert the uracil derivative to an aldehyde, form an oxime, and finally cyclize with an alkene via NCS chlorination to complete the isoxazoline ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling advantages that extend beyond mere chemical yield. The shift to a more stable and efficient process directly addresses the perennial challenges of cost volatility and supply continuity in the agrochemical sector. By eliminating the need for unstable intermediates and complex purification protocols, manufacturers can achieve a leaner production model that is both cost-effective and environmentally sustainable.
- Cost Reduction in Manufacturing: The most immediate impact of this technology is the drastic reduction in production costs driven by the tripling of overall yield compared to prior art. By avoiding the losses associated with the decomposition of unstable isoxazoline intermediates, the process ensures that a significantly higher proportion of expensive starting materials are converted into saleable product. Furthermore, the use of commercially available and inexpensive reagents like 2-fluoro-4-chloro-5-methylaniline eliminates the need for custom synthesis of exotic precursors, further driving down the cost of goods sold and enhancing margin potential for downstream herbicide formulations.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions creates a resilient supply chain that is less susceptible to disruptions. Since the raw materials are widely sourced and the process does not depend on scarce or highly specialized catalysts, procurement teams can secure long-term contracts with greater confidence. The robustness of the intermediates also means that inventory can be held at various stages of production without significant risk of degradation, providing flexibility in managing production schedules and meeting urgent customer demands for high-purity agrochemical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as reflux, extraction, and distillation that are easily adapted from pilot plant to multi-ton commercial reactors. The reduction in byproduct formation simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations. This ease of scale-up ensures that suppliers can rapidly respond to market surges in demand for herbicide active ingredients without the lengthy lead times typically associated with process optimization, thereby securing a competitive advantage in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis pathway. These insights are derived directly from the experimental data and process descriptions within the patent, providing a clear picture of what partners can expect when adopting this technology for their manufacturing portfolios.
Q: Why is the uracil-first synthesis route superior to conventional methods?
A: Conventional methods synthesize the isoxazoline ring first, which creates unstable intermediates prone to decomposition during the subsequent harsh uracil ring closure. The novel uracil-first approach ensures intermediate stability, significantly reducing byproduct formation and improving total yield.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes commercially available and cost-effective starting materials, primarily 2-fluoro-4-chloro-5-methylaniline, ethyl chloroformate, and ethyl 3-amino-4,4,4-trifluorocrotonate, ensuring a robust and reliable supply chain.
Q: How does this process impact industrial scalability?
A: The method employs standard unit operations such as reflux, extraction, and distillation without requiring exotic catalysts or extreme conditions. This simplicity facilitates easy scale-up from laboratory to multi-ton commercial production while maintaining high purity standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylisoxazoline Supplier
As the agrochemical industry evolves, the ability to access advanced intermediates through efficient and scalable routes becomes a critical differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in heterocyclic chemistry to deliver solutions that meet the rigorous demands of global pharmaceutical and agrochemical clients. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and reliable.
We invite you to engage with our technical procurement team to discuss how this optimized phenylisoxazoline synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
