Revolutionizing o-Aminobiphenyl Production: A Safe, One-Step Catalytic Strategy for Commercial Scale-Up
Revolutionizing o-Aminobiphenyl Production: A Safe, One-Step Catalytic Strategy for Commercial Scale-Up
The global demand for high-performance agrochemicals, particularly succinate dehydrogenase inhibitor (SDHI) fungicides like bixafen and fluxapyroxad, has placed immense pressure on the supply chains of their key intermediates. Patent CN107488113B introduces a transformative methodology for synthesizing o-aminobiphenyl compounds, addressing critical bottlenecks in yield, safety, and cost. This technology leverages a dual-metal catalytic system to achieve a continuous coupling and nitro reduction sequence in a single reactor vessel. By utilizing readily available o-nitro aromatic formates and substituted halobenzenes, this approach bypasses the complex multi-step preparations associated with traditional boronic acid chemistry. For R&D directors and procurement managers alike, this represents a paradigm shift towards more efficient, safer, and economically viable manufacturing processes for high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
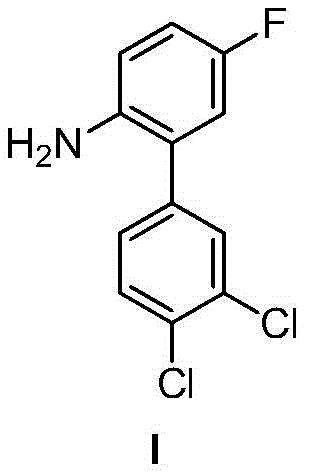
Historically, the synthesis of critical intermediates such as 3',4'-dichloro-2-amino-5-fluorobiphenyl has relied heavily on Suzuki-Miyaura coupling reactions. While effective in laboratory settings, these conventional routes suffer from severe industrial drawbacks. The primary bottleneck lies in the preparation of the requisite aryl boronic acid partners. Synthesizing species like 3,4-dichlorophenylboronic acid typically involves generating Grignard reagents at cryogenic temperatures of -60°C, followed by reaction with trialkylborates. This process is not only energy-intensive but also poses significant safety risks due to the handling of pyrophoric organometallics on a large scale. Furthermore, the subsequent nitro reduction step often necessitates high-pressure hydrogenation equipment and hazardous reducing agents, complicating facility requirements and increasing capital expenditure. Alternative methods involving diazonium salts or hydrazine derivatives frequently result in excessive waste generation and rely on silica gel chromatography for purification, a technique wholly unsuitable for ton-scale production.
The Novel Approach
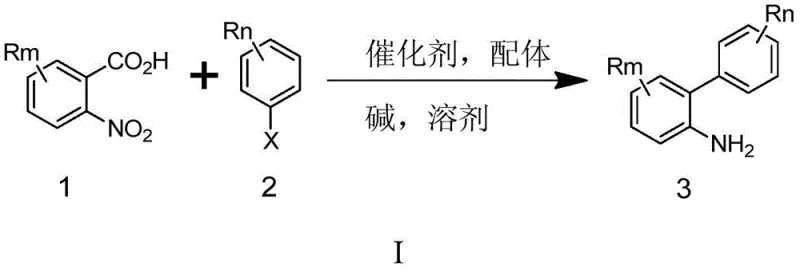
The methodology disclosed in CN107488113B offers a robust alternative by merging the biaryl bond formation and nitro group reduction into a unified operation. Instead of relying on unstable boronic acids, the process utilizes stable o-nitro aromatic carboxylic acid salts, such as potassium 2-nitro-5-fluorobenzoate, which are inexpensive and commercially accessible. The reaction proceeds via a decarboxylative cross-coupling mechanism facilitated by a synergistic palladium and copper catalyst system. Crucially, the reduction of the nitro group occurs simultaneously without external hydrogen gas, utilizing the polyalcohol solvent as an internal hydrogen donor. This innovation drastically simplifies the workflow, eliminating the need for separate reduction reactors and the associated safety infrastructure. The result is a streamlined process that delivers high-purity products with yields reaching up to 96%, effectively solving the purity and scalability issues plaguing older synthetic routes.
Mechanistic Insights into Dual Metal Catalyzed Decarboxylative Coupling
The core of this technological breakthrough lies in the intricate interplay between the palladium and copper catalysts within a high-boiling polar solvent matrix. The reaction initiates with the oxidative addition of the substituted halobenzene to the palladium center. Concurrently, the copper species facilitates the decarboxylation of the o-nitro aromatic formate, generating a reactive aryl-copper intermediate. Transmetallation between the copper and palladium centers forms a diaryl-palladium complex, which subsequently undergoes reductive elimination to forge the biaryl bond. Uniquely, the solvent environment, typically polyethylene glycol (PEG) or similar polyalcohols, plays a dual role: it acts as the reaction medium and serves as the hydrogen source for the reduction of the nitro group to an amine. This tandem transformation ensures that the potentially sensitive nitro-biaryl intermediate is immediately reduced to the stable amine, preventing side reactions and simplifying the impurity profile.
Impurity control is inherently managed by the choice of ligands and the specific reaction conditions. The use of phosphine ligands like triphenylphosphine in combination with nitrogen-containing ligands such as 1,10-phenanthroline stabilizes the catalytic cycle and suppresses homocoupling side reactions. The high temperature range of 190°C to 240°C ensures complete conversion of the starting materials, while the basic conditions provided by carbonates or hydroxides facilitate the initial decarboxylation step. This precise control over the reaction environment allows for the direct crystallization of the final product from the reaction mixture after simple aqueous workup, avoiding the need for complex chromatographic separations. Consequently, the process yields products with purity levels exceeding 98%, meeting the stringent specifications required for pharmaceutical and agrochemical active ingredient synthesis.
How to Synthesize 3',4'-Dichloro-2-amino-5-fluorobiphenyl Efficiently
Implementing this synthesis requires careful attention to the preparation of the nitro-benzoate salt and the selection of the catalyst loading. The process begins with the neutralization of 2-nitro-5-fluorobenzoic acid with potassium hydroxide in ethanol to form the dry potassium salt. This salt is then combined with 3,4-dichlorobromobenzene in a reactor containing anhydrous polyethylene glycol (PEG-400). The catalytic system, comprising palladium acetylacetonate and cuprous iodide along with the appropriate ligand mix, is added under an inert nitrogen atmosphere. The detailed standardized synthesis steps for optimizing yield and purity are outlined in the guide below.
- Prepare the o-nitro aromatic formate salt by reacting the corresponding acid with an alkali metal base in a solvent, followed by water removal.
- Combine the dried formate salt, substituted halobenzene, palladium catalyst (e.g., Pd(acac)2), copper catalyst (e.g., CuI), and ligands in a polyalcohol solvent like PEG-400.
- Heat the mixture to 190°C under nitrogen protection for approximately 22 hours to effect coupling and simultaneous nitro reduction, followed by extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits beyond mere technical elegance. The most significant advantage is the drastic simplification of the raw material supply chain. By eliminating the dependency on custom-synthesized boronic acids, which often have long lead times and volatile pricing due to their complex manufacture, manufacturers can source stable, commodity-grade halobenzenes and nitro-acids. This shift significantly enhances supply chain reliability and reduces exposure to raw material shortages. Furthermore, the removal of high-pressure hydrogenation steps lowers the barrier to entry for contract manufacturing organizations (CMOs) that may lack specialized hydrogenation infrastructure, thereby expanding the pool of potential suppliers and fostering competitive pricing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of multiple unit operations. Traditional routes require separate reactors for Grignard formation, boronic acid synthesis, coupling, and finally, nitro reduction. This one-pot method consolidates these steps, resulting in substantial savings in energy consumption, labor, and equipment depreciation. Additionally, the avoidance of expensive boronic acid precursors and the use of recyclable polyethylene glycol solvents contribute to a significantly lower cost of goods sold (COGS). The high atom economy and reduced waste generation further lower disposal costs, aligning with modern green chemistry mandates.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of robust, shelf-stable starting materials. Unlike Grignard reagents or diazonium salts which must be prepared fresh and handled under strict conditions, the o-nitro aromatic formates used here are stable solids that can be stockpiled. This stability allows for better inventory management and just-in-time manufacturing capabilities. The simplified workflow also reduces the risk of batch failures associated with transferring intermediates between different processing stages, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method is superior. The process generates minimal hazardous waste compared to the heavy metal sludge and acidic waste streams typical of traditional diazonium or Grignard routes. The use of PEG solvents, which are non-volatile and less toxic than traditional organic solvents like THF or DMF, simplifies solvent recovery and reduces VOC emissions. This environmental profile facilitates easier regulatory approval and permits for plant expansion, enabling seamless scale-up from pilot kilograms to multi-ton commercial production without extensive retrofitting of waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this decarboxylative coupling technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on its practical application in an industrial setting.
Q: What are the primary advantages of this method over traditional Suzuki coupling for aminobiphenyls?
A: This method eliminates the need for pre-synthesized boronic acids, which often require harsh Grignard conditions at -60°C. It utilizes commercially available halobenzenes and nitro-benzoic acid salts directly, significantly simplifying the supply chain and reducing raw material costs.
Q: How does this process address safety concerns regarding nitro group reduction?
A: Unlike traditional methods that require high-pressure hydrogenation with dangerous reducing agents, this protocol achieves nitro reduction in situ using the solvent as a hydrogen source. This removes the need for specialized hydrogenation equipment and mitigates explosion risks.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the process uses robust catalysts (Pd/Cu) and high-boiling, recyclable solvents like polyethylene glycol. It avoids column chromatography, relying instead on simple filtration and crystallization, making it highly amenable to multi-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3',4'-Dichloro-2-amino-5-fluorobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN107488113B can be fully realized in your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of o-aminobiphenyl intermediate meets the exacting standards required for next-generation fungicide production.
We invite you to collaborate with us to optimize your sourcing strategy for these critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced catalytic technologies can drive efficiency and reliability in your agrochemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
