Advanced Synthesis of Bioactive Quinoline Schiff Bases for Pharmaceutical Applications
The pharmaceutical landscape is constantly evolving with the demand for novel scaffolds that can address complex biological targets such as cancer and metabolic disorders. Patent CN108358838B introduces a significant advancement in this domain by disclosing a series of novel quinoline-substituted Schiff base derivatives. These compounds are engineered to function as dual inhibitors, targeting both Cell Cycle Division Protein 25B (CDC25B) and Protein Tyrosine Phosphatase 1B (PTP1B). The structural versatility of these molecules allows for extensive modification at the R1, R2, and R3 positions, enabling fine-tuning of their pharmacological profiles. As a reliable pharmaceutical intermediate supplier, understanding the underlying chemistry of such high-value scaffolds is crucial for developing robust supply chains. The core architecture features a quinoline ring linked via a methylene-hydrazono bridge to various aryl or acyl groups, creating a diverse library of potential drug candidates.
The development of these bioactive intermediates represents a strategic opportunity for cost reduction in API manufacturing, particularly for oncology and diabetes therapeutic areas. The patent highlights that the synthesis process is notably simple and cost-effective, avoiding the need for exotic catalysts or extreme reaction conditions that often plague complex heterocyclic synthesis. By leveraging a modular approach, manufacturers can rapidly generate analogues to optimize potency and selectivity. This flexibility is essential for R&D teams aiming to navigate the intricate path from lead identification to clinical candidate selection. Furthermore, the demonstrated inhibitory activities against CDC25B and PTP1B suggest that these intermediates are not merely academic curiosities but viable precursors for next-generation therapeutics with significant commercial potential.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of functionalized quinoline derivatives has been fraught with challenges related to regioselectivity and harsh reaction conditions. Conventional routes often rely on transition metal catalysis which introduces the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet stringent regulatory standards for pharmaceutical ingredients. Additionally, many classical methods suffer from poor atom economy and low yields when introducing sensitive functional groups at the 3- or 4-positions of the quinoline ring. The formation of the Schiff base linkage, while generally straightforward, can be complicated by the instability of the aldehyde precursors or the competing side reactions of the hydrazine components. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for supply chain heads who require consistent and reliable volumes of high-purity intermediates.
The Novel Approach
The methodology outlined in CN108358838B offers a transformative solution by employing a streamlined, multi-step sequence that maximizes yield and minimizes waste. The process begins with a base-catalyzed condensation that efficiently constructs the quinoline core, followed by a logical progression of functional group interconversions. This approach eliminates the need for precious metal catalysts, thereby reducing the environmental footprint and simplifying the downstream processing. The use of readily available starting materials such as isatin derivatives and substituted acetophenones ensures a stable supply of raw materials, mitigating the risk of procurement delays. Moreover, the reaction conditions are mild enough to tolerate a wide range of substituents, allowing for the rapid generation of diverse libraries without compromising the integrity of the molecular scaffold. This robustness is a key factor in enhancing supply chain reliability for complex pharmaceutical intermediates.
Mechanistic Insights into Quinoline Ring Construction and Schiff Base Formation
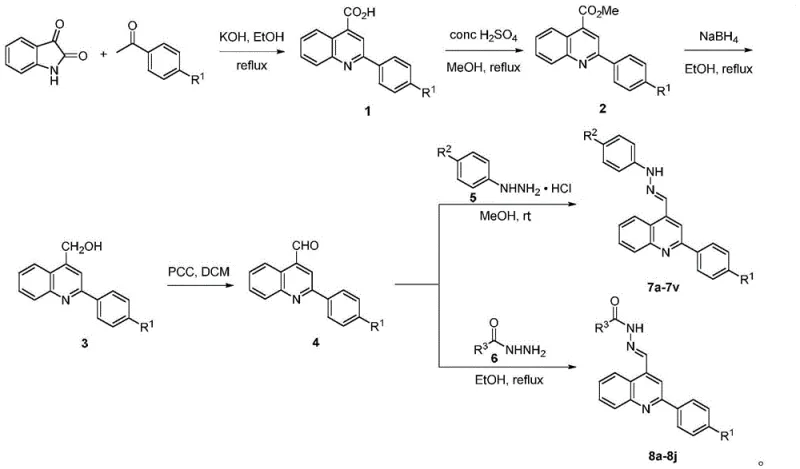
The synthetic strategy relies heavily on the classic Friedländer quinoline synthesis mechanism, adapted here for high efficiency. The initial step involves the condensation of 2,3-indole diketone (isatin) with a substituted acetophenone in the presence of potassium hydroxide. This base-catalyzed reaction facilitates the formation of the carbon-carbon double bond and the subsequent cyclization to form the quinoline-4-carboxylic acid derivative. The mechanistic pathway proceeds through an aldol-type condensation followed by dehydration and aromatization, driven by the thermodynamic stability of the resulting aromatic system. This step is critical as it establishes the core heterocyclic framework upon which all subsequent biological activity depends. The choice of ethanolic KOH provides a homogeneous reaction medium that promotes efficient mixing and heat transfer, ensuring consistent reaction kinetics across different scales of operation.
Following the construction of the quinoline core, the pathway diverges into a series of functional group transformations designed to install the reactive aldehyde handle necessary for Schiff base formation. The carboxylic acid is first esterified to a methyl ester using methanol and concentrated sulfuric acid, a standard Fischer esterification that protects the acid functionality and activates the carbonyl for reduction. Subsequent reduction with sodium borohydride converts the ester into a primary alcohol, which is then selectively oxidized to the corresponding aldehyde using pyridinium chlorochromate (PCC). This aldehyde serves as the electrophilic center for the final condensation with various hydrazines or acylhydrazines. The formation of the carbon-nitrogen double bond (imine or hydrazone linkage) is a reversible equilibrium process that is driven to completion by the removal of water, resulting in the stable Schiff base products depicted in the patent.
How to Synthesize Quinoline Schiff Base Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to ensure optimal yield and purity. The process is divided into distinct stages, each requiring specific attention to stoichiometry, temperature, and workup procedures. For R&D teams looking to replicate or scale this chemistry, understanding the nuances of each transformation is vital. The initial cyclization sets the foundation, while the subsequent redox steps determine the quality of the final coupling partner. Detailed protocols for each stage, including specific molar ratios and solvent choices, are essential for reproducibility. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale production.
- Perform base-catalyzed condensation of 2,3-indole diketone with substituted acetophenone using KOH in ethanol to form the quinoline carboxylic acid core.
- Convert the carboxylic acid to methyl ester using methanol and concentrated sulfuric acid, followed by reduction to the corresponding alcohol using sodium borohydride.
- Oxidize the alcohol to the aldehyde using PCC in DCM, then condense with substituted phenylhydrazines or acylhydrazines to yield the final Schiff base derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in CN108358838B offers compelling advantages that align with the strategic goals of modern pharmaceutical manufacturing. The elimination of expensive transition metal catalysts significantly reduces the raw material costs and simplifies the purification process, leading to substantial cost savings in the overall production budget. This is particularly relevant for procurement managers who are under constant pressure to reduce the cost of goods sold (COGS) without compromising quality. Furthermore, the use of commodity chemicals like KOH, methanol, and sodium borohydride ensures that the supply chain is resilient to market fluctuations, as these reagents are widely available from multiple global suppliers. This diversification of the supply base enhances supply chain reliability and reduces the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts such as palladium or platinum, which are not only expensive but also subject to volatile market pricing. By utilizing base-metal free chemistry, the manufacturing cost is significantly lowered. Additionally, the workup procedures involve simple filtration and washing steps rather than complex extraction or chromatographic separations for every intermediate, which reduces solvent consumption and labor costs. The high yields reported for key steps, such as the reduction of the ester to alcohol (92% yield), further contribute to cost efficiency by maximizing the output from a given amount of starting material.
- Enhanced Supply Chain Reliability: The starting materials, including isatin and various acetophenones, are bulk chemicals produced on a multi-ton scale globally. This abundance ensures a steady and reliable supply, minimizing the lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to consistent batch-to-batch performance. For supply chain heads, this predictability is invaluable for planning production schedules and meeting delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory experiments to kilogram and ton-scale commercial production. The reactions are conducted in common solvents like ethanol and methanol, which are easier to recover and recycle compared to chlorinated solvents, although dichloromethane is used in the oxidation step. The simplicity of the isolation procedures, often involving precipitation and filtration, reduces the generation of liquid waste and simplifies effluent treatment. This alignment with green chemistry principles supports environmental compliance and sustainability goals, which are increasingly important criteria for vendor selection in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quinoline derivatives. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating these intermediates into their development pipelines. The answers reflect the practical realities of scaling this chemistry and the specific biological activities that make these compounds attractive targets for drug discovery.
Q: What are the primary biological targets of these quinoline derivatives?
A: According to patent CN108358838B, these compounds act as potent inhibitors for Cell Cycle Division Protein 25B (CDC25B) and Protein Tyrosine Phosphatase 1B (PTP1B), indicating potential for anticancer and antidiabetic applications.
Q: Is the synthetic route suitable for large-scale manufacturing?
A: Yes, the process utilizes common reagents like KOH, NaBH4, and PCC, and involves standard unit operations such as reflux, filtration, and column chromatography, making it highly adaptable for commercial scale-up.
Q: How is the purity of the final intermediates ensured?
A: The patent describes rigorous purification steps including vacuum filtration, washing with specific solvent systems like petroleum ether and ethyl acetate, and column chromatography to achieve high-purity solids suitable for biological testing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Schiff Base Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from the bench to the market. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific analogues or large-scale supply of the core scaffolds described in CN108358838B, our infrastructure is designed to support your needs with speed and precision.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug discovery programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with us, you gain access to specific COA data and route feasibility assessments that can help you mitigate risks and reduce time-to-market. Contact us today to discuss how we can support your next breakthrough in oncology or metabolic disease research.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
