Scalable Fermentation and Semi-Synthesis of Bioactive Sulfur-containing Chromone Intermediates
Scalable Fermentation and Semi-Synthesis of Bioactive Sulfur-containing Chromone Intermediates
The pharmaceutical industry is constantly seeking novel scaffolds with potent antitumor activities to overcome drug resistance and improve therapeutic outcomes. Patent CN102603769A introduces a groundbreaking class of sulfur-containing chromone compounds derived from the marine-derived fungus Penicillium oxalicum CCTCC AF 93256. Unlike traditional synthetic chromones which often require harsh cyclization conditions, these natural product analogues feature a unique sulfur-bridged five-membered ring fused to the chromone core, offering distinct stereochemical properties and enhanced biological profiles. The patent details a robust biotechnological production method involving large-scale fermentation followed by efficient solvent extraction and chromatographic purification. This approach not only provides access to the parent compound but also enables the semi-synthetic generation of diverse acetylated and Mosher ester derivatives, creating a versatile library of high-purity pharmaceutical intermediates for oncology drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of complex chromone derivatives often relies on multi-step organic transformations that involve toxic reagents, heavy metal catalysts, and extreme temperatures, leading to significant environmental burdens and high production costs. Conventional routes to introduce sulfur moieties into heterocyclic systems frequently suffer from poor regioselectivity and low yields, necessitating extensive purification efforts that delay project timelines. Furthermore, achieving the specific stereochemistry required for optimal biological activity in synthetic chromones can be exceptionally challenging without employing expensive chiral auxiliaries or resolution techniques. These limitations create bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, making it difficult for manufacturers to supply consistent quality materials for preclinical and clinical evaluation. The reliance on petrochemical starting materials also exposes the supply chain to volatility in raw material pricing and availability.
The Novel Approach
The methodology disclosed in CN102603769A leverages the biosynthetic machinery of Penicillium oxalicum to construct the intricate sulfur-chromone skeleton under mild, ambient conditions, effectively bypassing the need for complex chemical cyclization steps. By utilizing a defined PDA (Potato Dextrose Agar) medium supplemented with sea salt, the fermentation process mimics the natural marine environment, triggering the expression of secondary metabolic pathways that produce the target compounds with high stereoselectivity. The downstream processing utilizes standard liquid-liquid extraction with ethyl acetate followed by gradient elution chromatography, which are well-established unit operations easily adaptable to industrial scales. This biotechnological route significantly simplifies the manufacturing workflow, reducing the number of synthetic steps and eliminating the need for hazardous reagents, thereby aligning with green chemistry principles and facilitating cost reduction in API manufacturing. The ability to subsequently derivatize the fermentation products via simple acetylation or esterification further enhances the utility of this platform for generating diverse analogue libraries.
Mechanistic Insights into Biosynthetic Assembly and Structural Elucidation
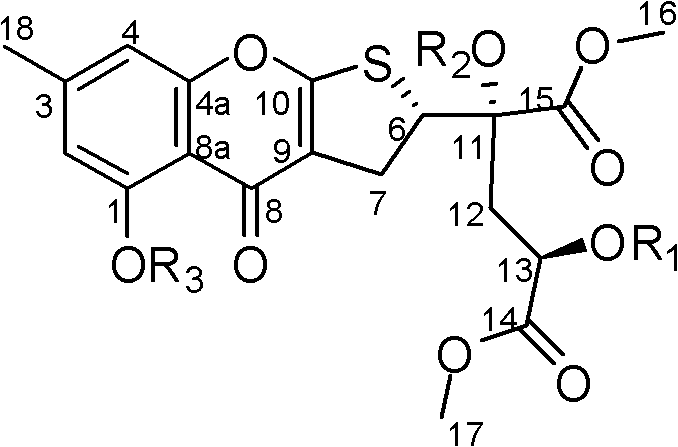
The structural integrity and stereochemistry of these sulfur-containing chromones are critical determinants of their antitumor efficacy. Detailed spectroscopic analysis, including 1D and 2D NMR techniques such as HMBC and COSY, revealed a unique 1,2,3,5-tetrasubstituted benzene ring fused to a sulfur-containing five-membered ring, which is further linked to a substituted methyl dipivalate fragment. The absolute configurations of the chiral centers at C-6, C-11, and C-13 were rigorously assigned as 6S, 11S, and 13R through a combination of NOESY correlations and modified Mosher esterification methods. This precise stereochemical control is inherent to the enzymatic processes within the fungus, ensuring that the produced intermediates possess the correct 3D orientation required for binding to biological targets. Understanding this biosynthetic logic allows chemists to predict the formation of specific impurities and optimize fermentation parameters to maximize the yield of the desired stereoisomer. For R&D teams, this level of structural definition provides a solid foundation for structure-activity relationship (SAR) studies, enabling the rational design of next-generation analogues with improved potency and selectivity profiles.
Impurity control in this bioprocess is managed through the specificity of the fungal metabolism and the selectivity of the purification steps. The patent describes the use of silica gel column chromatography with chloroform-methanol or chloroform-acetone solvent systems to separate the target compounds from other fungal metabolites. The distinct polarity differences between the parent chromones and their acetylated derivatives allow for effective separation during semi-preparative HPLC purification. By monitoring the elution profiles at 254 nm, manufacturers can ensure the removal of closely related structural analogues and residual solvents, achieving the stringent purity specifications required for pharmaceutical applications. The stability of the sulfur bridge under the described extraction and purification conditions indicates that the core scaffold is robust, minimizing the risk of degradation during processing. This reliability is essential for maintaining batch-to-batch consistency, a key requirement for regulatory compliance and successful technology transfer from laboratory to commercial production facilities.
How to Synthesize Sulfur-containing Chromone Compounds Efficiently
The production process outlined in the patent offers a clear pathway for manufacturing these valuable intermediates, starting from strain preservation and ending with high-purity isolation. The protocol begins with the activation of the Penicillium oxalicum strain on PDA agar plates, followed by seed culture expansion in liquid PDA medium under controlled shaking conditions to ensure adequate oxygen transfer. The main fermentation is conducted statically at room temperature for 30 days, allowing the fungus to fully develop its mycelial network and secrete the secondary metabolites into the broth. Following fermentation, the broth is extracted with ethyl acetate, concentrated, and subjected to a series of chromatographic separations to isolate Compound 1 and Compound 2. These parent compounds can then serve as substrates for chemical modification, such as acetylation with acetic anhydride catalyzed by DMAP, to generate a range of derivatives with varying lipophilicity and biological activity.
- Cultivate Penicillium oxalicum CCTCC AF 93256 in PDA medium at 28°C for 30 days to generate fermentation broth.
- Extract the broth with ethyl acetate, concentrate, and purify using silica gel column chromatography with chloroform-methanol gradients.
- Perform acetylation on the isolated Compound 1 using acetic anhydride and DMAP in pyridine to obtain derivatives 3-6.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this fermentation-based production route offers substantial strategic advantages over purely synthetic alternatives. The reliance on renewable biological resources rather than finite petrochemical feedstocks enhances the long-term sustainability of the supply chain, mitigating risks associated with fossil fuel price fluctuations. The fermentation process utilizes common, inexpensive media components such as potatoes, glucose, and sea salt, which are globally available and cost-effective, contributing to significant raw material cost optimization. Furthermore, the downstream processing employs standard solvent systems and chromatography resins that are readily sourced from multiple vendors, preventing single-source bottlenecks and ensuring supply continuity. The scalability of the fermentation step, demonstrated in the patent examples up to 30 liters, suggests a straightforward path to industrial-scale production using standard bioreactor technology, facilitating rapid response to market demand increases without requiring massive capital investment in new specialized equipment.
- Cost Reduction in Manufacturing: The elimination of complex multi-step organic synthesis reduces the consumption of expensive reagents, catalysts, and energy-intensive heating or cooling cycles. By shifting the burden of molecular complexity construction to the fungal host, the manufacturing process becomes inherently more efficient, lowering the overall cost of goods sold (COGS). The use of ambient temperature for key reaction steps, such as the acetylation of Compound 1, further decreases energy consumption and operational expenses. Additionally, the high selectivity of the biosynthetic pathway minimizes the formation of difficult-to-remove byproducts, reducing the waste disposal costs associated with hazardous chemical waste treatment. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediates, allowing pharmaceutical partners to allocate more resources to clinical development and marketing efforts.
- Enhanced Supply Chain Reliability: The use of a well-characterized fungal strain deposited in a public culture collection ensures that the biological starting material is secure and reproducible, eliminating the risk of strain drift or loss that can plague proprietary microbial processes. The fermentation timeline of 30 days is predictable and manageable within standard production scheduling, allowing for accurate lead time forecasting and inventory planning. Since the process does not depend on rare earth metals or exotic reagents that are subject to geopolitical trade restrictions, the supply chain remains resilient against global disruptions. The ability to store the fermentation broth or intermediate extracts provides flexibility in production scheduling, enabling manufacturers to build safety stock during periods of low demand to buffer against future supply shocks.
- Scalability and Environmental Compliance: The fermentation and extraction processes described are compatible with existing Good Manufacturing Practice (GMP) facilities, allowing for seamless technology transfer and scale-up from pilot to commercial volumes. The use of ethyl acetate and alcohols as primary solvents aligns with ICH Q3C guidelines for residual solvents, simplifying the regulatory approval process for the final drug substance. The biological nature of the production method generates less hazardous waste compared to traditional chemical synthesis, reducing the environmental footprint and easing the burden of environmental compliance and permitting. This eco-friendly profile is increasingly valued by global pharmaceutical companies seeking to meet their corporate sustainability goals and reduce their Scope 3 emissions associated with raw material sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these sulfur-containing chromone compounds. The answers are derived directly from the experimental data and specifications provided in patent CN102603769A, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of incorporating these intermediates into your drug discovery pipeline.
Q: What is the primary biological source of these sulfur-containing chromones?
A: The compounds are secondary metabolites isolated from the fermentation broth of the fungus Penicillium oxalicum strain CCTCC AF 93256.
Q: Which cancer cell lines show sensitivity to these compounds?
A: The compounds exhibit cytotoxic activity against malignant melanoma (A375), lung cancer (A549), cervical cancer (HeLa), breast cancer (MCF-7), laryngeal cancer (Hep-2), liver cancer (HepG2), and intestinal cancer (SW-620) cell lines.
Q: How are the absolute configurations of the chiral centers determined?
A: The absolute configurations at C-6, C-11, and C-13 were established through a combination of NOESY spectral analysis, Mosher esterification reactions, and circular dichroism (CD) spectroscopy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-containing Chromone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of the sulfur-containing chromone scaffold described in CN102603769A for developing next-generation antitumor therapies. As a leading CDMO partner, we possess the technical expertise and infrastructure to scale this fermentation-based pathway from laboratory benchtop to commercial manufacturing, ensuring a steady supply of high-quality intermediates for your clinical trials. Our facilities are equipped with state-of-the-art fermentation tanks and purification suites capable of handling volumes ranging from 100 kgs to 100 MT/annual commercial production, providing the flexibility needed to support your project at every stage of development. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest standards of quality and consistency required by global regulatory agencies.
We invite you to collaborate with us to leverage this innovative technology for your oncology portfolio. Our team of experienced process chemists and biologists can work with you to optimize the fermentation yield and streamline the purification process, delivering a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation. Let us help you accelerate your drug development timeline with our reliable supply of bioactive pharmaceutical intermediates.
