Advanced Chiral Resolution Technology for High-Purity L-Corey Lactone Intermediates
The pharmaceutical industry's relentless pursuit of efficient prostaglandin synthesis has long been hindered by the complexities associated with obtaining optically pure intermediates. Patent CN112939911A introduces a groundbreaking chiral resolution process specifically designed for the preparation of L-Corey lactone, a pivotal building block in the manufacture of various prostaglandin drugs. This technology represents a significant leap forward by transitioning from cumbersome, low-yield traditional methods to a streamlined, industrially viable protocol that prioritizes both optical purity and environmental sustainability. By leveraging a sophisticated diastereomeric salt formation strategy coupled with an innovative recycling loop for resolving agents, this patent addresses the critical bottlenecks of cost and scalability that have historically plagued the supply chain for these high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of L-Corey lactone has relied heavily on resolution methods that suffer from inherent inefficiencies, such as those documented by Feng Zewang et al., which were characterized by excessively complicated operational procedures and substantial solvent consumption. Traditional approaches often struggle with low yields due to the difficulty in separating enantiomers with high precision without multiple, labor-intensive recrystallization steps that degrade overall throughput. Furthermore, conventional processes frequently fail to effectively recover the expensive chiral resolving agents, leading to inflated production costs and significant chemical waste that complicates environmental compliance. The reliance on harsh conditions or exotic reagents in older methodologies also poses safety risks and limits the feasibility of scaling these reactions to the multi-ton quantities required by the global pharmaceutical market.
The Novel Approach
In stark contrast, the novel methodology disclosed in CN112939911A simplifies the entire workflow by integrating hydrolysis and salt formation into a cohesive, continuous operation that minimizes unit operations. The process initiates with the hydrolysis of racemic compound I under mild basic conditions to form diastereomeric salt II, followed by the direct addition of a chiral amine solution to induce selective crystallization of the desired L-enantiomer. This approach not only streamlines the purification sequence but also incorporates a closed-loop system where mother liquors are treated to recover the resolving agent and racemize the unwanted enantiomer for reuse. 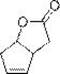 This holistic design ensures that the process is not merely a laboratory curiosity but a robust manufacturing platform capable of delivering consistent quality while adhering to green chemistry principles.
This holistic design ensures that the process is not merely a laboratory curiosity but a robust manufacturing platform capable of delivering consistent quality while adhering to green chemistry principles.
Mechanistic Insights into Diastereomeric Salt Crystallization
The core of this technological breakthrough lies in the precise control of diastereomeric salt formation, where the interaction between the racemic acid derivative and the chiral amine resolving agent creates distinct physical properties for each enantiomeric salt. By carefully adjusting the pH to a narrow range of 8 to 9 during the crystallization phase, the process exploits the solubility differences between the diastereomers to selectively precipitate the target L-enantiomer salt while keeping the unwanted D-enantiomer in the solution phase. The use of specific solvents such as ethyl acetate or ethanol further enhances this selectivity, allowing for the isolation of a crude salt with high initial optical purity that serves as an excellent foundation for subsequent purification steps. This mechanistic precision is critical for pharmaceutical applications where even trace amounts of the wrong enantiomer can compromise the safety and efficacy of the final drug product.
Furthermore, the patent details a rigorous impurity control mechanism through a dedicated recrystallization step that elevates the optical purity of the intermediate to pharmaceutical-grade standards. Following the initial crystallization, the crude L-enantiomer salt is subjected to recrystallization in esters like isopropyl acetate, which effectively strips away residual impurities and any co-crystallized undesired isomers. The final acidification step, conducted at a controlled pH of 3 to 4, liberates the free L-Corey lactone intermediate (Compound III) with exceptional stereochemical integrity, as evidenced by specific rotation values matching literature standards. This multi-stage purification strategy ensures that the final product meets the stringent specifications required for downstream synthesis of complex prostaglandin analogs without requiring additional chromatographic separations.
How to Synthesize L-Corey Lactone Intermediate Efficiently
The synthesis of this critical pharmaceutical intermediate follows a logical sequence of hydrolysis, resolution, and recovery that maximizes material efficiency. The process begins by dissolving the racemic starting material in a compatible organic solvent and treating it with an aqueous base to generate the reactive salt species necessary for resolution. Subsequent addition of the chiral amine resolving agent triggers the formation of the diastereomeric salt, which is then isolated through controlled crystallization and filtration. For a complete understanding of the specific reaction parameters, solvent ratios, and temperature controls required to replicate this high-yield process, please refer to the detailed standardized synthesis guide provided below.
- Hydrolyze racemic compound I in an organic solvent with an aqueous base solution to form diastereomeric salt II.
- Add chiral amine solution to the salt II mixture, adjust pH to 8-9, and crystallize to obtain the crude L-enantiomer salt.
- Recrystallize the crude salt, followed by acidification and extraction to isolate the final high-purity L-compound III.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented resolution technology translates directly into enhanced operational resilience and significant cost optimization across the manufacturing value chain. By eliminating the need for expensive transition metal catalysts often used in asymmetric synthesis, the process removes a major cost driver and reduces the regulatory burden associated with heavy metal clearance in the final API. The simplified operational workflow, which relies on standard unit operations like filtration and extraction rather than complex chromatography, allows for faster batch turnover times and reduced labor costs, thereby improving the overall economic viability of producing these high-value intermediates.
- Cost Reduction in Manufacturing: The most profound economic benefit arises from the integrated recycling loop that recovers the chiral resolving agent from the mother liquors with high efficiency. Instead of discarding the expensive chiral amine after a single use, the process regenerates it for repeated cycles, drastically lowering the raw material cost per kilogram of the final product. Additionally, the ability to racemize the unwanted enantiomer and feed it back into the process as starting material effectively doubles the theoretical yield from the initial racemic input, providing a substantial reduction in the cost of goods sold without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydroxide, ethanol, and ethyl acetate ensures that the supply chain is not vulnerable to the volatility often seen with specialized reagents or scarce natural products. This accessibility of raw materials guarantees a stable and continuous supply of the L-Corey lactone intermediate, mitigating the risk of production stoppages due to material shortages. Furthermore, the robustness of the crystallization process ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that could disrupt downstream API manufacturing schedules.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed to minimize waste generation through its closed-loop recycling system, aligning perfectly with modern green chemistry mandates. The reduction in solvent usage and the elimination of toxic heavy metal catalysts simplify wastewater treatment and lower the environmental footprint of the facility. This eco-friendly profile not only facilitates easier regulatory approval but also supports the long-term sustainability goals of multinational pharmaceutical companies seeking responsible suppliers for their critical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These answers are derived directly from the experimental data and process descriptions found in the patent documentation to provide clarity on feasibility and performance. Understanding these details is essential for technical teams evaluating the integration of this route into existing manufacturing frameworks.
Q: What are the primary advantages of this chiral resolution process over traditional asymmetric synthesis?
A: This process offers significant operational simplicity and eliminates the need for expensive chiral catalysts often required in asymmetric synthesis. By utilizing a robust diastereomeric salt formation and recycling loop for the resolving agent, it achieves high optical purity while drastically reducing raw material costs and waste generation compared to conventional methods.
Q: How does the patent address the issue of low yield in previous resolution methods?
A: The patent introduces a comprehensive recycling strategy where mother liquors containing the unwanted enantiomer are subjected to racemization under strong alkaline conditions. This converts the waste back into the racemic starting material, which can be re-fed into the process, thereby theoretically maximizing the overall yield and minimizing material loss.
Q: Is this process suitable for large-scale industrial production of prostaglandin intermediates?
A: Yes, the process is explicitly designed for industrialization. It utilizes common solvents like ethanol and ethyl acetate, operates at moderate temperatures, and features a straightforward work-up procedure involving filtration and extraction, making it highly scalable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Corey Lactone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the successful development of life-saving prostaglandin therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot scale to full industrial manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering L-Corey lactone intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this advanced chiral resolution process can optimize your specific supply chain requirements. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project's unique needs. Let us help you secure a reliable, cost-effective, and high-quality supply of this essential pharmaceutical building block.
