Scalable Production of 3-Bromo-6-Chloropyridine-2-Carboxylic Acid via Directed Metalation
Scalable Production of 3-Bromo-6-Chloropyridine-2-Carboxylic Acid via Directed Metalation
The pharmaceutical and agrochemical industries continuously demand high-purity heterocyclic building blocks that possess multiple functional handles for downstream diversification. A recent technological breakthrough documented in patent CN114890942A introduces a highly efficient, one-step synthesis strategy for 3-bromo-6-chloropyridine-2-carboxylic acid, a critical intermediate characterized by its polyhalogenated picolinic acid structure. This compound serves as a versatile scaffold where the chlorine and bromine atoms act as distinct reactive sites for cross-coupling reactions such as Suzuki, Buchwald-Hartwig, or Sonogashira couplings, while the carboxyl group offers further condensation possibilities. The disclosed method leverages the unique reactivity of the magnesium amide base TMPMgCl•LiCl to achieve regioselective metalation at the C2 position of the pyridine ring, followed by immediate carboxylation with dry ice. This innovation represents a significant leap forward for any reliable agrochemical intermediate supplier or pharma partner seeking to streamline their supply chain for complex nitrogenous heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-bromo-6-chloropyridine-2-carboxylic acid has been plagued by severe safety hazards and operational inefficiencies that hinder large-scale adoption. One prevalent prior art method involves the nucleophilic substitution of 2-fluoro-3-bromo-6-chloropyridine using virulent sodium cyanide, followed by acidic hydrolysis. This route not only necessitates the handling of extremely toxic cyanide salts, posing grave risks to personnel and the environment, but also generates substantial chemical waste that requires costly disposal protocols. Another existing pathway relies on the oxidation of 3-bromo-6-chloropyridine to its N-oxide derivative using urea peroxide and trifluoroacetic anhydride, followed by cyanation and hydrolysis. This multi-step sequence introduces unstable nitrogen oxide intermediates which present potential explosion hazards, alongside harsh reaction conditions involving strong acids and elevated temperatures. Consequently, these conventional routes are often restricted to small laboratory scales due to their poor atom economy, high toxicity profiles, and complex purification requirements involving chromatographic separation.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent data utilizes a direct C-H functionalization strategy that dramatically simplifies the synthetic landscape. By employing 5-bromo-2-chloropyridine, a commercially abundant and cost-effective starting material, the process bypasses the need for pre-functionalized precursors like fluoro-derivatives or N-oxides. The core of this innovation lies in the use of the mixed magnesium-lithium amide base, TMPMgCl•LiCl, which facilitates a highly selective deprotonation at the C2 position adjacent to the nitrogen atom. Following metalation, the intermediate is quenched directly with dry ice to install the carboxylic acid moiety in a single pot. This approach eliminates the generation of high-risk intermediates and avoids the use of heavy metal catalysts or toxic cyanide sources entirely. Furthermore, the workup procedure is remarkably straightforward, involving simple acidification and solvent extraction, which obviates the need for energy-intensive column chromatography, thereby significantly reducing solvent consumption and production time.
Mechanistic Insights into TMPMgCl•LiCl Mediated Directed Metalation
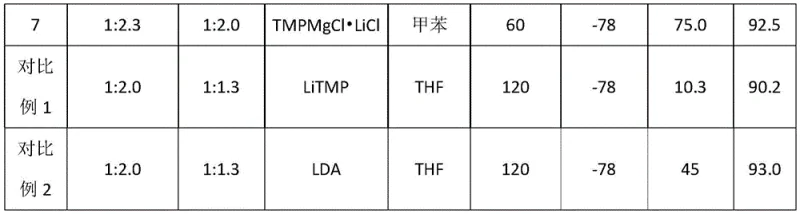
The success of this synthesis hinges on the exceptional properties of the TMPMgCl•LiCl reagent, often referred to as a "Turbo Grignard" base, which combines the basicity of a lithium amide with the nucleophilicity and solubility characteristics of a Grignard reagent. In the context of the pyridine substrate 5-bromo-2-chloropyridine, the nitrogen atom acts as a powerful directing group, coordinating with the magnesium center to facilitate deprotonation specifically at the C2 position. This regioselectivity is crucial because the C3, C4, C5, and C6 positions are either sterically hindered or electronically deactivated by the halogen substituents. The presence of LiCl in the complex enhances the solubility of the organomagnesium species in non-polar solvents like toluene and stabilizes the transition state, allowing the reaction to proceed smoothly even at cryogenic temperatures such as -78°C. This low-temperature operation is vital for suppressing side reactions, such as halogen-metal exchange at the bromine or chlorine sites, which would lead to undesired byproducts and lower the overall yield of the target acid.
From an impurity control perspective, the mechanism ensures a clean reaction profile by avoiding the formation of polymeric tars or decomposition products often seen in harsher nucleophilic substitutions. The kinetic control exerted by the bulky TMP ligand prevents over-metallation or attack on the sensitive carbon-halogen bonds, preserving the integrity of the bromine and chlorine atoms which are essential for downstream coupling chemistry. Upon addition of dry ice, the magnesiated intermediate undergoes rapid carboxylation to form the magnesium carboxylate salt. The subsequent acidic workup with dilute hydrochloric acid protonates the carboxylate to release the free acid, which precipitates or partitions into the organic phase depending on the pH adjustment. The patent data indicates that maintaining the pH between 4 and 5 during workup is optimal for maximizing recovery while minimizing the co-extraction of inorganic salts, resulting in a crude product with purity levels exceeding 97% without further purification.
How to Synthesize 3-Bromo-6-Chloropyridine-2-Carboxylic Acid Efficiently
Implementing this synthesis in a pilot or production plant requires strict adherence to temperature controls and stoichiometric ratios to ensure reproducibility and safety. The process begins with the dissolution of the starting material in an anhydrous solvent like toluene under an inert atmosphere, followed by the controlled addition of the base at cryogenic conditions. The reaction mixture is then transferred to a slurry of dry ice, allowing the exothermic carboxylation to occur safely before warming to ambient temperature for completion. While the general concept is straightforward, precise execution regarding mixing rates, temperature ramps, and quenching protocols is essential to maintain the high yields reported in the patent examples. For detailed operational parameters, including specific flow rates, agitation speeds, and safety interlocks required for GMP compliance, please refer to the standardized synthesis steps provided below.
- Dissolve 5-bromo-2-chloropyridine in toluene, cool to -78°C, and add TMPMgCl•LiCl solution dropwise, maintaining temperature for 1 hour.
- Prepare a slurry of dry ice in toluene and add the metallated intermediate solution to it, allowing the reaction to warm to room temperature over 12 hours.
- Quench the reaction with hydrochloric acid to pH 4.5, extract with ethyl acetate, wash with brine, dry, and concentrate to obtain the pure acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this TMPMgCl•LiCl mediated route offers transformative benefits that extend far beyond simple yield improvements. The elimination of toxic cyanide reagents and unstable N-oxide intermediates fundamentally alters the risk profile of the manufacturing process, leading to substantial reductions in insurance premiums, waste disposal costs, and regulatory compliance burdens. By removing the necessity for chromatographic purification, the process drastically cuts down on the volume of organic solvents required, which directly translates to lower raw material costs and a smaller environmental footprint. This efficiency gain is particularly critical in the current market landscape where solvent prices are volatile and environmental regulations regarding VOC emissions are becoming increasingly stringent globally.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the simplification of the unit operations and the avoidance of expensive reagents. Traditional methods often require multiple isolation steps, specialized equipment for handling toxic gases or solids, and extensive purification protocols that consume significant labor and utility resources. In contrast, this one-pot metalation-carboxylation sequence consolidates the synthesis into fewer steps, reducing the overall cycle time and equipment occupancy. The use of 5-bromo-2-chloropyridine as a starting material is particularly advantageous as it is produced on a multi-ton scale globally, ensuring a stable and competitive pricing baseline compared to custom-synthesized fluoro-precursors. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the loss of valuable material during purification and maximizing the effective output per batch.
- Enhanced Supply Chain Reliability: Supply continuity is a paramount concern for downstream drug manufacturers, and this process significantly de-risks the supply chain by relying on robust, commodity-grade chemicals. The reagents used, such as TMPMgCl•LiCl and dry ice, are widely available from multiple global suppliers, preventing bottlenecks associated with sole-source specialty reagents. The operational simplicity of the method means that it can be easily transferred between different manufacturing sites or scaled up from pilot plants to commercial reactors without requiring bespoke engineering solutions. This flexibility ensures that production schedules can be maintained even in the face of logistical disruptions, providing a reliable source of high-purity intermediates for time-sensitive drug development programs.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry principles, this method aligns perfectly with sustainability goals by designing out hazards at the molecular level. The absence of heavy metal catalysts eliminates the need for costly and complex metal scavenging steps, which are often required to meet strict residual metal limits in pharmaceutical ingredients. The aqueous waste stream generated from the acidic workup is relatively benign compared to the cyanide-laden effluents of older methods, simplifying wastewater treatment and reducing the environmental liability of the manufacturing site. This inherent safety and cleanliness make the process highly scalable, capable of supporting production volumes ranging from hundreds of kilograms to multi-ton annual capacities without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-bromo-6-chloropyridine-2-carboxylic acid using this advanced metalation strategy. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing synthesis pipelines.
Q: Why is the TMPMgCl•LiCl method superior to traditional cyanide substitution?
A: Traditional methods rely on toxic sodium cyanide and harsh hydrolysis conditions, generating hazardous waste. The TMPMgCl•LiCl route avoids cyanide entirely, uses safer reagents, and achieves higher purity without chromatographic purification.
Q: What is the expected yield and purity for this synthesis?
A: Under optimized conditions described in patent CN114890942A, the process achieves yields up to 87.5% with purity reaching 97.5% after simple extraction and crystallization, eliminating the need for column chromatography.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes commercially available raw materials like 5-bromo-2-chloropyridine and avoids unstable intermediates like pyridine N-oxides. The simple workup involving acidification and extraction makes it highly scalable for ton-level production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-6-Chloropyridine-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN114890942A can be reliably replicated on an industrial scale. We operate state-of-the-art facilities equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 3-bromo-6-chloropyridine-2-carboxylic acid meets the exacting standards required for API synthesis and advanced agrochemical formulations. Our commitment to quality assurance means that we meticulously monitor every critical process parameter, from the moisture content of the solvents to the precise temperature profiles during metalation, to deliver consistent product performance.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient route for your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your unique supply chain needs, ensuring that you secure a competitive advantage through superior raw material sourcing and process optimization.
