Optimizing Iopromide Production: A Technical Breakthrough in Contrast Agent Manufacturing
Optimizing Iopromide Production: A Technical Breakthrough in Contrast Agent Manufacturing
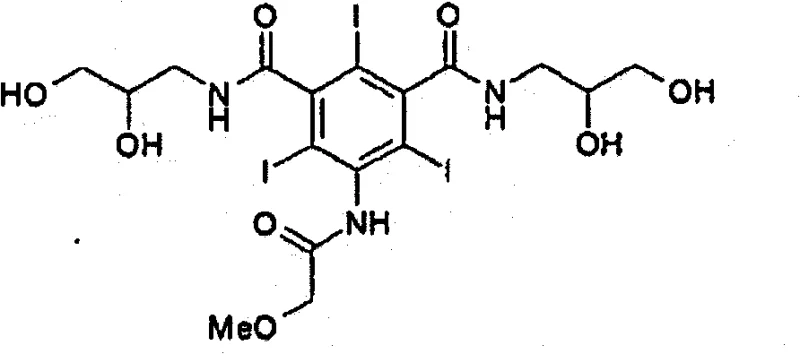
The pharmaceutical industry continuously seeks robust synthetic routes for critical diagnostic agents, particularly non-ionic X-ray contrast media like Iopromide. Patent CN102015624B discloses a novel preparation method that fundamentally addresses the longstanding challenge of bismer byproduct formation. Traditional synthesis pathways often struggle with the generation of symmetric diamide impurities that are structurally similar to the target molecule, making purification arduous and yield-limiting. This innovation introduces specific acetylated intermediates, namely 5-methoxyacetamido-2,4,6-triiodoisophthalic acid (2,3-diacetoxypropyl) amide chloride and its diamide derivative, to streamline the production workflow. By integrating these intermediates, the process achieves high-purity Iopromide with significantly improved yield efficiency, eliminating the need for excessive recrystallization cycles that typically plague conventional manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iopromide has been hindered by the inevitable formation of bismer byproducts, specifically the N,N'-bis(2,3-dihydroxypropyl) diamide derivative. In standard procedures, such as those disclosed in earlier patents like U.S. Patent No. 4,364,921, the reaction of 5-amino-2,4,6-triiodoisophthalic acid dichloride with amines often lacks sufficient selectivity. When attempting to introduce the second amine group, excess reagent or improper stoichiometry leads to the formation of the symmetric bismer impurity. This byproduct possesses physicochemical properties very similar to the desired asymmetric product, rendering standard filtration or simple washing ineffective. Consequently, manufacturers are forced to employ multiple, labor-intensive crystallization and filtration steps using large volumes of organic solvents to achieve acceptable purity levels. This not only drastically reduces the overall process yield due to material loss during purification but also increases the environmental burden and operational costs associated with solvent recovery and waste disposal.
The Novel Approach
The patented methodology circumvents these issues by employing a strategic protection-deprotection sequence centered on acetylation. Instead of reacting the fully deprotected diol directly in the final amidation step, the process utilizes an intermediate where the hydroxyl groups are protected as acetates (Formula 19). This structural modification is pivotal; it allows for the selective crystallization and removal of the symmetric bismer byproduct (Formula 21) at an intermediate stage, rather than at the final API stage. The acetylated bismer exhibits different solubility characteristics compared to the target mono-amide chloride intermediate, facilitating easy separation. Furthermore, the subsequent conversion to the final diamide (Formula 20) and eventual hydrolysis proceeds with higher fidelity. This approach effectively decouples the purification challenge from the final step, ensuring that the bulk of impurities are removed early in the synthesis, thereby safeguarding the yield and purity of the final Iopromide product without requiring additional, post-synthesis purification treatments.
Mechanistic Insights into Selective Amidation and Acetylation
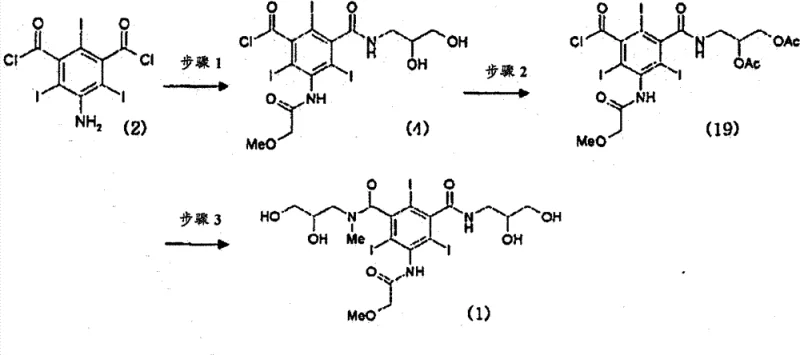
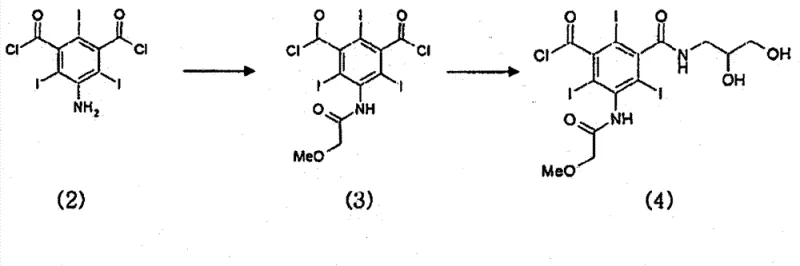
The core of this technical advancement lies in the precise control of functional group reactivity during the amidation phases. In the initial step, 5-amino-2,4,6-triiodoisophthalic acid dichloride is reacted with methoxyacetyl chloride to protect the amino group, followed by selective mono-amidation with 2,3-dihydroxypropylamine. The stoichiometry here is critical; using approximately 0.6 to 0.7 equivalents of the amine ensures that the formation of the bis-amide is minimized from the outset. The resulting intermediate retains a reactive acid chloride group while possessing free hydroxyls. The subsequent acetylation step using acetic anhydride and a catalytic amount of sulfuric acid transforms these hydroxyls into acetate esters. This transformation is not merely protective; it serves a purification function. The symmetric bismer byproduct, if formed, becomes fully acetylated (Formula 21), which can be separated from the desired mono-amide chloride (Formula 19) via crystallization. This mechanistic intervention exploits the subtle differences in crystal lattice energy and solubility between the asymmetric target and the symmetric impurity, a distinction that is far less pronounced in the non-acetylated species.
Furthermore, the stability of the acetylated intermediate allows for robust handling and storage prior to the final coupling reaction. In the final stage, the acetylated mono-amide chloride reacts with 2,3-dihydroxy-N-methylpropylamine to form the di-amide structure. The presence of the acetate groups prevents unwanted side reactions at the hydroxyl positions during this coupling. Finally, a mild hydrolysis step removes the acetate protecting groups to reveal the free hydroxyls required for the water solubility of the final contrast agent. This sequence ensures that the chiral centers and functional groups remain intact throughout the synthesis, minimizing the formation of regioisomers or degradation products. The rigorous control over the reaction environment, particularly the use of dimethylacetamide as a solvent and triethylamine as a base, further suppresses side reactions, contributing to the high purity profile observed in the final product.
How to Synthesize Iopromide Efficiently
The synthesis of Iopromide via this patented route involves a carefully orchestrated sequence of protection, amidation, and deprotection steps designed to maximize yield and minimize impurity carryover. The process begins with the activation of the isophthalic acid core, followed by the strategic introduction of the side chains. Detailed operational parameters, including temperature controls, solvent ratios, and stoichiometric precision, are critical for replicating the high success rates reported in the patent data. Manufacturers aiming to adopt this route should pay close attention to the crystallization conditions of the acetylated intermediate, as this is the primary purification checkpoint.
- React 5-amino-2,4,6-triiodoisophthalic acid dichloride with methoxyacetyl chloride, followed by selective amidation with 2,3-dihydroxypropylamine to form the mono-amide chloride intermediate.
- Perform acetylation on the hydroxyl groups of the intermediate using acetic anhydride and sulfuric acid catalyst, enabling the removal of symmetric bismer byproducts via crystallization.
- React the purified acetylated intermediate with 2,3-dihydroxy-N-methylpropylamine, followed by hydrolysis to yield the final high-purity Iopromide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical elegance. The primary advantage lies in the significant simplification of the downstream processing workflow. By eliminating the need for multiple, repetitive crystallization steps to remove the bismer byproduct, the overall processing time is drastically reduced. This reduction in unit operations translates directly into lower utility consumption, reduced labor hours, and decreased wear and tear on processing equipment. Furthermore, the ability to remove impurities at an intermediate stage rather than at the final API stage minimizes the risk of losing high-value iodinated material late in the process, thereby improving the effective mass balance of the production run. This efficiency gain is crucial for maintaining cost competitiveness in the global market for contrast media.
- Cost Reduction in Manufacturing: The streamlined process eliminates the requirement for extensive solvent usage associated with repeated purification cycles. Conventional methods often demand large volumes of organic solvents for recrystallization to meet purity specifications, which incurs high costs for solvent purchase, recovery, and waste treatment. By achieving high purity through a single crystallization of the acetylated intermediate, this method significantly lowers the variable costs per kilogram of produced Iopromide. Additionally, the improved yield means that less raw material is required to produce the same amount of final product, further enhancing the economic viability of the manufacturing process without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply chain stability. Conventional processes that rely on complex purification are more susceptible to batch failures or variability in purity, which can lead to production delays and inconsistent supply. The new method's ability to consistently remove the difficult bismer impurity ensures a more predictable production schedule. This reliability is essential for meeting the rigorous delivery timelines demanded by pharmaceutical clients who require uninterrupted supply of critical diagnostic agents. The simplified workflow also reduces the dependency on specialized purification equipment, making the process easier to scale across different manufacturing sites if necessary.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with modern green chemistry principles. The reduction in solvent usage directly correlates to a lower environmental footprint, reducing the volume of hazardous waste generated per unit of product. This is increasingly important as regulatory bodies impose stricter limits on solvent emissions and waste disposal. The process is inherently scalable, as the key steps involve standard chemical transformations like amidation and esterification that are well-understood in industrial settings. The ability to produce high-purity material with fewer processing steps facilitates easier technology transfer and scale-up from pilot plant to commercial production scales, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the quality implications of the new route. The answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance to industrial application.
Q: How does this new method address the bismer byproduct issue in Iopromide synthesis?
A: The method introduces a specific acetylation step on the hydroxyl groups of the intermediate (Formula 19). This modification alters the solubility profile, allowing the symmetric bismer byproduct (Formula 21) to be effectively removed through simple crystallization without needing complex additional purification steps.
Q: What are the key intermediates used in this patented process?
A: The process relies on two critical intermediates: 5-methoxyacetamido-2,4,6-triiodoisophthalic acid (2,3-diacetoxypropyl) amide chloride (Formula 19) and its subsequent diamide derivative (Formula 20). These intermediates facilitate better control over the reaction stoichiometry and impurity profile.
Q: Why is the removal of the bismer byproduct critical for X-ray contrast agents?
A: Bismer byproducts, such as N,N'-bis(2,3-dihydroxypropyl) derivatives, can significantly lower the purity of the final API. For injectable contrast agents like Iopromide, high purity is mandatory to ensure patient safety and meet stringent pharmacopeial standards for heavy metals and organic impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iopromide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving diagnostic agents like Iopromide. Our technical team has extensively analyzed advanced synthetic routes, including the innovative acetylation strategy detailed in recent patents, to ensure our manufacturing capabilities remain at the forefront of the industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities or full-scale commercial supply, our facilities are equipped to deliver. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against global pharmacopeial standards.
We invite pharmaceutical partners to collaborate with us to leverage these advanced manufacturing efficiencies. By optimizing the synthesis of complex iodinated intermediates, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our expertise in fine chemical synthesis can enhance your supply chain resilience and drive down the total cost of ownership for your contrast agent portfolio.
