Optimized Industrial Synthesis of High-Purity Ioversol Intermediates for Global Contrast Media Manufacturing
Optimized Industrial Synthesis of High-Purity Ioversol Intermediates for Global Contrast Media Manufacturing
The global demand for non-ionic X-ray contrast media continues to surge, driven by the increasing prevalence of diagnostic imaging procedures in modern healthcare systems. At the heart of this market lies Ioversol, a highly water-soluble and stable agent known commercially as Optiray. The economic and technical viability of producing Ioversol depends heavily on the efficiency of synthesizing its key precursor, 5-chloroacetamide-N,N'-bi(2,3-dihydroxyl propyl)-2,4,6-triiodo-1,3-benzenedicarboxamide. A pivotal advancement in this domain is detailed in patent CN101654417B, which discloses a revolutionary preparation method that fundamentally restructures the synthetic pathway. Unlike legacy processes that rely on cumbersome nitro-reduction sequences, this innovation utilizes 5-amino-2,4,6-triiodo-1,3-phthalic acid as a direct starting material. This strategic shift not only simplifies the operational workflow but also addresses critical pain points regarding yield stability and reagent consumption. For R&D directors and procurement specialists alike, understanding this mechanistic pivot is essential for securing a competitive edge in the pharmaceutical intermediates sector.
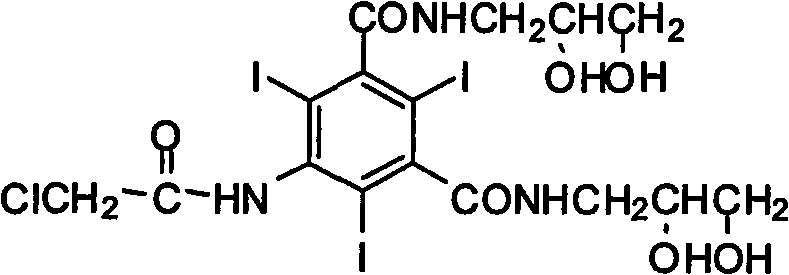
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this critical intermediate has been plagued by inefficiencies inherent in multi-step functional group transformations. Traditional routes, such as those described in earlier US patents, typically commence with 5-nitro-1,3-phthalic acid methyl esters. This necessitates an initial amidation with 3-amino-1,2-propanediol, followed by a catalytic hydrogenation reduction to convert the nitro group into an amine. Subsequently, an iodination step using iodine monochloride is required to introduce the three iodine atoms essential for X-ray opacity. Finally, the amino group must be acylated with chloroacetyl chloride. However, a significant drawback arises in this final stage: the presence of four hydroxyl groups on the molecule leads to unavoidable esterification side reactions with chloroacetyl chloride. Consequently, a subsequent basic hydrolysis step is mandatory to cleave these unwanted esters, drastically inflating the consumption of chloroacetyl chloride and complicating the purification process. This convoluted pathway, illustrated in the reaction scheme below, results in lower overall yields and higher production costs due to the excessive use of reagents and energy-intensive separation units.
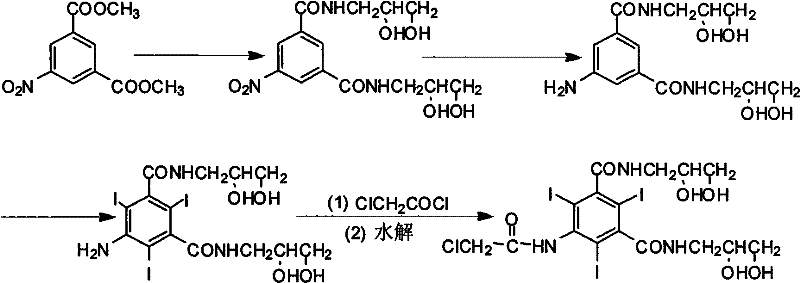
The Novel Approach
In stark contrast, the methodology outlined in CN101654417B introduces a streamlined strategy that bypasses these historical bottlenecks by altering the sequence of bond formation. The novel approach initiates with 5-amino-2,4,6-triiodo-1,3-phthalic acid, a substrate that already possesses the requisite iodine pattern and amino functionality. By converting this acid directly into its diacyl chloride derivative using thionyl chloride, the process creates a highly reactive intermediate ready for immediate functionalization. Crucially, the chloroacetylation is performed on this diacyl chloride species before the introduction of the diol moiety. Since the diacyl chloride lacks hydroxyl groups, the reaction with chloroacetyl chloride proceeds with high selectivity at the amino position, completely eliminating the formation of ester byproducts. This ingenious reordering of steps removes the need for the downstream hydrolysis stage entirely. The resulting 5-chloroacetamide-2,4,6-triiodo-1,3-benzenedicarbonyl dichloride is then coupled with 3-amino-1,2-propanediol to yield the final product. This reduction in unit operations translates directly to enhanced process robustness and significantly improved atom economy.
Mechanistic Insights into Selective Acylation and Amidation
The core chemical innovation of this process lies in the precise control of nucleophilicity and electrophilicity during the acylation stages. In the first critical transformation, 5-amino-2,4,6-triiodo-1,3-phthalic acid reacts with thionyl chloride in ethyl acetate. The mechanism involves the nucleophilic attack of the carboxylic acid oxygen on the sulfur atom of thionyl chloride, followed by the elimination of SO2 and HCl gases, driving the equilibrium towards the formation of the diacyl chloride. Maintaining the reaction temperature between 50°C and 90°C is vital to ensure complete conversion while preventing thermal degradation of the sensitive triiodo-aromatic core. The absence of hydroxyl groups in this intermediate is the key differentiator; it ensures that when chloroacetyl chloride is introduced in the subsequent step, the only available nucleophile is the aromatic amine. This prevents the competitive acylation of alcohol groups that plagues the conventional route. The reaction is conducted in N,N-dimethylacetamide (DMAC), a polar aprotic solvent that stabilizes the transition state and solubilizes the highly iodinated intermediate effectively.
Furthermore, the final amidation step demonstrates exceptional selectivity through the use of triethylamine as a proton scavenger. When 3-amino-1,2-propanediol is introduced to the 5-chloroacetamide-diacyl chloride, the primary amine of the propanediol acts as a potent nucleophile, attacking the carbonyl carbons of the acid chloride groups. The triethylamine neutralizes the HCl generated during this exothermic process, preventing the protonation of the amine nucleophile which would otherwise deactivate it. The reaction conditions are carefully controlled, with the addition occurring below 10°C to manage the exotherm, followed by heating to 30-70°C to drive the reaction to completion over 8 to 16 hours. The workup procedure is equally refined; instead of complex chromatographic separations, the product is isolated by adjusting the pH of the aqueous solution to 3 using hydrochloric acid. This precise pH control precipitates the target molecule while keeping soluble impurities in the mother liquor, ensuring a high-purity profile suitable for parenteral applications without the need for recrystallization from hazardous solvents.
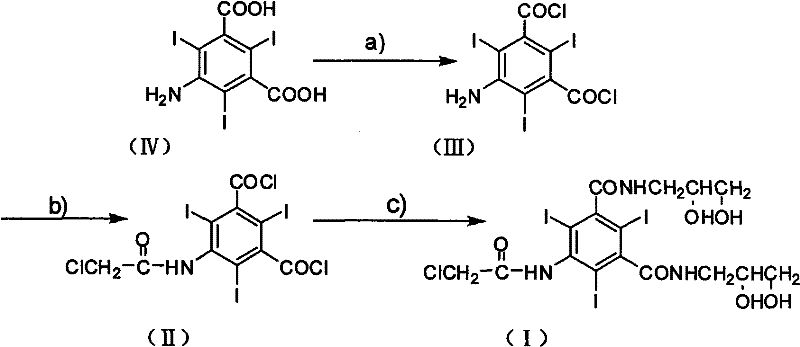
How to Synthesize 5-Chloroacetamide-N,N'-bi(2,3-dihydroxyl propyl)-2,4,6-triiodo-1,3-benzenedicarboxamide Efficiently
Implementing this optimized synthesis requires strict adherence to the stoichiometric ratios and thermal profiles defined in the patent to maximize yield and safety. The process begins with the activation of the phthalic acid derivative, followed by the sequential introduction of the chloroacetyl group and finally the diol side chains. Each step builds upon the purity of the previous intermediate, emphasizing the importance of in-process controls. While the general chemistry is straightforward, the scale-up from laboratory to commercial production demands rigorous attention to gas evolution during the thionyl chloride step and heat management during the amidation. The following guide outlines the standardized operational framework derived from the patent examples, providing a roadmap for technical teams aiming to replicate this high-efficiency pathway.
- Convert 5-amino-2,4,6-triiodo-1,3-phthalic acid to its corresponding diacyl chloride using thionyl chloride in ethyl acetate at elevated temperatures.
- Perform selective chloroacetylation on the amino group of the diacyl chloride intermediate using chloroacetyl chloride in DMAC solvent.
- React the resulting 5-chloroacetamide-2,4,6-triiodo-1,3-benzenedicarbonyl dichloride with 3-amino-1,2-propanediol in the presence of triethylamine to form the final diamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic benefits beyond mere technical elegance. The primary advantage is the substantial reduction in raw material costs, driven by the elimination of the hydrolysis step and the associated waste of chloroacetyl chloride. In the conventional process, a significant portion of this expensive reagent is consumed in forming transient esters that are later destroyed; the new method directs nearly 100% of the chloroacetyl chloride towards the desired amide bond. Additionally, the shortened reaction sequence reduces the overall cycle time, allowing for faster turnover of production assets and improved responsiveness to market demand fluctuations. The use of common, industrially available solvents like ethyl acetate and DMAC further simplifies logistics, reducing the reliance on specialized or hazardous reagents that often face supply volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the workflow. By removing the hydrolysis unit operation, manufacturers save on the costs of base reagents, acid for neutralization, and the energy required for additional heating and cooling cycles. Furthermore, the high selectivity of the chloroacetylation step means that less chloroacetyl chloride is required per kilogram of product, directly lowering the bill of materials. The ability to use the crude diacyl chloride directly in the next step without purification also eliminates solvent exchange and drying costs, contributing to a leaner, more cost-effective manufacturing model that enhances margin potential in a competitive generic drug market.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this route mitigates the risk of production delays. Traditional methods involving catalytic hydrogenation and iodine monochloride handling are prone to variability and safety incidents that can halt production lines. The new method relies on stable, non-catalytic liquid-phase reactions that are easier to control and scale. The starting material, 5-amino-2,4,6-triiodo-1,3-phthalic acid, is a stable solid that can be sourced or stockpiled effectively, decoupling the production schedule from the complexities of on-site iodination. This stability ensures a consistent flow of high-purity pharmaceutical intermediates, safeguarding the downstream formulation of the final contrast agent against upstream disruptions.
- Scalability and Environmental Compliance: The environmental footprint of the manufacturing process is significantly reduced, aligning with increasingly stringent global regulatory standards. The elimination of the hydrolysis step reduces the volume of saline wastewater generated, lowering the burden on effluent treatment plants. Moreover, the reaction generates gaseous byproducts like SO2 and HCl which can be efficiently scrubbed and recovered, minimizing atmospheric emissions. The high yields reported in the patent examples (exceeding 89% in key steps) imply less waste generation per unit of product. This green chemistry profile not only facilitates regulatory approval but also future-proofs the supply chain against tightening environmental regulations, ensuring long-term operational continuity for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These insights are derived directly from the experimental data and claims within patent CN101654417B, providing a reliable foundation for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term value proposition of this supply route.
Q: Why is the new synthesis route for Ioversol intermediate more cost-effective than traditional methods?
A: The novel route eliminates the need for a hydrolysis step required in conventional methods. By performing chloroacetylation on the amino-phthalic acid derivative before amidation with the diol, side reactions forming esters are prevented, significantly reducing the consumption of expensive chloroacetyl chloride and simplifying purification.
Q: What are the critical quality control parameters for this contrast media intermediate?
A: Critical parameters include the purity of the 5-amino-2,4,6-triiodo-1,3-phthalic acid starting material, precise temperature control during the exothermic acyl chloride formation (50-90°C), and strict pH adjustment during the final isolation to ensure complete removal of unreacted amines and acidic byproducts.
Q: How does this process improve supply chain reliability for contrast agent manufacturers?
A: By shortening the synthetic sequence and utilizing robust, scalable reaction conditions such as ambient pressure acyl chloride formation, the process reduces production lead times and minimizes the risk of batch failures associated with complex multi-step reductions and iodinations found in older patents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ioversol Intermediate Supplier
The transition to this optimized synthetic pathway represents a significant opportunity for contrast media manufacturers to enhance their competitive positioning through cost efficiency and supply security. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and corrosive challenges of acyl chloride chemistry, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to continuous process improvement, we deliver high-purity Ioversol intermediates that consistently exceed the demanding standards required for injectable pharmaceutical products.
We invite global partners to collaborate with us to unlock the full potential of this efficient manufacturing route. Whether you require a Customized Cost-Saving Analysis for your existing supply chain or need to validate the technical feasibility of this new method for your specific application, our technical procurement team is ready to assist. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and reliability in your contrast media production.
