Efficient Metal-Free Synthesis of Beta-Acyl Allyl Methyl Sulfones for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex molecular scaffolds, particularly those containing sulfone motifs which are prevalent in bioactive molecules. A recent technological breakthrough documented in patent CN115304524A introduces a highly efficient, one-pot synthesis strategy for beta-acyl allyl methyl sulfone compounds that addresses many of the historical pain points associated with sulfone chemistry. This novel approach leverages a Selectfluor-promoted cascade coupling reaction between readily available aryl methyl ketones and dimethyl sulfoxide (DMSO), operating under mild conditions without the need for transition metal catalysts. For R&D directors and procurement specialists alike, this represents a significant paradigm shift, offering a pathway to high-purity intermediates with a drastically simplified workup procedure and enhanced safety profile compared to traditional multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-acyl allyl methyl sulfone derivatives has relied on methodologies that are often fraught with operational complexities and economic inefficiencies. Prior art, such as the p-toluenesulfonic acid (TsOH) and acetic acid promoted reactions reported in literature, typically requires harsh thermal conditions reaching up to 150°C and often necessitates a two-step process that lowers overall throughput. Furthermore, alternative strategies employing Iron(III) catalysis, while effective, introduce the significant burden of transition metal contamination, which is a critical quality attribute concern for pharmaceutical intermediates intended for human consumption. The removal of residual iron to meet stringent regulatory specifications often requires additional purification steps, such as specialized scavenging resins or repeated recrystallizations, which inevitably drive up manufacturing costs and extend production lead times. These conventional routes also frequently suffer from limited substrate scope, struggling to accommodate sensitive functional groups that are common in modern drug discovery pipelines.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115304524A utilizes a unique combination of Selectfluor and hydroxylamine hydrochloride to facilitate a direct cross-coupling between methyl ketones and DMSO in a mixed solvent system of toluene and DMSO (7:1 ratio). This innovative protocol operates at a moderate temperature of 120°C and completes the transformation in a single pot within approximately 10 hours, demonstrating exceptional chemoselectivity and regioselectivity. By completely avoiding the use of precious or transition metals, this process inherently produces a cleaner crude reaction profile, significantly reducing the burden on downstream purification units. The ability to use DMSO not just as a solvent but as a dual-purpose reagent providing both the vinyl and methyl sulfone fragments represents a remarkable atom-economic advancement. This streamlined approach allows for the direct conversion of simple, commercially available acetophenones into highly functionalized sulfone derivatives with impressive yields, as evidenced by the successful synthesis of various substituted derivatives including fluoro, chloro, and methoxy variants.
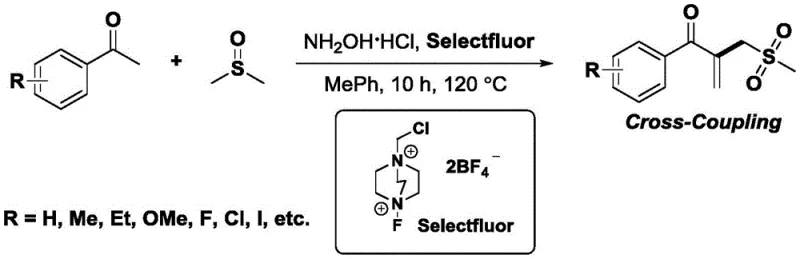
Mechanistic Insights into Selectfluor-Promoted Cross-Coupling
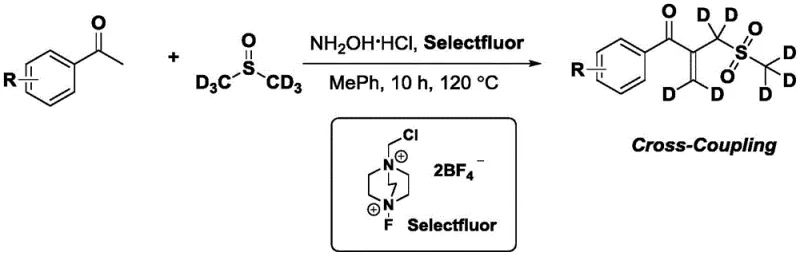
The mechanistic underpinning of this transformation is a fascinating cascade that highlights the versatility of hypervalent iodine-free fluorinating agents in organic synthesis. The reaction initiates with the activation of the methyl ketone by the Selectfluor reagent in the presence of hydroxylamine hydrochloride, generating a reactive intermediate that is poised for nucleophilic attack. DMSO, traditionally viewed merely as a polar aprotic solvent, plays a pivotal role here as a carbon and sulfur source; mechanistic studies involving deuterium labeling have confirmed that the dimethyl sulfoxide molecule provides both the vinyl group and the methyl sulfone moiety incorporated into the final product structure. This dual-donor capability eliminates the need for pre-functionalized vinyl sulfone reagents, which are often expensive and unstable. The reaction proceeds through a series of oxidative and elimination steps that are carefully balanced by the reaction conditions, ensuring that the carbonyl group of the starting ketone remains intact while the alpha-position is functionalized. This level of control is crucial for maintaining the integrity of the beta-dicarbonyl-like motif found in the product, which is essential for subsequent cyclization reactions to form thiophenes or other heterocycles.
Furthermore, the tolerance of this catalytic system towards a wide array of electronic environments on the aromatic ring suggests a robust radical or cationic manifold that is not easily perturbed by steric or electronic hindrance. Whether the substrate bears strong electron-withdrawing groups like trifluoromethyl or nitro groups, or electron-donating groups like methoxy and alkyl chains, the reaction maintains high efficiency and selectivity. This broad substrate scope is validated by extensive experimental data within the patent, covering ortho-, meta-, and para-substituted acetophenones as well as heteroaromatic substrates like acetylthiophene. The absence of metal catalysts also means there is no risk of metal-mediated side reactions such as homocoupling or unwanted reductions, which further contributes to the high purity of the isolated products. Understanding these mechanistic nuances allows process chemists to confidently predict the outcome of the reaction on novel substrates, facilitating faster route scouting and optimization campaigns.
How to Synthesize Beta-Acyl Allyl Methyl Sulfones Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized conditions described in the patent to ensure maximum yield and reproducibility. The process is designed to be operationally simple, utilizing standard glassware and heating equipment without the need for specialized high-pressure reactors or inert gas manifolds beyond standard nitrogen protection. The key to success lies in the precise stoichiometric balance of the Selectfluor promoter and the hydroxylamine salt, as well as the maintenance of anhydrous conditions to prevent hydrolysis of the reactive intermediates. Operators should be prepared to manage the exothermic nature of the initial mixing and ensure adequate stirring to maintain homogeneity in the toluene-DMSO biphasic-like system. Detailed standardized synthetic steps for the preparation of these valuable intermediates are provided in the guide below, ensuring that technical teams can replicate the high performance observed in the patent examples.
- Charge a reaction vessel with Selectfluor (3 equiv) and NH2OH·HCl (1 equiv) under nitrogen atmosphere.
- Add the aryl methyl ketone substrate, ultra-dry DMSO, and toluene (MePh) in a 1: 7 solvent ratio.
- Stir the mixture at 120°C for 10 hours, then cool, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free synthesis protocol offers profound advantages for procurement managers and supply chain heads who are tasked with minimizing costs and mitigating supply risks. The most immediate benefit is the drastic simplification of the raw material portfolio; by relying on commodity chemicals like acetophenones and DMSO rather than specialized organometallic reagents or custom-synthesized vinyl sulfones, companies can leverage existing bulk purchasing agreements and reduce inventory complexity. The elimination of transition metal catalysts translates directly into substantial cost savings by removing the need for expensive metal scavengers and the associated waste disposal fees for heavy metal contaminated streams. Moreover, the one-pot nature of the reaction reduces the number of unit operations, which lowers energy consumption and labor costs associated with intermediate isolation and handling. This efficiency gain allows for a more agile response to market demand fluctuations, as production cycles can be shortened significantly compared to multi-step traditional routes.
- Cost Reduction in Manufacturing: The economic impact of removing precious metal catalysts from the process cannot be overstated, as it eliminates a major variable cost driver and the capital expenditure associated with metal recovery systems. By utilizing Selectfluor, a stable and commercially available solid reagent, the process avoids the price volatility often seen with palladium or rhodium-based catalysts. Additionally, the high atom economy of using DMSO as a reagent means less waste is generated per kilogram of product, leading to lower environmental compliance costs and reduced raw material consumption. The simplified purification process, often requiring only standard column chromatography or crystallization, further reduces the consumption of silica gel and solvents, contributing to a leaner and more cost-effective manufacturing operation overall.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials that are produced on a massive global scale, minimizing the risk of shortages due to geopolitical issues or single-source supplier failures. Unlike complex organometallic reagents that may have long lead times and strict storage requirements, the reagents for this process are stable and easy to transport, allowing for flexible inventory management strategies. The robustness of the reaction conditions, which tolerate a wide range of substrates without needing bespoke optimization for each new derivative, ensures consistent output quality even when switching between different batches of raw materials. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the absence of hazardous pyrophoric reagents or high-pressure hydrogenation steps, making it safer for large-scale implementation in multipurpose chemical plants. The reduced generation of heavy metal waste aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals, simplifying the permitting process for new production lines. The use of toluene and DMSO, while requiring proper handling, is well-understood in the industry with established recycling protocols, further enhancing the green chemistry profile of the manufacture. This combination of safety, scalability, and environmental friendliness makes the technology an attractive candidate for long-term strategic partnerships and contract development manufacturing agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, derived directly from the detailed specifications and examples provided in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this route for their own supply chains. Understanding these specifics helps in making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the advantages of this synthesis method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts like Iron(III), thereby removing the costly and complex heavy metal removal steps required for pharmaceutical grade intermediates.
Q: Does this reaction tolerate diverse functional groups on the acetophenone substrate?
A: Yes, the protocol demonstrates impressive functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and cyano substituents.
Q: What is the role of DMSO in this cross-coupling reaction?
A: Dimethyl sulfoxide (DMSO) serves a dual function in this cascade reaction, acting simultaneously as both the vinyl donor and the methyl sulfone donor to construct the complex sulfone skeleton in a single pot.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Acyl Allyl Methyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for producing high-value pharmaceutical intermediates and specialty chemicals. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this Selectfluor-promoted reaction, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to leveraging this advanced chemistry to deliver superior value to our clients, combining technical expertise with operational excellence to drive innovation in the fine chemical sector.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific target molecules. By collaborating with our technical procurement team, you can access specific COA data and comprehensive route feasibility assessments that demonstrate how this technology can optimize your supply chain. Whether you require small quantities for clinical trials or metric tons for commercial launch, our dedicated support ensures that your project timelines are met with precision and reliability. Reach out today to explore how we can integrate this cutting-edge synthesis method into your production strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
